Get your patient on Ibtrozi - Taletrectinib capsule (Taletrectinib)
Ibtrozi - Taletrectinib capsule prescribing information
INDICATIONS AND USAGE
IBTROZI ® (taletrectinib) is indicated for the treatment of adult patients with locally advanced or metastatic ROS1 -positive non-small cell lung cancer (NSCLC) [see Dosage and Administration (2.1 )] .
DOSAGE AND ADMINISTRATION
- Select patients for the treatment of locally advanced or metastatic NSCLC based on the presence of ROS1 rearrangement(s). (2.1 )
- Recommended Dosage: 600 mg orally once daily on an empty stomach (no food intake at least 2 hours before and 2 hours after taking IBTROZI). (2.3 )
- Continue treatment until disease progression or unacceptable toxicity. (2.3 )
Patient Selection
Select patients for the treatment of locally advanced or metastatic NSCLC with IBTROZI based on the presence of ROS1 rearrangement(s) in tumor specimens [see Clinical Studies (14.1 )] .
An FDA-approved test to detect ROS1 rearrangement(s) for selecting patients for treatment with IBTROZI is not currently available.
Recommended Testing and Evaluation Before Initiating IBTROZI
Recommended Dosage and Administration
- The recommended dosage of IBTROZI is 600 mg orally once daily on an empty stomach (no food intake at least 2 hours before and 2 hours after taking IBTROZI) [see Clinical Pharmacology (12.2 , 12.3 )] until disease progression or unacceptable toxicity.
- Take IBTROZI at approximately the same time each day. Swallow IBTROZI capsules whole. Do not open, chew, crush, or dissolve the capsule prior to swallowing.
- Avoid food or drink containing grapefruit during treatment with IBTROZI.
- Minimize sun exposure and use sun protection, including broad-spectrum sunscreen, during treatment with IBTROZI and for at least 5 days after discontinuation [see Adverse Reactions (6.1 )] .
Missed Dose
- If a dose is missed, take the next dose at its scheduled time on the following day.
Vomiting
- If vomiting occurs at any time after taking a dose, take the next dose at its scheduled time on the following day.
Dosage Modifications for Adverse Reactions
The recommended dosage reductions for the management of adverse reactions are provided in Table 1 .
| Dosage Reduction | Recommended Dosage |
| First Dose Reduction | 400 mg once daily |
| Second Dose Reduction | 200 mg once daily |
| Permanently discontinue IBTROZI capsules in patients unable to tolerate 200 mg once daily. | |
The recommended dosage modifications of IBTROZI for the management of adverse reactions are provided in Table 2 .
| Adverse Reaction | Severity• | Dosage Modification |
| Hepatotoxicity (Elevation of ALT or AST) [see Warnings and Precautions (5.1 )] | Grade 3 (>5 - 20 × ULN) | Withhold IBTROZI until recovery to Grade ≤1 or baseline.
Recurrence:
|
| Grade 4 (>20 × ULN) | Withhold IBTROZI until recovery to Grade ≤1 or baseline.
Recurrence:
| |
| ALT or AST ≥3 × ULN with concurrent total bilirubin ≥2 × ULN (in the absence of cholestasis or hemolysis) | Permanently discontinue IBTROZI. | |
| ILD/pneumonitis [see Warnings and Precautions (5.2 )] | Grade 1 | Withhold IBTROZI if ILD/pneumonitis occurs or is suspected until recovery to Grade 0 or baseline.
Recurrence:
|
| Grade 2 | Withhold IBTROZI if ILD/pneumonitis occurs or is suspected until recovery to Grade 0 or baseline.
Recurrence:
| |
| Grade 3 or 4 | Permanently discontinue IBTROZI. | |
| QTc Interval Prolongation [see Warnings and Precautions (5.3 )] | Grade 2 (QTc interval 481-500 msec) | Withhold IBTROZI until recovery to Grade ≤1 or baseline.
|
Abbreviations: ALT = alanine aminotransferase; AST = aspartate aminotransferase; ULN = Upper limit of normal | ||
•Graded per National Cancer Institute Common Terminology Criteria for Adverse Events (NCI CTCAE), version 5.0. | ||
| Adverse Reaction | Severity• | Dosage Modification |
| Grade 3 (QTc interval ≥501 msec or QTc interval increase of >60 msec from baseline) | Withhold IBTROZI until recovery to Grade ≤1 or baseline.
| |
| Grade 4 (Torsade de pointes; polymorphic ventricular tachycardia; signs/symptoms of serious arrhythmia) | Permanently discontinue IBTROZI. | |
| Hyperuricemia [see Warnings and Precautions (5.4 )] | Grade 3 or 4 | Withhold IBTROZI until improvement of signs or symptoms.
|
| Creatine Phosphokinase Elevation [see Warnings and Precautions (5.5 )] | CPK elevation >5 times ULN | Withhold until recovery to baseline or ≤2.5 times ULN, then resume at same dose. |
| CPK elevation >10 times ULN or second occurrence of CPK elevation of >5 times ULN | Withhold until recovery to baseline or ≤2.5 times ULN, then resume at a reduced dose. | |
| Other Adverse Reactions [see Adverse Reactions (6.1 )] | Grade 3 | Withhold IBTROZI until recovery to Grade ≤1 or baseline.
Recurrence:
|
| Grade 4 | Withhold IBTROZI until recovery to Grade ≤1 or baseline. Resume IBTROZI at reduced dose or permanently discontinue as clinically indicated. Recurrence:
| |
DOSAGE FORMS AND STRENGTHS
Capsules: 200 mg, immediate release, white, opaque, size 0, hard capsules filled with white to light yellow powder, imprinted with “TAL” and “200” in blue ink on the body of the capsule. Each capsule contains 272 mg of taletrectinib adipate equivalent to 200 mg of taletrectinib.
