Get your patient on Indocyanine Green - Indocyanine Green And Water (Indocyanine Green And Water)
Indocyanine Green - Indocyanine Green And Water prescribing information
INDICATIONS AND USAGE
Indocyanine Green for Injection USP is indicated:
Visual assessment of blood vessels, blood flow and related tissue perfusion
For visual assessment of blood vessels, blood flow and related tissue perfusion with OLYMPUS infrared compatible endoscopic imaging system
Visual assessment of the major extrahepatic bile duct
For visual assessment of the major extrahepatic bile ducts with OLYMPUS infrared imaging endoscopic imaging system.
Viewing intra-operative blood flow in the cerebral area
For viewing intra-operative blood flow in the cerebral area with OLYMPUS infrared compatible video microscopic imaging system
DOSAGE AND ADMINISTRATION
For visual assessment of blood vessels, blood flow and related tissue perfusion (2.1 )
Under sterile conditions, the Indocyanine Green for Injection USP powder should be dissolved with the 10 mL Sterile Water for Injection, USP provided and the solution used within 6 hours after it is prepared. The usual doses of Indocyanine Green for Injection USP is 0.1 mg/kg (0.04 mL/kg) – 0.3 mg/kg (0.12 mL/kg). Immediately follow each ICG intravenous injection with a tight bolus injection of approximately 10 - 12 mL of normal saline immediately before IR imaging. Multiple administrations can be performed, up to 2 mg/kg (0.8 mL/kg) per patient.
For visual assessment of the major extrahepatic bile ducts (2.2 )
Under sterile conditions, the Indocyanine Green for Injection USP powder should be dissolved with the 10 mL Sterile Water for Injection, USP provided and the solution used within 6 hours after it is prepared. The usual doses of Indocyanine Green for Injection USP is 3.0 mg (1.2 mL). Immediately follow each ICG intravenous injection with a tight bolus injection of approximately 10 - 12 mL of normal saline at least 30 minutes prior to IR imaging. Multiple administrations can be performed, up to 2 mg/kg (0.8 mL/kg) per patient.
For viewing intra-operative blood flow in the cerebral area (2.3 )
Under sterile conditions, the Indocyanine Green for Injection USP powder should be dissolved with the 10 mL Sterile Water for Injection, USP provided and the solution used within 6 hours after it is prepared. The usual doses of Indocyanine Green for Injection USP is 0.1 mg/kg (0.04 mL/kg) – 0.3 mg/kg (0.12 mL/kg). Immediately follow each ICG intravenous injection with a tight bolus injection of approximately 10 - 12 mL of normal saline immediately before IR imaging Multiple administrations can be performed, up to 2 mg/kg (0.8 mL/kg) per patient.
Visual assessment of blood vessels, blood flow and related tissue perfusion
Under sterile conditions, the Indocyanine Green for Injection USP powder should be dissolved with the 10 mL Sterile Water for Injection, USP provided for this product, and the solution used within 6 hours after it is prepared. If a precipitate is present, discard the solution.
The patient should be weighed and the dosage for one administration should be calculated on the basis of 0.1 mg/kg (0.04 mL/kg) - 0.3 mg/kg (0.12 mL/kg) of body weight. Multiple administrations can be performed, up to 2 mg/kg (0.8 mL/kg) per patient.
ICG should be administrated immediately before IR imaging.
Item | Note |
Indocyanine Green for Injection, USP | 25 mg vials of ICG powder |
Sterile Water for Injection (for dissolving ICG) | 10 mL vial of sterile water |
Syringe (for injecting sterile water into the ICG vial) | Use the syringe whose minimum volume is 10 mL |
Syringes (for each administration) | Considering the administration volume, select the appropriate size |
Sterile normal saline (for each saline flush) | Approximately 10 – 12 mL following each ICG administration |
Syringes (for each saline flush) | Use the syringe whose minimum volume is 12 mL |
Prepare the syringes filled with the weight-scaled dose of ICG solution, and the syringes filled with 10 - 12 mL of normal saline for the tight bolus injection.
Immediately before IR imaging, administer the prepared dose of ICG solution intravenously. Immediately follow each ICG injection with a tight bolus injection of approximately 10 - 12 mL of normal saline.
Visual assessment of the major extrahepatic bile duct
Under sterile conditions, the Indocyanine Green for Injection USP powder should be dissolved with the 10 mL Sterile Water for Injection, USP provided for this product, and the solution used within 6 hours after it is prepared. If a precipitate is present, discard the solution.
