Get your patient on Klayesta - Nystatin Topical Powder powder (Nystatin Topical Powder)
Klayesta - Nystatin Topical Powder powder prescribing information
INDICATIONS AND USAGE
KLAYESTA is indicated in the treatment of cutaneous or mucocutaneous mycotic infections caused by Candida albicans and other susceptible Candida species.
KLAYESTA is not indicated for systemic, oral, intravaginal or ophthalmic use.
DOSAGE AND ADMINISTRATION
Very moist lesions are best treated with the topical dusting powder.
Adults and Pediatric Patients (Neonates and Older)
Apply to candidal lesions two or three times daily until healing is complete. For fungal infection of the feet caused by Candida species, the powder should be dusted on the feet, as well as, in all foot wear.
CONTRAINDICATIONS
KLAYESTA is contraindicated in patients with a history of hypersensitivity to any of its components.
ADVERSE REACTIONS
The frequency of adverse events reported in patients using KLAYESTA is less than 0.1%. The more common events that were reported include allergic reactions, burning, itching, rash, eczema, and pain on application. (See PRECAUTIONS, General .)
DESCRIPTION
Nystatin is a polyene antifungal antibiotic obtained from Streptomyces noursei. The molecular formula for Nystatin is C 47 H 75 NO 17 . The molecular weight of Nystatin is 926.09.
Structural formula:
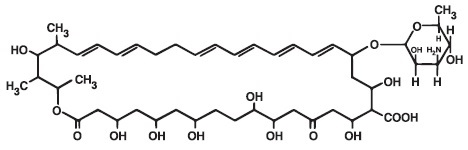
KLAYESTA is for dermatologic use.
KLAYESTA contains 100,000 USP nystatin units per gram.
Inactive ingredients: magnesium stearate and light kaolin.
CLINICAL PHARMACOLOGY
Pharmacokinetics
KLAYESTA is not absorbed from intact skin or mucous membrane.
Microbiology
Nystatin is an antibiotic which is both fungistatic and fungicidal in vitro against a wide variety of yeasts and yeast-like fungi, including Candida albicans, C. parapsilosis, C. tropicalis, C. guilliermondi, C. pseudotropicalis, C. krusei, Torulopsis glabrata, Tricophyton rubrum, T. mentagrophytes.
Nystatin acts by binding to sterols in the cell membrane of susceptible species resulting in a change in membrane permeability and the subsequent leakage of intracellular components. On repeated subculturing with increasing levels of nystatin, Candida albicans does not develop resistance to nystatin. Generally, resistance to nystatin does not develop during therapy. However, other species of Candida (C, tropicalis, C. guilliermondi, C. krusei, and C. stellatoides) become quite resistant on treatment with nystatin and simultaneously become cross resistant to amphotericin as well. This resistance is lost when the antibiotic is removed.
Nystatin exhibits no appreciable activity against bacteria, protozoa, or viruses.
HOW SUPPLIED
KLAYESTA (nystatin topical powder, USP) is off-white to light yellow powder, and is supplied as 100,000 units nystatin per gram in plastic squeeze bottles.
15 g (NDC 42806-186-15)
30 g (NDC 42806-186-30)
60 g (NDC 42806-186-60)