USE IN SPECIFIC POPULATIONS
- Lactation: Advise not to breastfeed. (8.2 )
Pregnancy
Risk Summary
Based on literature reports in humans with congenital mutations leading to changes in TRK signaling, findings from animal studies, and its mechanism of action [see Clinical Pharmacology (12.1 )] , IBTROZI can cause fetal harm when administered to a pregnant woman. Limited data from case reports with IBTROZI used in pregnant women are insufficient to inform a drug-associated risk of adverse developmental outcomes. In animal reproduction studies, oral administration of taletrectinib to pregnant rats and rabbits during the period of organogenesis resulted in embryo-fetal mortalities and structural abnormalities at exposures that were below or equal to the human exposure based on AUC at the recommended dose ( see Data ) . Advise pregnant women of the potential risk to a fetus.
In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Data
Human Data
Published reports of individuals with congenital mutations in TRK pathway proteins suggest that decreases in TRK-mediated signaling are correlated with obesity, developmental delays, cognitive impairment, insensitivity to pain, and anhidrosis.
Animal Data
In an embryo-fetal development study, taletrectinib was administered orally to pregnant rats during the period of organogenesis from gestation day 6 to 17 at doses of 10, 30, and 100 mg/kg/day. Taletrectinib caused fetal abnormalities including abnormal ossification of the pelvis at 100 mg/kg/day (1.3 times the human exposure based on AUC at the recommended dose).
In an embryo-fetal development study, taletrectinib was administered orally to pregnant rabbits during the period of organogenesis from gestation day 6 to 19 at doses of 15, 30, and 90 mg/kg/day. Maternal lethality and increased total pregnancy loss were observed at doses ≥15 mg/kg/day (≥0.04 times the human exposure based on AUC at the recommended dose). Fetal malformations, including undeveloped or no development of eyes, nose, and mouth, ventricular malformations, thoracic vascular malformations, and skull malformations were observed at 30 mg/kg/day (0.1 times the human exposure based on AUC at the recommended dose).
Lactation
Risk Summary
There are no data on the presence of taletrectinib or its metabolites in human milk or their effects on a breastfed child or on milk production. Because of the potential for adverse reactions in breastfed children, advise women not to breastfeed during treatment with IBTROZI and for 3 weeks after the last dose.
Females and Males of Reproductive Potential
IBTROZI can cause fetal harm when administered to a pregnant woman [see Use in Specific Populations (8.1 )].
Pregnancy Testing
Verify the pregnancy status of females of reproductive potential prior to initiating IBTROZI [see Use in Specific Populations (8.1 )] .
Contraception
Females
Advise females of reproductive potential to use effective contraception during treatment with IBTROZI and for 3 weeks after the last dose [see Nonclinical Toxicology (13.1 )] .
Males
Advise male patients with female partners of reproductive potential to use effective contraception during treatment with IBTROZI and for 3 weeks after the last dose [see Nonclinical Toxicology (13.1 )] .
Infertility
Based on findings in animals, IBTROZI may impair fertility in males and females [see Nonclinical Toxicology 13.1 ]. The effects on animal fertility were reversible.
Pediatric Use
The safety and effectiveness of IBTROZI in pediatric patients has not been established.
Geriatric Use
Of the 352 patients who received IBTROZI 600 mg orally once daily, 88 (25%) patients were 65 years of age and older, and 14 (4%) patients were 75 years of age and older. There were no clinically meaningful differences in safety and efficacy between patients younger than 65 years of age and patients 65 years of age or older.
CONTRAINDICATIONS
None.
WARNINGS AND PRECAUTIONS
- Hepatotoxicity: Monitor liver function tests prior to initiating, every 2 weeks during the first 2 months of treatment, then monthly thereafter as clinically indicated, with more frequent testing in patients who develop transaminase elevations. Based on severity and resolution, withhold and then resume at reduced dose, or permanently discontinue. (2.4 , 5.1 )
- Interstitial Lung Disease (ILD)/Pneumonitis: Monitor patients for new or worsening pulmonary symptoms indicative of ILD/pneumonitis. Immediately withhold in patients with suspected ILD/pneumonitis. Based on severity and resolution, resume at the same or reduced dose, or permanently discontinue. (2.4 , 5.2 )
- QTc Interval Prolongation: Monitor ECG and electrolytes prior to initiating and periodically during treatment. Based on severity and resolution, withhold and then resume at same or reduced dose, or permanently discontinue. (2.4 , 5.3 )
- Hyperuricemia: Monitor serum uric acid levels prior to initiating and periodically during treatment. Initiate treatment with urate-lowering medications as clinically indicated. Withhold and resume at same or reduced dose or permanently discontinue based on severity. (2.4 , 5.4 )
- Myalgia with Creatine Phosphokinase (CPK) Elevation: Monitor serum CPK levels during treatment in patients reporting unexplained muscle pain, tenderness, or weakness. Based on severity, withhold and resume at same or reduced dose upon improvement. (2.4 , 5.5 )
- Skeletal Fractures: Promptly evaluate patients with signs or symptoms (e.g., pain, changes in mobility, deformity) of fractures. (5.6 )
- Embryo-Fetal Toxicity: Can cause fetal harm. Advise patients of the potential risk to a fetus and to use effective contraception. (5.7 )
Hepatotoxicity
IBTROZI can cause hepatotoxicity, including drug-induced liver injury and fatal adverse reactions.
