Get your patient on Kurvelo - Levonorgestrel And Ethinyl Estradiol (Levonorgestrel And Ethinyl Estradiol)
Kurvelo - Levonorgestrel And Ethinyl Estradiol prescribing information
WARNING: CIGARETTE SMOKING AND SERIOUS CARDIOVASCULAR EVENTS
Cigarette smoking increases the risk of serious cardiovascular events from combination oral contraceptive (COC) use. This risk increases with age, particularly in women over 35 years of age, and with the number of cigarettes smoked. For this reason, COCs, including Kurvelo, are contraindicated in women who are over 35 years of age and smoke [see CONTRAINDICATIONS and WARNINGS (1)].
INDICATIONS AND USAGE
Kurvelo is indicated for use by females of reproductive potential to prevent pregnancy.
DOSAGE AND ADMINISTRATION
1.How to Start and Take Kurvelo
Kurvelo is dispensed in a compact dispenser containing 28 tablets (see HOW SUPPLIED ). Kurvelo may be started using either a Day 1 start or a Sunday start (see Table 3). For the first cycle of a Sunday start regimen, an additional method of contraception should be used until after the first 7 consecutive days of administration.
| Starting Kurvelo in females with no current use of hormonal contraception | Day 1 start • Take first tablet without regard to meals on the first day of menses • Take subsequent tablets once daily at the same time each day • Begin each subsequent pack on the same day of the week as the first cycle pack (i.e., on the day after taking the last tablet) |
| Sunday start • Take first tablet without regard to meals on the first Sunday after the onset of menstrual period • Take subsequent tablets once daily at the same time each day • Use additional nonhormonal contraception for the first seven days of product use • Begin each subsequent pack on the same day of the week as the first cycle pack (i.e., on the day after taking the last tablet) | |
| Switching from another contraceptive method • A COC | Start Kurvelo: • On the day when the new pack of the previous COC would have been started |
| • Transdermal patch | • On the day when next application would have been scheduled |
| • Vaginal ring | • On the day when next insertion would have been scheduled |
| • Injection | • On the day when next injection would have been scheduled |
| • Intrauterine contraceptive | • On the day of removal |
| • Implant | • On the day of removal |
Starting Kurvelo after Abortion or Miscarriage
First-trimester
- After a first-trimester abortion or miscarriage, Kurvelo may be started immediately. An additional method of contraception is not needed if Kurvelo is started immediately.
- If Kurvelo is not started within 5 days after termination of the pregnancy, the patient should use additional non-hormonal contraception (such as condoms or spermicide) for the first seven days of her first cycle of Kurvelo.
Second-trimester
- Do not start until 4 weeks after a second-trimester abortion or miscarriage, due to the increased risk of thromboembolic disease. Start Kurvelo following the instructions in Table 3 for Day 1 or Sunday start. Use additional non-hormonal contraception (such as condoms or spermicide) for the first seven days of the patient's first cycle of Kurvelo (see CONTRAINDICATIONS , WARNINGS (1) , PRECAUTIONS (10) and FDA-APPROVED PATIENT LABELING ) .
Starting Kurvelo after Childbirth
- Do not start until 4 weeks after delivery, due to the increased risk of thromboembolic disease. Start contraceptive therapy with Kurvelo following the instructions in Table 3 for women not currently using hormonal contraception.
- Kurvelo is not recommended for use in lactating women (see PRECAUTIONS (7 ) and FDA-APPROVED PATIENT LABELING ).
- If the woman has not yet had a period postpartum, consider the possibility of ovulation and conception occurring prior to use of Kurvelo (see CONTRAINDICATIONS , WARNINGS (9) , PRECAUTIONS (6) and FDA APPROVED PATIENT LABELING) .
2.Dosing Kurvelo
Instruct patients to take one tablet by mouth at the same time every day. To achieve maximum contraceptive effectiveness, patients must take Kurvelo as directed, in the order directed on the blister pack. The failure rate may increase when pills are missed or taken incorrectly.
3.Missed doses
Instruct patients about the handling of missed doses (e.g., to take single missed pills as soon as possible) and to follow the dosing instructions provided in the FDA-approved patient labeling.
| • If one active tablet is missed in Weeks 1, 2, or 3 | Take the tablet as soon as possible. Continue taking one tablet a day until the pack is finished. |
| • If two active tablets are missed in Week 1 or Week 2 | Take the two missed tablets as soon as possible and the next two active tablets the next day. Continue taking one tablet a day until the pack is finished. Additional nonhormonal contraception (such as condoms or spermicide) should be used as back-up if the patient has sex within 7 days after missing tablets. |
| • If two active tablets are missed in the third week or three or more active tablets are missed in a row in Weeks 1, 2, or 3 | Day 1 start: Throw out the rest of the pack and start a new pack that same day. Sunday start: Continue taking one tablet a day until Sunday, then throw out the rest of the pack and start a new pack that same day. Additional nonhormonal contraception (such as condoms or spermicide) should be used as back-up if the patient has sex within 7 days after missing tablets. |
4.Advice in Case of Gastrointestinal Disturbances
If vomiting occurs within 3 to 4 hours after taking Kurvelo, the patient should proceed as if she missed a tablet. In case of prolonged vomiting or diarrhea, absorption may not be complete and additional contraceptive measures should be taken.
