Get your patient on Ipratropium Bromide - Ipratropium Bromide spray, Metered (Ipratropium Bromide)
Ipratropium Bromide - Ipratropium Bromide spray, Metered prescribing information
INDICATIONS AND USAGE:
Ipratropium Bromide Nasal Solution 0.03% (Nasal Spray) is indicated for the symptomatic relief of rhinorrhea associated with allergic and nonallergic perennial rhinitis in adults and children age 6 years and older. Ipratropium Bromide Nasal Solution 0.03% (Nasal Spray) does not relieve nasal congestion, sneezing, or postnasal drip associated with allergic or nonallergic perennial rhinitis.
DOSAGE AND ADMINISTRATION:
The recommended dose of Ipratropium Bromide Nasal Solution 0.03% (Nasal Spray) is two sprays (42 mcg) per nostril two or three times daily (total dose 168 to 252 mcg/day) for the symptomatic relief of rhinorrhea associated with allergic and nonallergic perennial rhinitis in adults and children age 6 years and older. Optimum dosage varies with the response of the individual patient.
Initial pump priming requires seven sprays of the pump. If used regularly as recommended, no further priming is required. If not used for more than 24 hours, the pump will require two sprays, or if not used for more than seven days, the pump will require seven sprays to reprime. Avoid spraying into eyes .
CONTRAINDICATIONS:
Ipratropium Bromide Nasal Solution 0.03% (Nasal Spray) is contraindicated in patients with a history of hypersensitivity to atropine or its derivatives, or to any of the other ingredients.
ADVERSE REACTIONS:
Adverse reaction information on Ipratropium Bromide Nasal Solution 0.03% (Nasal Spray) in patients with perennial rhinitis was derived from four multicenter, vehicle-controlled clinical trials involving 703 patients (356 patients on ipratropium bromide and 347 patients on vehicle), and a one-year, open-label, follow-up trial. In three of the trials, patients received Ipratropium Bromide Nasal Solution 0.03% (Nasal Spray) three times daily, for eight weeks. In the other trial, Ipratropium Bromide Nasal Solution 0.03% (Nasal Spray) was given to patients two times daily for four weeks. Of the 285 patients who entered the open-label, follow-up trial, 232 were treated for 3 months, 200 for 6 months, and 159 up to one year. The majority (>86%) of patients treated for one year were maintained on 42 mcg per nostril, two or three times daily, of Ipratropium Bromide Nasal Solution 0.03% (Nasal Spray).
Table 1 shows adverse events, and the frequency that these adverse events led to the discontinuation of treatment, reported for patients who received Ipratropium Bromide Nasal Solution 0.03% (Nasal Spray) at the recommended dose of 42 mcg per nostril, or vehicle two or three times daily for four or eight weeks. Only adverse events reported with an incidence of at least 2.0% in the ipratropium bromide group and higher in the ipratropium bromide group than in the vehicle group are shown.
Ipratropium Bromide Nasal | ||
Solution 0.03% (Nasal Spray) | ||
(n=356) | ||
Incidence% | Discontinued% | |
Headache | 9.8 | 0.6 |
Upper respiratory tract infection | 9.8 | 1.4 |
Epistaxis Epistaxis reported by 7.0% of ipratropium bromide patients and 2.3% of vehicle patients, blood-tinged mucus by 2.0% of ipratropium bromide patients and 2.3% of vehicle patients. | 9.0 | 0.3 |
Rhinitis All events are listed by their WHO term; rhinitis has been presented by descriptive terms for clarification. | ||
Nasal dryness | 5.1 | 0.0 |
Nasal irritation Nasal irritation includes reports of nasal itching, nasal burning, nasal irritation, and ulcerative rhinitis. | 2.0 | 0.0 |
Other nasal symptoms Other nasal symptoms include reports of nasal congestion, increased rhinorrhea, increased rhinitis, posterior nasal drip, sneezing, nasal polyps, and nasal edema. | 3.1 | 1.1 |
Pharyngitis | 8.1 | 0.3 |
Nausea | 2.2 | 0.3 |
Vehicle Control | ||
(n=347) | ||
Incidence% | Discontinued% | |
Headache | 9.2 | 0.0 |
Upper respiratory tract infection | 7.2 | 1.4 |
Epistaxis | 4.6 | 0.3 |
Rhinitis | ||
Nasal dryness | 0.9 | 0.3 |
Nasal irritation | 1.7 | 0.6 |
Other nasal symptoms | 1.7 | 0.3 |
Pharyngitis | 4.6 | 0.0 |
Nausea | 0.9 | 0.0 |
Ipratropium Bromide Nasal Solution 0.03% (Nasal Spray) was well tolerated by most patients. The most frequently reported nasal adverse events were transient episodes of nasal dryness or epistaxis. These adverse events were mild or moderate in nature, none was considered serious, none resulted in hospitalization and most resolved spontaneously or following a dose reduction. Treatment for nasal dryness and epistaxis was required infrequently (2% or less) and consisted of local application of pressure or a moisturizing agent (e.g., petroleum jelly or saline nasal spray). Patient discontinuation for epistaxis or nasal dryness was infrequent in both the controlled (0.3% or less) and one-year, open-label (2% or less) trials. There was no evidence of nasal rebound (i.e., a clinically significant increase in rhinorrhea, posterior nasal drip, sneezing or nasal congestion severity compared to baseline) upon discontinuation of double-blind therapy in these trials.
Adverse events reported by less than 2% of the patients receiving Ipratropium Bromide Nasal Solution 0.03% (Nasal Spray) during the controlled clinical trials or during the open-label follow-up trial, which are potentially related to ipratropium bromide’s local effects or systemic anticholinergic effects include: dry mouth/throat, dizziness, ocular irritation, blurred vision, conjunctivitis, hoarseness, cough, and taste perversion.
There were infrequent reports of skin rash in both the controlled and uncontrolled clinical studies.
To report SUSPECTED ADVERSE REACTIONS, contact Bausch & Lomb Incorporated at 1-800-321-4576 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions:
No controlled clinical trials were conducted to investigate potential drug-drug interactions. There is potential for an additive interaction with other concomitantly administered medications with anticholinergic properties, including ipratropium bromide for oral inhalation.
DESCRIPTION:
The active ingredient in Ipratropium Bromide Nasal Solution 0.03% (Nasal Spray) is ipratropium bromide monohydrate. It is an anticholinergic agent chemically described as 8-azoniabicyclo[3.2.1] octane, 3-(3-hydroxy-1-oxo-2 phenylpropoxy)-8-methyl-8-(1-methylethyl)-, bromide monohydrate (3-endo, 8-syn)-: a synthetic quaternary ammonium compound, chemically related to atropine. Its structural formula is:
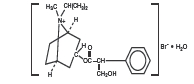
ipratropium bromide monohydrate
C 20 H 30 BrNO 3 • H 2 O Mol. Wt. 430.4
Ipratropium bromide is a white to off-white crystalline substance, freely soluble in water and methanol, sparingly soluble in ethanol, and insoluble in non-polar media. In aqueous solution, it exists in an ionized state as a quaternary ammonium compound.
Ipratropium Bromide Nasal Solution 0.03% (Nasal Spray) is a metered-dose, manual pump spray unit which delivers 21 mcg (70 microliters) ipratropium bromide per spray on an anhydrous basis in an isotonic, aqueous solution, with pH-adjusted to 4.7 with hydrochloric acid and/or sodium hydroxide (if needed). It also contains benzalkonium chloride, edetate disodium, sodium chloride and purified water. Each bottle contains 345 sprays.
CLINICAL PHARMACOLOGY:
Clinical Trials:
The clinical trials for Ipratropium Bromide Nasal Solution 0.03% (Nasal Spray) were conducted in patients with nonallergic perennial rhinitis (NAPR) and in patients with allergic perennial rhinitis (APR). APR patients were those who experienced symptoms of nasal hypersecretion and nasal congestion or sneezing when exposed to specific perennial allergens (e.g., dust mites, molds) and were skin test positive to these allergens. NAPR patients were those who experienced symptoms of nasal hypersecretion and nasal congestion or sneezing throughout the year, but were skin test negative to common perennial allergens.
In four controlled, four- and eight-week comparisons of Ipratropium Bromide Nasal Solution 0.03% (Nasal Spray) (42 mcg per nostril, two or three times daily) with its vehicle, in patients with allergic or nonallergic perennial rhinitis, there was a statistically significant decrease in the severity and duration of rhinorrhea in the ipratropium bromide group throughout the entire study period. An effect was seen as early as the first day of therapy.
There was no effect of Ipratropium Bromide Nasal Solution 0.03% (Nasal Spray) on degree of nasal congestion, sneezing, or postnasal drip. The response to Ipratropium Bromide Nasal Solution 0.03% (Nasal Spray) did not appear to be affected by the type of perennial rhinitis (NAPR or APR), age, or gender. No controlled clinical trials directly compared the efficacy of BID versus TID treatment.
HOW SUPPLIED:
Ipratropium Bromide Nasal Solution 0.03% (Nasal Spray) is supplied as 30 mL of solution in a high density polyethylene (HDPE) bottle fitted with a metered nasal spray pump, a safety clip to prevent accidental discharge of the spray, and a plastic dust cap. It contains 31.1 g of product formulation, 345 sprays, each delivering 21 mcg (70 microliters) of ipratropium per spray, or 28 days of therapy at the maximum recommended dose (two sprays per nostril three times a day).
- NDC 24208-398-30 Bottle of 30 mL
Mechanism of Action:
Ipratropium bromide is an anticholinergic (parasympatholytic) agent which, based on animal studies, appears to inhibit vagally-mediated reflexes by antagonizing the action of acetylcholine, the transmitter agent released at the neuromuscular junctions in the lung. In humans, ipratropium bromide has anti-secretory properties and, when applied locally, inhibits secretions from the serous and seromucous glands lining the nasal mucosa. Ipratropium bromide is a quaternary amine that minimally crosses the nasal and gastrointestinal membranes and the blood-brain barrier, resulting in a reduction of the systemic anticholinergic effects (e.g., neurologic, ophthalmic, cardiovascular, and gastrointestinal effects) that are seen with tertiary anticholinergic amines.