In the pooled safety population [see Adverse Reactions (6.1 )], based on laboratory values, 88% of patients treated with IBTROZI experienced increased aspartate aminotransferase (AST), including 10% Grade 3 or 4. Increased alanine aminotransferase (ALT) occurred in 85% of patients treated with IBTROZI, including 13% Grade 3 or 4. The median time to first onset of AST or ALT elevation was 15 days (range: 3 days to 20.8 months).
Increased AST or ALT each led to dose interruption in 7% of patients. Increased AST and ALT leading to dose reduction occurred in 5% and 9% of patients, respectively. Increased AST and ALT each led to permanent discontinuation of IBTROZI in 0.3% of patients. Other liver-related adverse reactions leading to permanent discontinuation of IBTROZI were hepatotoxicity (0.6% of patients) and increased bilirubin (0.3% of patients).
Concurrent elevations in AST or ALT ≥3 times the upper limit of normal (ULN) and total bilirubin ≥2 times the ULN, with normal alkaline phosphatase, occurred in 2 (0.6%) patients treated with IBTROZI. Fatal liver events occurred in 2 (0.6%) patients.
Monitor liver function tests (AST, ALT, and bilirubin) prior to administration of IBTROZI, every 2 weeks during the first 2 months of treatment, and then monthly thereafter as clinically indicated with more frequent testing in patients who develop transaminase elevations. Withhold, then resume at reduced dose upon improvement, or permanently discontinue IBTROZI based on severity [see Dosage and Administration (2.4 )] .
Interstitial Lung Disease/Pneumonitis
IBTROZI can cause severe, life-threatening, or fatal interstitial lung disease (ILD) or pneumonitis.
In the pooled safety population [see Adverse Reactions (6.1 )], interstitial lung disease (ILD)/pneumonitis occurred in 2.3% of patients treated with IBTROZI, including Grade 3 or 4 in 1.1% of patients. The median time to first onset of ILD/pneumonitis was 3.8 months (range: 12 days to 11.8 months).
ILD/pneumonitis led to dose interruption of IBTROZI in 1.1% of patients. ILD/pneumonitis required dose reduction in 0.6% of patients and permanent discontinuation of IBTROZI in 0.6% of patients. One fatal ILD case occurred in a patient who received 400 mg once daily dose of IBTROZI.
Monitor patients for new or worsening pulmonary symptoms indicative of ILD/pneumonitis. Immediately withhold IBTROZI in patients with suspected ILD/pneumonitis. Withhold, then reduce the dose or permanently discontinue IBTROZI if Grade ≥2 ILD/pneumonitis is confirmed [see Dosage and Administration (2.4 )] .
QTc Interval Prolongation
IBTROZI can cause QTc interval prolongation, which can increase the risk for ventricular tachyarrhythmias (e.g., torsades de pointes) or sudden death. IBTROZI prolongs the QTc interval in a concentration-dependent manner [see Clinical Pharmacology (12.2 )] .
In the pooled safety population [see Adverse Reactions (6.1 )], of the 351 patients who underwent at least one post baseline ECG assessment, 13% experienced an increase in QTcF of >60 msec compared to baseline after receiving IBTROZI and 2.6% had an increase in QTcF to >500 msec. Overall, 3.4% of patients had Grade ≥3 QTc interval prolongation. The median time from the first dose of IBTROZI to the onset of ECG QT prolongation was 22 days (range: 1 day to 38.7 months). QTc prolongation led to dose interruption and dose reduction, each in 2.8% of patients treated with IBTROZI.
Monitor ECGs and electrolytes prior to administration of IBTROZI, and then periodically thereafter as clinically indicated during treatment. Adjust the frequency of monitoring based on risk factors such as known long QT syndromes, clinically significant bradyarrhythmias, severe or uncontrolled heart failure, and concomitant medications associated with QTc interval prolongation.
Significant prolongation of the QTc interval may occur when IBTROZI is taken with food, strong and moderate CYP3A inhibitors, and/or drugs with a known potential to prolong QTc. Administer IBTROZI on an empty stomach. Avoid coadministration of IBTROZI with strong and moderate CYP3A inhibitors and/or drugs with a known potential to prolong QTc [see Dosage and Administration (2.3 ), Drug Interactions (7.1 , 7.2 )] .
Withhold, then resume at the same or reduced dose, or permanently discontinue IBTROZI based on severity [see Dosage and Administration (2.4 )] .
Hyperuricemia
IBTROZI can cause hyperuricemia.
In the pooled safety population [see Adverse Reactions (6.1 )], 14% of patients treated with IBTROZI experienced hyperuricemia reported as an adverse reaction, with 16% of these patients requiring urate-lowering medication without pre-existing gout or hyperuricemia. Hyperuricemia Grade ≥3 occurred in one patient. The median time to first onset of hyperuricemia was 2.1 months (range: 7 days to 35.8 months). Hyperuricemia leading to dose interruption occurred in 0.3% of patients.
Monitor serum uric acid levels prior to administration of IBTROZI and periodically during treatment. Initiate treatment with urate-lowering medications as clinically indicated. Withhold, then resume at the same or reduced dose, or permanently discontinue IBTROZI based on severity [see Dosage and Administration (2.4 )].
Myalgia with Creatine Phosphokinase Elevation
IBTROZI can cause myalgia with or without creatine phosphokinase (CPK) elevation.
In the pooled safety population [see Adverse Reactions (6.1 )], myalgia occurred in 10% of patients treated with IBTROZI. The median time to first onset of myalgia was 11 days (range: 2 days to 10 months).
Concurrent myalgia with increased CPK within a 7-day time period was observed in 0.9% of patients. IBTROZI was interrupted in one patient (0.3%) with myalgia, who also presented with concurrent CPK elevation.
Advise patients to report any unexplained muscle pain, tenderness, or weakness. Monitor serum CPK levels during IBTROZI treatment every 2 weeks during the first month of treatment and then as clinically indicated in patients reporting unexplained muscle pain, tenderness, or weakness. Withhold, then resume at the same or reduced dose upon improvement [see Dosage and Administration (2.4 )].
Skeletal Fractures
IBTROZI can increase the risk of fractures. ROS1 inhibitors as a class have been associated with skeletal fractures.
In the pooled safety population [see Adverse Reactions (6.1 )], 3.4% of patients experienced fractures including 1.4% Grade 3. Some fractures occurred in the setting of an accidental fall or other predisposing factors such as osteoporosis, bone metastasis, and age-related degenerative conditions. Fractures involved the ribs (1.4%), spine (0.9%), femur (0.6%), humerus (0.3%), and acetabulum (0.3%). The median time to first onset of fracture was 10.7 months (range: 26 days to 29.1 months). Dose interruption occurred in 0.3% of patients.
Promptly evaluate patients with signs or symptoms (e.g., pain, changes in mobility, deformity) of fractures. There are no data on the effects of IBTROZI on healing of known fractures and risk of future fractures.
Embryo-Fetal Toxicity
Based on literature reports in humans with congenital mutations leading to changes in tropomyosin receptor kinase (TRK) signaling, findings from animal studies and its mechanism of action, IBTROZI can cause fetal harm when administered to a pregnant woman.
In animal reproduction studies, oral administration of taletrectinib to pregnant rats during the period of organogenesis caused structural abnormalities. Oral administration of taletrectinib to pregnant rabbits during the period of organogenesis resulted in embryo-fetal mortalities and structural abnormalities. Findings in both species occurred at doses resulting in exposures below or equal to the human exposure based on AUC at the recommended dose.
Advise pregnant women and females of reproductive potential of the potential risk to a fetus. Advise females of reproductive potential to use effective contraception during treatment with IBTROZI and for 3 weeks after the last dose. Advise male patients with female partners of reproductive potential to use effective contraception during treatment with IBTROZI and for 3 weeks after the last dose [see Use in Specific Populations (8.1 , 8.3 )] .
ADVERSE REACTIONS
The following adverse reactions are discussed in greater detail in the WARNINGS AND PRECAUTIONS section:
- Hepatotoxicity [see Warnings and Precautions (5.1 )]
- Interstitial Lung Disease/Pneumonitis [see Warnings and Precautions (5.2 )]
- QTc Interval Prolongation [see Warnings and Precautions (5.3 )]
- Hyperuricemia [see Warnings and Precautions (5.4 )]
- Myalgia with Creatine Phosphokinase Elevation [see Warnings and Precautions (5.5 )]
- Skeletal fractures [see Warnings and Precautions (5.6 )]
Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The pooled safety population described in the WARNINGS AND PRECAUTIONS section and below reflects exposure to IBTROZI as a single agent at 600 mg orally once daily until disease progression or unacceptable toxicity in 352 patients with ROS1 -positive NSCLC (N=337) and other solid tumors (N=15). Among the 352 patients who received IBTROZI, 68% were exposed for at least 6 months, and 47% were exposed for greater than 1 year. In this pooled safety population, the most common (≥20%) adverse reactions were diarrhea, nausea, vomiting, dizziness, rash, constipation, and fatigue. The most common (≥2%) Grade 3 or 4 laboratory abnormalities were increased ALT, increased AST, decreased neutrophils, increased creatine phosphokinase, decreased lymphocytes, increased magnesium, decreased hemoglobin, and increased triglycerides.
Locally Advanced or Metastatic ROS1-Positive NSCLC
The safety of IBTROZI was evaluated in the TRUST-I and TRUST-II studies [see Clinical Studies (14.1 )]. Key eligibility criteria were histologically confirmed, locally advanced or metastatic, ROS1 -positive NSCLC, ECOG performance status ≤1, and measurable disease per RECIST v1.1. Patients received IBTROZI as a single agent at 600 mg orally once daily until disease progression or unacceptable toxicity. Among patients who received IBTROZI, 68% were exposed for 6 months or longer and 47% were exposed for greater than one year.
The median age of patients who received IBTROZI was 56 years (range: 26 to 83); 56% female; 76% Asian, 15% White, 0.6% Black or African American, 8% unknown or other races; and 1.8% were of Hispanic or Latino ethnicity.
Serious adverse reactions occurred in 31% of patients who received IBTROZI. Serious adverse reactions in ≥2% of patients included pneumonia (7%), pleural effusion (4.7%), and hepatotoxicity (2.4%). Fatal adverse reactions occurred in 18 (5%) patients who received IBTROZI, including pneumonia (2.4%), multiple organ dysfunction syndrome (0.6%), hepatotoxicity (0.6%), cardiac arrest (0.6%), cardiac failure (0.3%), cardiopulmonary failure (0.3%), respiratory failure (0.3%), and death not otherwise specified (0.3%).
Permanent discontinuation of IBTROZI was required in 7% of patients due to adverse reactions. Adverse reactions resulting in permanent discontinuation of IBTROZI in ≥2 patients were pneumonia, ILD, and hepatotoxicity.
Dosage interruptions of IBTROZI due to an adverse reaction occurred in 41% of patients. Adverse reactions which required dosage interruption in ≥5% of patients included increased AST and increased ALT.