The dosage for one administration should be 3.0 mg (1.2 mL) per patient. Multiple administration can be performed, up to 2 mg/kg (0.8 mL/kg) per patient.
ICG should be administrated at least 30 minutes prior to IR imaging.
Item | Note |
Indocyanine Green for Injection, USP | 25 mg vials of ICG powder |
Sterile Water for Injection (for dissolving ICG) | 10 mL vial of sterile water |
Syringe (for injecting sterile water into the ICG vial) | Use the syringe whose minimum volume is 10 mL |
Syringes (for each administration) | Considering the administration volume, select the appropriate size |
Sterile normal saline (for each saline flush) | Approximately 10 – 12 mL following each ICG administration |
Syringes (for each saline flush) | Use the syringe whose minimum volume is 12 mL |
Prepare the syringes filled with 3.0 mg (1.2 mL) of ICG solution, and the syringes filled with 10 - 12 mL of normal saline for the tight bolus.
At least 30 minutes prior to IR imaging, administer the prepared dose of ICG solution intravenously. Immediately follow each ICG injection with a tight bolus injection of approximately 10 - 12 mL of normal saline.
Viewing intra-operative blood flow in the cerebral area
Under sterile conditions, the Indocyanine Green for Injection USP powder should be dissolved with the 10 mL Sterile Water for Injection, USP provided for this product, and the solution used within 6 hours after it is prepared. If a precipitate is present, discard the solution.
The patient should be weighed and the dosage for one administration should be calculated on the basis of 0.1 mg/kg (0.04 mL/kg) - 0.3 mg/kg (0.12 mL/kg) of body weight. Multiple administration can be performed, up to 2 mg/kg (0.8 mL/kg) per patient.
ICG should be administrated immediately before IR imaging.
Item | Note |
Indocyanine Green for Injection, USP | 25 mg vials of ICG powder |
Sterile Water for Injection (for dissolving ICG) | 10 mL vial of sterile water |
Syringe (for injecting sterile water into the ICG vial) | Use the syringe whose minimum volume is 10 mL |
Syringes (for each administration) | Considering the administration volume, select the appropriate size |
Sterile normal saline (for each saline flush) | Approximately 10 – 12 mL following each ICG administration |
Syringes (for each saline flush) | Use the syringe whose minimum volume is 12 mL |
Prepare the syringes filled with the weight-scaled dose of ICG solution, and the syringes filled with 10 - 12 mL of normal saline for the tight bolus.
Immediately before IR imaging, administer the prepared dose of ICG solution intravenously. Immediately follow each ICG injection with a tight bolus injection of approximately 10 - 12 mL of normal saline.
DOSAGE FORMS AND STRENGTHS
Indocyanine Green for Injection USP is a sterile, lyophilized green powder containing 25 mg of indocyanine green with no more than 5% sodium iodide.
USE IN SPECIFIC POPULATIONS
Pregnancy
Animal reproduction studies have not been conducted with Indocyanine Green for Injection USP. It is also not known whether Indocyanine Green for Injection USP can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. Indocyanine Green for Injection USP should be given to a pregnant woman only if clearly indicated.
Nursing Mothers
It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when Indocyanine Green for Injection USP is administered to a nursing woman.
Pediatric Use
The safety and effectiveness of indocyanine green for visual assessment of blood vessels, blood flow and related tissue perfusion, for visual assessment of the major extrahepatic bile duct and for intra-operative blood flow in the cerebral area using this IR imaging application has not been evaluated in pediatric patients.
Geriatric Use
No overall differences in safety or effectiveness have been observed between elderly and adult patients.
CONTRAINDICATIONS
Indocyanine Green for Injection USP contains sodium iodide and should be used with caution in patients who have a history of allergy to iodides because of the risk of anaphylaxis.
WARNINGS AND PRECAUTIONS
- Deaths due to anaphylaxis have been reported following Indocyanine Green for Injection USP administration during cardiac catheterization. (5.1 )
- Indocyanine Green for Injection USP is unstable in aqueous solution and must be used within 6 hours. (5.2 )
- Radioactive iodine uptake studies should not be performed for at least a week following the use of Indocyanine Green for Injection USP. (5.3 )
Anaphylaxis
Deaths from anaphylaxis have been reported following Indocyanine Green for Injection USP administration during cardiac catheterization.