CONTRAINDICATIONS
Kurvelo is contraindicated in females who are known to have the following conditions:
- A high risk of arterial or venous thrombotic diseases. Examples include women who are known to:
- Smoke, if over age 35 [see BOXED WARNING and WARNINGS (1) ].
- Have current or history of deep vein thrombosis or pulmonary embolism [see WARNINGS (1) ] .
- Have cerebrovascular disease [see WARNINGS (1) ] .
- Have coronary artery disease [see WARNINGS (1) ] .
- Have thrombogenic valvular or thrombogenic rhythm diseases of the heart (for example, subacute bacterial endocarditis with valvular disease, or atrial fibrillation) [see WARNINGS (1) ] .
- Have inherited or acquired hypercoagulopathies [see WARNINGS (1) ] .
- Have uncontrolled hypertension or hypertension with vascular disease [see WARNINGS (3) ] .
- Have diabetes mellitus and are over age 35, diabetes mellitus with hypertension or vascular disease or other end-organ damage, or diabetes mellitus of >20 years duration [see WARNINGS (7) ] .
- Have headaches with focal neurological symptoms, migraine headaches with aura, or over age 35 with any migraine headaches [see WARNINGS (8) ] .
- Current diagnosis of, or history of, breast cancer, which may be hormone-sensitive.
- Liver tumors, acute viral hepatitis, or severe (decompensated) cirrhosis [see WARNINGS (2 )] .
- Use of Hepatitis C drug combinations containing ombitasvir/paritaprevir/ritonavir, with or without dasabuvir, due to the potential for ALT elevations [see WARNINGS (5 )] .
- Undiagnosed abnormal uterine bleeding [see WARNINGS (9 )] .
DESCRIPTION
Kurvelo™ (levonorgestrel and ethinyl estradiol tablets USP) is a combination oral contraceptive (COC) consisting of 21 light-orange active tablets, each containing 0.15 mg of levonorgestrel, a synthetic progestin and 30 mcg of ethinyl estradiol, an estrogen, and 7 pink inert tablets (without hormones).
The structural formulas for the active components are:
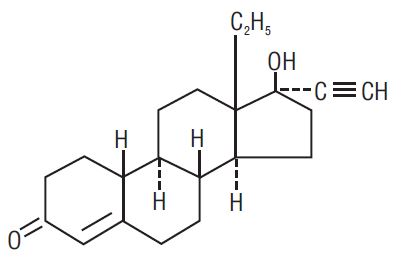
Levonorgestrel C 21 H 28 O 2 MW: 312.4
Levonorgestrel is chemically 18,19-Dinorpregn-4-en-20-yn-3-one, 13-ethyl-17-hydroxy-,(17α)-(-)-
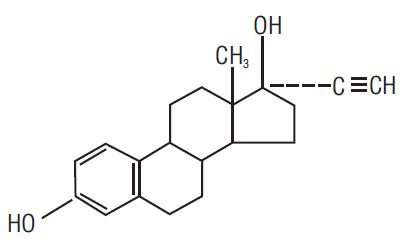
Ethinyl Estradiol C 20 H 24 O 2 MW: 296.4
Ethinyl Estradiol is (19-nor-17α-pregna-1,3,5 (10)-trien-20-yne-3,17-diol).
Each light-orange active tablet contains the following inactive ingredients: croscarmellose sodium, FD&C Yellow #6, lactose monohydrate, magnesium stearate, microcrystalline cellulose and povidone.
Each pink inert tablet contains the following inactive ingredients: croscarmellose sodium, D & C Red #30, lactose monohydrate, magnesium stearate and microcrystalline cellulose.
CLINICAL PHARMACOLOGY
Combination oral contraceptives prevent pregnancy primarily by suppressing ovulation.
HOW SUPPLIED/STORAGE AND HANDLING
Kurvelo Tablets (levonorgestrel and ethinyl estradiol tablets USP) (3 x 28) are available in packages of 3 blisters, each containing 28 tablets as follows:
Each blister strip contains 21 light orange tablets, each containing of 0.15 mg levonorgestrel and 30 mcg ethinyl estradiol, debossed with "LU" on one side and "U31" on the other side.
7 inert pink, round, biconvex tablets, debossed with "LU" on one side and "U32" on the other side.
They are supplied as follows:
Kurvelo tablets are available in a blister (NDC 68180-844-71) containing 28 tablets, such 3 blisters are packed in a carton (NDC 68180-844-73).
Store at 25° C (77° F); excursions permitted to 15° to 30° C (59° to 86° F) [see USP Controlled Room Temperature].
Distributed by:
Lupin Pharmaceuticals, Inc.
Naples, FL 34108
United States
Manufactured by:
Lupin Limited
Pithampur (M.P.) - 454 775
INDIA
Revised: December 2025