Dose reductions of IBTROZI due to an adverse reaction occurred in 29% of patients. Adverse reactions that required dosage reductions in ≥5% of patients included increased ALT and increased AST.
Table 3 summarizes the adverse reactions in this population.
1 Based on NCI CTCAE version 5.0 a Includes enterocolitis | |||
b Includes vertigo, and vertigo positional | |||
c Includes dysesthesia, hypoesthesia, neuralgia, paresthesia, and peripheral sensory neuropathy d Includes ageusia | |||
e Includes dermatitis, dermatitis acneiform, drug eruption, eczema, eyelid rash, palmar-plantar erythrodysesthesia syndrome, rash | |||
maculo-papular, rash papular, skin exfoliation, and drug reaction with eosinophilia and systemic symptoms (DRESS) | |||
f Includes asthenia | |||
g Includes productive cough | |||
| Adverse Reaction 1 | IBTROZI N=337 | ||
| All Grades (%) | Grade 3 or 4 (%) | ||
| Gastrointestinal Disorders | |||
| Diarrhea a | 64 | 2.1 | |
| Nausea | 47 | 1.5 | |
| Vomiting | 43 | 1.5 | |
| Constipation | 21 | 0 | |
| Nervous System Disorders | |||
| Dizziness b | 22 | 0.3 | |
| Peripheral neuropathy c | 17 | 0.3 | |
| Dysgeusia d | 15 | 0 | |
| Skin and Subcutaneous Tissue | |||
| Rash e | 22 | 1.8 | |
| General Disorders | |||
| Fatigue f | 20 | 0.9 | |
| Cardiac | |||
| Electrocardiogram QT prolonged | 19 | 3.6 | |
| Metabolism and Nutritional | |||
| Decreased appetite | 16 | 0.3 | |
| Respiratory, thoracic and mediastinal disorders | |||
| Cough g | 16 | 0 | |
Clinically relevant adverse reactions in <15% of patients receiving IBTROZI were pneumonia, eye disorders, myalgia, skeletal fracture, ILD/pneumonitis, dermatologic adverse reactions including drug reaction with eosinophilia and systemic symptoms (DRESS), and photosensitivity reactions.
Table 4 summarizes the laboratory abnormalities.
Abbreviations: ALT = alanine aminotransferase; AST = aspartate aminotransferase | ||
1 Based on NCI CTCAE version 5.0 | ||
2 The denominator used to calculate the rate varied from 149 to 336 based on the number of patients with a baseline value and at least one post-treatment value. | ||
| Laboratory Abnormality 1 | IBTROZI 2 | |
| All Grades (%) | Grade 3 or 4 (%) | |
| Hematology | ||
| Hemoglobin decreased | 48 | 3.6 |
| Lymphocytes decreased | 38 | 4.8 |
| Neutrophils decreased | 25 | 5 |
| Chemistry | ||
| AST increased | 87 | 10 |
| ALT increased | 85 | 13 |
| Creatine phosphokinase increased | 53 | 5 |
| Cholesterol increased | 41 | 0 |
| Triglycerides increased | 41 | 2.5 |
| Creatinine increased | 39 | 0.3 |
| Uric acid increased | 38 | 0 |
| Gamma glutamyl transferase increased | 36 | 1.8 |
| Alkaline phosphatase increased | 30 | 0 |
| Calcium decreased | 28 | 1.8 |
| Albumin decreased | 25 | 0.9 |
| Bilirubin increased | 24 | 0.6 |
| Potassium increased | 21 | 1.2 |
| Sodium increased | 20 | 0.9 |
DRUG INTERACTIONS
- Strong and Moderate CYP3A inhibitors : Avoid concomitant use. (7.1 )
- Strong and Moderate CYP3A inducers: Avoid concomitant use. (7.1 )
- Gastric Acid Reducing Agents: Avoid concomitant use with proton pump inhibitors (PPIs) and H2 receptor antagonists. If an acid-reducing agent cannot be avoided, administer IBTROZI 2 hours before or 2 hours after taking a locally acting antacid. (7.1 )
- Drugs That Prolong the QTc Interval: Avoid concomitant use. (7.2 )
Effects of Other Drugs on IBTROZI
Strong and Moderate CYP3A Inhibitors
Avoid concomitant use with strong or moderate CYP3A inhibitors.
Taletrectinib is a CYP3A substrate. Concomitant use of IBTROZI with a strong or moderate CYP3A inhibitor increases taletrectinib exposure [see Clinical Pharmacology (12.3 )] , which may increase the risk of IBTROZI adverse reactions.
Strong and Moderate CYP3A Inducers
Avoid concomitant use with strong or moderate CYP3A inducers.
Concomitant use of IBTROZI with a strong or moderate CYP3A inducer decreases taletrectinib exposure [see Clinical Pharmacology (12.3 )] , which may reduce the effectiveness of IBTROZI.
Gastric Acid Reducing Agents
Avoid concomitant use with proton pump inhibitors (PPI) and H2 receptor antagonists. Administer locally acting antacids at least 2 hours before or 2 hours after taking IBTROZI [see Clinical Pharmacology (12.3 )].
Concomitant use of a proton pump inhibitor decreases taletrectinib exposure [see Clinical Pharmacology (12.3 )], which may reduce the effectiveness of IBTROZI.
Drugs That Prolong the QTc Interval
Avoid concomitant use of IBTROZI with other drug(s) with a known potential to prolong the QTc interval, such as antiarrhythmic drugs. If concomitant use cannot be avoided, adjust the frequency of monitoring as recommended [see Warnings and Precautions (5.3 ), Clinical Pharmacology (12.2 )]. Withhold IBTROZI if the QTc interval is >500 msec or the change from baseline is >60 msec [see Dosage and Administration (2.4 )].
IBTROZI causes QTc interval prolongation [see Warnings and Precautions (5.3 ), Clinical Pharmacology (12.2 )] . Concomitant use of IBTROZI with other drugs known to prolong the QTc interval may increase the risk of QTc interval prolongation.
DESCRIPTION
IBTROZI (taletrectinib) capsules contain taletrectinib adipate, a kinase inhibitor for oral use. The molecular formula for taletrectinib adipate is C 23 H 24 FN 5 O•C 6 H 10 O 4 and the molecular weight is 551.61 g/mol. The chemical name is 3-{4-[(2 R )-2-aminopropoxy]phenyl}- N -[(1 R )-1-(3-fluorophenyl)ethyl]imidazo [1,2 b ]pyridazin-6-amine monoadipate. The chemical structure of taletrectinib adipate is as follows:
Figure 1: Structure of Taletrectinib Adipate
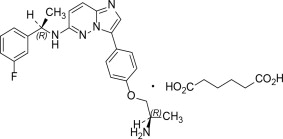
(R) Indicates chiral center
Taletrectinib adipate is a white to yellowish powder and is practically insoluble or insoluble in water, anhydrous ethanol, isopropanol, acetonitrile, and n-hexane, sparingly soluble in methanol, and slightly soluble in N-methyl pyrrolidone. The aqueous solubility is pH dependent and decreases with increasing pH. The pKa1 is 5.39 and pKa2 is 8.65.
IBTROZI (taletrectinib) capsules for oral use are supplied as immediate-release hard capsules containing 272 mg of taletrectinib adipate (equivalent to 200 mg of taletrectinib free base). Inactive ingredients are colloidal silicon dioxide, low-substituted hydroxypropyl cellulose, mannitol, pregelatinized starch, and sodium stearyl fumarate.
The white opaque capsule shell contains hypromellose and titanium dioxide. The blue printing ink (SB-6008) contains butyl alcohol, dehydrated alcohol, FD&C Blue#1 Aluminum Lake, isopropyl alcohol, propylene glycol, shellac, strong ammonia solution, and titanium dioxide.
CLINICAL PHARMACOLOGY
Mechanism of Action
Taletrectinib is an inhibitor of tyrosine kinase ROS1, including ROS1 resistance mutations. Taletrectinib also showed inhibitory effects on tropomyosin receptor kinases (TRKs) TRKA, TRKB, and TRKC.
Fusion proteins that include ROS1 domains can drive tumorigenic potential through hyperactivation of downstream signaling pathways leading to unconstrained cell proliferation. Taletrectinib inhibited growth of cancer cells expressing ROS1 fusion genes and mutations.
In mice subcutaneously implanted with tumors harboring ROS1 fusions, including the G2032R mutation, administration of taletrectinib resulted in tumor growth inhibition. Taletrectinib had anticancer activity in an intracranial NSCLC xenograft model harboring a ROS1 fusion.
Pharmacodynamics
Exposure-response relationship
Higher taletrectinib exposure is associated with an increased risk of Grade ≥3 increased AST/ALT.
Taletrectinib exposure-response relationships for efficacy and the time course of pharmacodynamic response have not been fully characterized.
Cardiac Electrophysiology
The largest mean increase in the QTc interval was 12.8 msec (upper confidence interval of 15.4 msec) at the mean steady-state maximum concentration (C max,ss ) after administration of IBTROZI 600 mg once daily. The increase in the QTc internal was concentration-dependent [see Warnings and Precautions (5.3 )]. At plasma concentrations achieved with administration of IBTROZI 600 mg once daily with high fat food, the predicted increase in the QTc interval is 20.5 (16.3, 24.7) msec.
Pharmacokinetics
Taletrectinib pharmacokinetics (PK) were characterized at steady state for the approved recommended dosage and the PK parameters presented below are as mean (CV%), unless otherwise specified.
Taletrectinib maximum concentration (C max ) is 476 (36%) ng/mL and systemic exposure (AUC) is 9,649 (36%) ng•h/mL. Taletrectinib C max and AUC increase in a dose proportional manner over the dose range of 50 mg to 1200 mg (0.08 to 2 times the approved recommended dosage). Taletrectinib accumulation is approximately 4-fold and steady state reached within 7 days.
Absorption
Taletrectinib time to maximum plasma concentration (Tmax) is 2 to 6 hours.
Effect of Food
Both taletrectinib AUC and Cmax increased by 1.5-fold following administration with high-fat food (1000 calories, 50% fat).
Distribution
The estimated apparent (oral) volume of distribution is 9,820 L.
Taletrectinib in vitro plasma protein binding is concentration-dependent and decreases with increasing taletrectinib concentrations (from 96% at 100 ng/mL to 93% at 10,000 ng/mL). The blood-to-plasma ratio is 1.3 to 1.4 in vitro.
Elimination
Taletrectinib effective half-life is approximately 66 hours with an apparent (oral) clearance of 63 L/h (36%).
Metabolism
Taletrectinib is eliminated by CYP450 and non-CYP450 (i.e., sulfation and acetylation) metabolism. CYP3A and, to a lesser extent, CYP2C8 and CYP2C9 are the CYP450 enzymes involved in taletrectinib metabolism.
Excretion
Following a single oral dose of radiolabeled taletrectinib 200 mg, 75% of the dose was recovered in feces (15% as unchanged) and 11% in urine (2.9% as unchanged).
Specific Populations
No clinically significant differences in the pharmacokinetics of taletrectinib were observed based on age (18 to 80 years), sex, mild to moderate renal impairment (eGFR 30 to 89 mL/min), or mild hepatic impairment (total bilirubin >1 to 1.5 times ULN or AST >ULN). The effect of moderate (total bilirubin >1.5 to 3 times ULN with any AST) or severe (total bilirubin >3 x ULN with any AST) hepatic impairment, severe renal impairment (eGFR<30 mL/min), or dialysis on taletrectinib pharmacokinetics is unknown.
A 30% increase in taletrectinib exposure (AUC) was observed in Asian patients compared to White patients. No clinically significant differences in the AUC were observed between White and Black patients.
Drug Interaction Studies
Clinical Studies and Model-Informed Approaches
Strong CYP3A and P-gp Inhibitors : Taletrectinib C max increased by 1.8-fold and AUC by 3.3-fold following concomitant administration of itraconazole (strong CYP3A and P-gp inhibitor) 200 mg daily.
Moderate CYP3A Inhibitors : Concomitant use with moderate CYP3A inhibitors (fluconazole, erythromycin, or verapamil) is predicted to increase taletrectinib AUC up to 2.6-fold and C max up to 1.5-fold.
Strong CYP3A Inducers and P-gp Inducers : Taletrectinib C max decreased by 42% and AUC by 86% following concomitant administration of rifampin (strong CYP3A and P-gp inducer) 600 mg daily.
Moderate CYP3A Inducers : Concomitant use with a moderate CYP3A inducer (efavirenz) is predicted to reduce taletrectinib AUC by 66% and its C max by 40%.
Proton Pump Inhibitors (PPIs) : Taletrectinib displays a pH-dependent aqueous solubility [see Description (11 )]. Taletrectinib C max decreased by 65% and AUC by 40% following concomitant administration of omeprazole (PPI) 40 mg daily.
P-gp substrates : No clinically significant difference in the exposure of digoxin (P-gp substrate) was observed when used concomitantly with taletrectinib.
In vitro Studies
CYP450 Enzymes : Taletrectinib inhibits CYP2C8, CYP2D6, and CYP3A. Taletrectinib induces CYP1A2 and CYP3A4.
Transporter Systems : Taletrectinib is a P-gp substrate. Taletrectinib inhibits P-gp, BCRP, OATP1B1, MATE1 and MATE2-K.
NONCLINICAL TOXICOLOGY
Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
Carcinogenicity studies with taletrectinib were not conducted.
Mutagenesis
Taletrectinib was not mutagenic in the in vitro bacterial reverse mutation (Ames) assay and additional mutagenicity tests. Taletrectinib was not clastogenic or aneugenic in the in vivo micronucleus study in rats in bone marrow and liver.
Impairment of Fertility
In a fertility and early embryonic development study in rats, male animals were treated with taletrectinib at doses of 4, 25, or 60 mg/kg/day orally for 70 days before mating and during 13 days of the mating period. Female rats, paired with treated males, were administered taletrectinib at doses of 4, 25, or 100 mg/kg/day orally for 14 to 21 days before mating, during the mating period and up to gestational Day 6. No effects on mating or fertility in males or females were observed at doses below or equal to the human exposure based on AUC at the recommended dose. Sperm morphological abnormalities were observed in males at exposures as low as 0.03 times the human exposure based on AUC at the recommended dose.
In repeat-dose toxicity studies up to 12 weeks in duration with oral administration of taletrectinib in rats, adverse effects in reproductive organs included epithelial vacuolation of the seminal vesicle in male rats and endometrial glandular epithelium vacuolation in the uterus with cervix in female rats at exposures 4 times the human exposure based on AUC at the recommended dose for both male and female rats. Findings in the reproductive organs were reversible.
CLINICAL STUDIES
Locally Advanced or Metastatic ROS1 -Positive NSCLC
The efficacy of IBTROZI was evaluated in 270 patients with ROS1 -positive locally advanced or metastatic NSCLC who received IBTROZI at a dose of 600 mg orally once daily, enrolled in two multicenter, single-arm, open-label clinical trials: TRUST-I (NCT04395677) or TRUST-II (NCT04919811). In both trials, patients were required to have histologically confirmed, locally advanced or metastatic, ROS1 -positive NSCLC, ECOG performance status of 0 or 1, and measurable disease per Response Evaluation Criteria in Solid Tumors (RECIST) v1.1. Identification of ROS1 gene fusions in tumor specimens was determined in local laboratories using next-generation sequencing (NGS), polymerase chain reaction (PCR), fluorescence in situ (FISH), or immunohistochemistry (IHC).
The major efficacy outcome measures were confirmed overall response rate (ORR) and duration of response (DOR) according to RECIST v1.1 as assessed by a blinded independent central review (BICR). Intracranial response according to modified RECIST v1.1 was assessed by BICR. Tumor assessments with imaging were performed every 6 weeks for the first 24 weeks, every 9 weeks for the following year, and every 12 weeks thereafter. The efficacy populations included 157 patients naïve to treatment with a ROS1 TKI and 113 patients who received one prior ROS1 TKI. Patients could be chemotherapy-naïve or have received prior chemotherapy for locally advanced disease.
ROS1 TKI-Naïve
TRUST-I: Among 103 patients with ROS1 TKI-naïve NSCLC treated in TRUST-I, the median age was 56 years (range: 26 to 78); 55% were female; 100% were Asian; 73% never smoked; and 81% had ECOG performance status of 1. At baseline, 91% of patients had metastatic disease; 17% of patients had CNS metastases by BICR; 96% had adenocarcinoma; and 19% of patients had prior platinum-based chemotherapy for advanced disease.
TRUST-II: Among 54 patients with ROS1 TKI-naïve NSCLC treated in TRUST-II, the median age was 57 years (range: 27 to 82); 56% were female; 65% were Asian; 22% were White; 1.9% were Black or African American; 11% were of unknown race; 1.9% were of Hispanic or Latino ethnicity; 50% never smoked; and 61% had ECOG performance status of 1. At baseline, 91% of patients had metastatic disease, 35% of patients had CNS metastases by BICR; 98% had adenocarcinoma; and 19% of patients had prior platinum-based chemotherapy for advanced disease.
Efficacy results are summarized in Table 5 .
Abbreviations: CI = Confidence interval; NR = Not reached; “+” indicating ongoing response | ||
a Median DOR not included for TRUST-II given the shorter duration of follow-up. | ||
b Based on observed duration of response | ||
| Efficacy Parameters | ROS1 TKI-Naïve N=157 | |
| TRUST-I | TRUST-II | |
| n=103 | n=54 | |
| Response Rate, % (95% CI) | 90% (83, 95) | 85% (73, 93) |
| Complete Response | 5% | 7% |
| Partial Response | 85% | 78% |
| Duration of Response (DOR) | n= 93 | n= 46 |
| Median DOR (95% CI) | NR (30.4, NR) | -- a |
| Range (months) | 1.1, 46.9+ | 1.4+, 30.4+ |
| % with DOR b ≥12 months | 72 | 63 |
Among 157 ROS1 TKI-naïve patients across TRUST-I and TRUST-II, 15 had measurable CNS metastases at baseline as assessed by BICR and had not received radiation therapy to the brain within 2 months prior to study entry; responses in intracranial lesions were observed in 11 patients.
ROS1 TKI-Pretreated
TRUST-I: Among the 66 patients with ROS1 TKI-pretreated NSCLC, the median age was 51 years (range: 31 to 77); 61% were female; 100% were Asian; 74% never smoked; and 71% had ECOG performance status of 1. At baseline, 97% of patients had metastatic disease, 42% of patients had CNS metastases by BICR; 92% had adenocarcinoma; 35% of patients had prior platinum-based chemotherapy for advanced disease and 100% had prior treatment with crizotinib.
TRUST-II: Among 47 patients with ROS1 TKI-pretreated NSCLC, the median age was 55 years (range: 27 to 79); 57% were female; 47% were Asian; 34% were White; 2.1% were Black or African American, 17% were of unknown or other races; 2.1% were Hispanic or Latino; 62% never smoked; and 55% had ECOG performance status of 1. At baseline, 98% of patients had metastatic disease; 57% of patients had CNS metastases by BICR; 98% had adenocarcinoma; 40% of patients had prior platinum-based chemotherapy for advanced disease, 79% had prior treatment with crizotinib, and 21% had prior treatment with entrectinib.
Efficacy results are summarized in Table 6 .
Abbreviations: CI = Confidence interval; “+” indicating ongoing response | ||
a Median DOR not included for TRUST-II given the shorter duration of follow-up. | ||
b Based on observed duration of response. | ||
| Efficacy Parameters | ROS1 TKI-Pretreated N=113 | |
| TRUST-I | TRUST-II | |
| n=66 | n=47 | |
| Response Rate, % (95% CI) | 52% (39, 64) | 62% (46, 75) |
| Complete Response | 0% | 11% |
| Partial Response | 52% | 51% |
| Duration of Response (DOR) | n= 34 | n= 29 |
| Median DOR (95% CI) | 13.2 (7.7, 24.9) | -- a |
| Range (months) | 1.4, 38.7+ | 1.7+, 30.4+ |
| % with DOR b ≥6 months | 74 | 83 |
| % with DOR b ≥12 months | 44 | 45 |
Among 113 ROS1 TKI-pretreated patients, across TRUST-I and TRUST-II, 24 had measurable CNS metastases at baseline as assessed by BICR and had not received radiation therapy to the brain within 2 months prior to study entry; responses in intracranial lesions were observed in 15 patients.
Among 32 patients who had re-biopsied samples tested by next-generation sequencing after failure of a prior ROS1 TKI, 15 had resistance mutations. Responses were observed in 8 of these 15 patients; all responding patients had tumors with solvent front mutation G2032R.
HOW SUPPLIED/STORAGE AND HANDLING
IBTROZI is supplied as 200 mg, Size 0 white opaque hard capsules imprinted with “TAL” and “200” in blue ink as follows:
| Packaging Configuration | NDC |
| Carton of three bottles (90 capsules total) – each bottle contains 30 capsules | 84651-200-93 – each bottle 84651-200-30 |
| Carton of one bottle containing 90 capsules | 84651-200-90 |
Store IBTROZI at 20°C to 25°C (68°F to 77°F); excursions are permitted to 15°C to 30°C (59°F to 86°F) [ see USP Controlled Room Temperature ].
Mechanism of Action
Taletrectinib is an inhibitor of tyrosine kinase ROS1, including ROS1 resistance mutations. Taletrectinib also showed inhibitory effects on tropomyosin receptor kinases (TRKs) TRKA, TRKB, and TRKC.
Fusion proteins that include ROS1 domains can drive tumorigenic potential through hyperactivation of downstream signaling pathways leading to unconstrained cell proliferation. Taletrectinib inhibited growth of cancer cells expressing ROS1 fusion genes and mutations.
In mice subcutaneously implanted with tumors harboring ROS1 fusions, including the G2032R mutation, administration of taletrectinib resulted in tumor growth inhibition. Taletrectinib had anticancer activity in an intracranial NSCLC xenograft model harboring a ROS1 fusion.