Drug Instability
Indocyanine Green for Injection USP is unstable in aqueous solution and must be used within 6 hours. However, the dye is stable in plasma and whole blood so that samples obtained in discontinuous sampling techniques may be read hours later. Sterile techniques should be used in handling the dye solution as well as in the performance of the dilution curves. If a precipitate is present, discard the solution.
Drug/Laboratory Test Interactions
Radioactive iodine uptake studies should not be performed for at least a week following the use of Indocyanine Green for Injection USP.
ADVERSE REACTIONS
Anaphylactic or urticarial reactions have been reported in patients with or without history of allergy to iodides. If such reactions occur, treat with the appropriate agents, e.g., epinephrine, antihistamines, and corticosteroids.
DRUG INTERACTIONS
Preparations containing sodium bisulfite including some heparin products reduce the absorption peak of Indocyanine Green for Injection USP in blood and, therefore, should not be used as an anticoagulant for the collection of samples for analysis.
DESCRIPTION
Indocyanine Green for Injection USP is a sterile, lyophilized green powder containing 25 mg of indocyanine green with no more than 5% sodium iodide. It is packaged with Sterile Water for Injection, USP used to dissolve the indocyanine green. Indocyanine Green for Injection USP is to be administered intravenously.
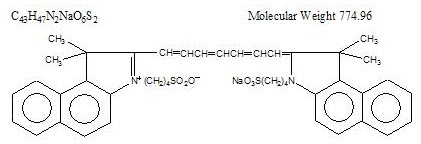
Indocyanine green is a water soluble, tricarbocyanine dye with a peak spectral absorption at 800 nm. The chemical name for Indocyanine Green is 1 H -Benz[ e ]indolium, 2-[7-[1,3-dihydro-1,1-dimethyl-3-(4-sulfobutyl)-2 H -benz[ e ] indol-2-ylidene]-1,3,5-heptatrienyl]-1,1-dimethyl-3-(4-sulfobutyl)-,hydroxide, inner salt, sodium salt. Indocyanine Green for Injection USP has a pH of approximately 6.5 when reconstituted. Each vial of Indocyanine Green for Injection USP contains 25 mg of indocyanine green as a sterile lyophilized powder.
CLINICAL PHARMACOLOGY
Indocyanine Green for Injection USP permits recording of the indicator-dilution curves for both diagnostic and research purposes independently of fluctuations in oxygen saturation. Following intravenous injection, Indocyanine Green for Injection USP is rapidly bound to plasma protein, of which albumin is the principle carrier (95%). Indocyanine Green for Injection USP undergoes no significant extrahepatic or enterohepatic circulation; simultaneous arterial and venous blood estimations have shown negligible renal, peripheral, lung or cerebro-spinal uptake of the dye. Indocyanine Green for Injection USP is taken up from the plasma almost exclusively by the hepatic parenchymal cells and is secreted entirely into the bile. After biliary obstruction, the dye appears in the hepatic lymph, independently of the bile, suggesting that the biliary mucosa is sufficiently intact to prevent diffusion of the dye, though allowing diffusion of bilirubin. These characteristics make Indocyanine Green for Injection USP a helpful index of hepatic function.
The peak absorption and emission of Indocyanine Green for Injection USP lie in a region (800 to 850 nm) where transmission of energy by the pigment epithelium is more efficient than in the region of visible light energy. Indocyanine Green for Injection USP also has the property of being nearly 98% bound to blood protein, and therefore, excessive dye extravasation does not take place in the highly fenestrated choroidal vasculature. It is, therefore, useful in both absorption and fluorescence infrared angiography of the choroidal vasculature when using appropriate filters and film in a fundus camera.
The plasma fractional disappearance rate at the recommended 0.5 mg/kg dose has been reported to be significantly greater in women than in men, although there was no significant difference in the calculated value for clearance.
NONCLINICAL TOXICOLOGY
Carcinogenesis, Mutagenesis, Impairment of Fertility
No studies have been performed to evaluate the carcinogenicity, mutagenicity, or impairment of fertility.
HOW SUPPLIED/STORAGE AND HANDLING
Indocyanine Green for Injection USP is supplied in a kit (NDC 73624-424-02) containing six 25 mg Indocyanine Green for Injection USP vials and six 10 mL Sterile Water for Injection, USP plastic vials:
NDC 70100-424-01 Indocyanine Green for Injection USP vial. 25 mg fill in 25 mL vial.
NDC 63323-185-10 (or NDC 0409-4887-17) Sterile Water for Injection, USP, 10 mL fill in 10 mL plastic vials.
STORAGE: Store at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature].