Get your patient on Levalbuterol - Levalbuterol Hydrochloride solution (Levalbuterol Hydrochloride)
Levalbuterol - Levalbuterol Hydrochloride solution prescribing information
Recent Major Changes to the Dosage and Administration Section
Levalbuterol Inhalation Solution, USP is for oral inhalation only. Administer by nebulization using with a standard jet nebulizer (with a face mask or mouthpiece) connected to an air compressor. Do not exceed recommended dose.
(Added the word "with" to read "Administer by nebulization using with a standard jet nebulizer...")
1 INDICATIONS AND USAGE
Levalbuterol Inhalation Solution, USP is indicated for the treatment or prevention of bronchospasm in adults, adolescents, and children 6 years of age and older with reversible obstructive airway disease.
2 DOSAGE AND ADMINISTRATION
Levalbuterol Inhalation Solution, USP is for oral inhalation only. Administer by nebulization using with a standard jet nebulizer (with a face mask or mouthpiece) connected to an air compressor. Do not exceed recommended dose.
Children 6-11 years old: The recommended dosage of Levalbuterol Inhalation Solution, USP for patients 6-11 years old is 0.31 mg administered three times a day, by nebulization. Routine dosing should not exceed 0.63 mg three times a day.
Adults and Adolescents ≥ 12 years old: The recommended starting dosage of Levalbuterol Inhalation Solution, USP for patients 12 years of age and older is 0.63 mg administered three times a day, every 6 to 8 hours, by nebulization.
Patients 12 years of age and older with more severe asthma or patients who do not respond adequately to a dose of 0.63 mg of Levalbuterol Inhalation Solution, USP may benefit from a dosage of 1.25 mg three times a day.
Patients receiving the highest dose of Levalbuterol Inhalation Solution, USP should be monitored closely for adverse systemic effects, and the risks of such effects should be balanced against the potential for improved efficacy.
The use of Levalbuterol Inhalation Solution, USP can be continued as medically indicated to help control recurring bouts of bronchospasm. During this time, most patients gain optimal benefit from regular use of the inhalation solution.
If a previously effective dosage regimen fails to provide the usual response this may be a marker of destabilization of asthma and requires reevaluation of the patient and the treatment regimen, giving special consideration to the possible need for anti-inflammatory treatment, e.g., corticosteroids.
The drug compatibility (physical and chemical), efficacy, and safety of Levalbuterol Inhalation Solution, USP when mixed with other drugs in a nebulizer have not been established.
The safety and efficacy of Levalbuterol Inhalation Solution, USP have been established in clinical trials when administered using the PARI LC Jet™ and PARI LC Plus™ nebulizers, and the PARI Master ® Dura-Neb ® 2000 and Dura-Neb ® 3000 compressors. The safety and efficacy of Levalbuterol Inhalation Solution, USP when administered using other nebulizer systems have not been established.
Recent Major Changes to the Dosage and Administration Section
Levalbuterol Inhalation Solution, USP is for oral inhalation only. Administer by nebulization using with a standard jet nebulizer (with a face mask or mouthpiece) connected to an air compressor. Do not exceed recommended dose.
(Added the word "with" to read "Administer by nebulization using with a standard jet nebulizer...")
3 DOSAGE FORMS AND STRENGTHS
Inhalation Solution 3 mL unit-dose, vials in three dosage strengths of levalbuterol; 0.31 mg, 0.63 mg, 1.25 mg. Each strength of Levalbuterol Inhalation Solution, USP is available in a shelf carton containing one or more foil pouches, each pouch containing one or more unit-dose vials.
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
There are no adequate and well-controlled studies of Levalbuterol Inhalation Solution, USP in pregnant women. There are clinical considerations with the use of Levalbuterol Inhalation Solution, USP in pregnant women [see Clinical Considerations] .
Following oral administration of levalbuterol HCl to pregnant rabbits, there was no evidence of teratogenicity at doses up to 25 mg/kg/day [approximately 108 times the maximum recommended human daily inhalation dose (MRHDID) of levalbuterol HCl for adults on a mg/m 2 basis]; however, racemic albuterol sulfate was teratogenic in mice (cleft palate) and rabbits (cramioschisis) at doses slightly higher than the human therapeutic range ( see Data ).
The estimated background risk of major birth defects and miscarriage for the indicated population(s) are unknown. In the U.S. general population, the estimated risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4% and 15 to 20% respectively.
Clinical Considerations
Disease-Associated Maternal and/or Embryo/Fetal Risk
In women with poorly or moderately controlled asthma, there is an increased risk of preeclampsia in the mother and prematurity, low birth weight, and small for gestational age in the neonate. Pregnant women should be closely monitored and medication adjusted as necessary to maintain optimal control.
Labor or Delivery
Because of the potential for beta-adrenergic agonists to interfere with uterine contractility, the use of Levalbuterol Inhalation Solution, USP for the treatment of bronchospasm during labor should be restricted to those patients for whom the benefits clearly outweigh the risk.
Levalbuterol Inhalation Solution, USP has not been approved for the management of preterm labor. The benefit-risk ratio when Levalbuterol Inhalation Solution, USP is administered for tocolysis has not been established. Serious adverse reactions, including maternal pulmonary edema, have been reported during or following treatment of premature labor with beta 2 -agonists, including racemic albuterol.
Data
Animal Data
The oral administration of levalbuterol HCl to pregnant New Zealand White rabbits during the period of organogenesis found no evidence of teratogenicity at doses up to 25 mg/kg/day (approximately 108 times the MRHDID of levalbuterol HCl for adults on a mg/m 2 basis). In a rat developmental study, racemic albuterol sulfate administered by inhalation did not produce any teratogenic effects at exposures approximately 63 times the MRHDID (on a mg/m 2 basis at a maternal dose of 10.5 mg/kg).
However, other developmental studies with the racemic albuterol sulfate, did result in teratogenic effects in mice and rabbits at doses slightly higher than the human therapeutic range. In a rabbit developmental study, orally administered albuterol sulfate induced cranioschisis in 7 of 19 fetuses (37%) at approximately 215 times the MRHDID for adults (on a mg/m 2 basis at a maternal dose of 50 mg/kg). In a mouse developmental study, subcutaneously administered albuterol sulfate produced cleft palate formation in 5 of 111 (4.5%) fetuses at an exposure approximately 0.3 times the MRHDID for adults (on a mg/m 2 basis at a maternal dose of 0.25 mg/kg/day) and in 10 of 108 (9.3%) fetuses at approximately 3 times the MRHDID (on a mg/m 2 basis at a maternal dose of 2.5 mg/kg/day). Similar effects were not observed at approximately 0.03 times the MRHDID for adults on a mg/m 2 basis at a maternal dose of 0.025 mg/kg/day (i.e., less than the therapeutic dose). Cleft palate also occurred in 22 of 72 (30.5%) fetuses from females treated subcutaneously with isoproterenol (positive control).
Non-Teratogenic Effects:
A study in which pregnant rats were dosed with radiolabeled racemic albuterol sulfate demonstrated that drug-related material is transferred from the maternal circulation to the fetus.
8.2 Lactation
Risk Summary
There are no available data on the presence of levalbuterol in human milk, the effects on the breastfed child, or the effects on milk production.
The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for Levalbuterol Inhalation Solution, USP and any potential adverse effects on the breastfed child from Levalbuterol Inhalation Solution, USP or from the underlying maternal condition.
8.4 Pediatric Use
Pediatric Patients 6 Years of Age and Older
The safety and efficacy of Levalbuterol Inhalation Solution, USP have been established in pediatric patients 6 years of age and older in an adequate and well-controlled clinical trial [see Adverse Reactions (6) and Clinical Studies (14) ].
Pediatric Patients less than 6 Years of Age
Levalbuterol Inhalation Solution, USP is not indicated for pediatric patients less than 6 years of age.
Clinical trials with Levalbuterol Inhalation Solution, USP in this age group failed to meet the primary efficacy endpoint and demonstrated an increased number of asthma related adverse reactions following chronic Levalbuterol Inhalation Solution, USP treatment.
Levalbuterol Inhalation Solution, USP was studied in 379 pediatric patients less than 6 years of age with asthma or reactive airway disease - (291 patients 2 to 5 years of age, and 88 patients from birth to less than 2 years of age). Efficacy and safety data for Levalbuterol Inhalation Solution, USP in this age group are primarily available from one 3-week, multicenter, randomized, double-blind, active and placebo-controlled study (Study 1) in 211 pediatric patients between the ages of 2 and 5 years, of whom 119 received Levalbuterol Inhalation Solution, USP. Over the 3 week treatment period, there were no significant treatment differences in the Pediatric Asthma Questionnaire (PAQ) total score between groups receiving Levalbuterol Inhalation Solution, USP 0.31 mg, Levalbuterol Inhalation Solution, USP 0.63 mg, racemic albuterol, and placebo. Additional safety data following chronic dosing is available from a 4-week, multicenter, randomized, modified-blind, placebo-controlled study (Study 2) of 196 patients between the ages of birth and 3 years, of whom 63 received open-label Levalbuterol Inhalation Solution, USP. In these two studies, treatment-emergent asthma exacerbations or asthma-related adverse reactions and treatment discontinuations due to asthma occurred at a higher frequency in Levalbuterol Inhalation-treated subjects compared to control (Table 5). Other adverse reactions were consistent with those observed in the clinical trial population of patients 6 years of age and older [see Adverse Reactions (6.1) ].
| Asthma Exacerbations• n (%) | Treatment Discontinuations due to Asthma n (%) | Asthma-related Adverse Reactions•• n (%) | |
| Study 1 | |||
| Levalbuterol Inhalation Solution, USP 0.31 mg, n=58 | 6 (10) | 4 (7) | -- |
| Levalbuterol Inhalation Solution, USP 0.63 mg, n=51 | 7 (14) | 6 (12) | -- |
| Racemic albuterol, n=52 | 3 (6) | 2 (4) | -- |
| Placebo, n=50 | 2 (4) | 2 (4) | -- |
| Study 2 | |||
| Levalbuterol Inhalation Solution, USP 0.31 mg, n=63 | -- | 2 (3) | 6 (10) |
| Levalbuterol HFA inhalation aerosol, n=65 | -- | 1 (2) | 8 (12) |
| Placebo, n=68 | -- | 0 | 3 (4) |
• Asthma exacerbation defined as worsening of asthma symptoms or pulmonary function that required any of the following: emergency department visit, hospitalization, therapeutic intervention with oral or parenteral steroids, unscheduled clinic visit to treat acute asthma symptoms
•• Includes the following Preferred Terms (whether considered by the investigator to be related or unrelated to drug): asthma, cough, hypoxia, status asthmaticus, tachypnea.
8.5 Geriatric Use
Clinical studies of Levalbuterol Inhalation Solution, USP did not include sufficient numbers of subjects aged 65 years and older to determine whether they respond differently from younger subjects. Only 5 patients 65 years of age and older were treated with Levalbuterol Inhalation Solution, USP in a 4-week clinical study [see Clinical Pharmacology (12) and Clinical Studies (14) ] (n = 2 for 0.63 mg and n = 3 for 1.25 mg). In these patients, bronchodilation was observed after the first dose on day 1 and after 4 weeks of treatment. In general, patients 65 years of age and older should be started at a dose of 0.63 mg of Levalbuterol Inhalation Solution, USP. If clinically warranted due to insufficient bronchodilator response, the dose of Levalbuterol Inhalation Solution, USP may be increased in elderly patients as tolerated, in conjunction with frequent clinical and laboratory monitoring, to the maximum recommended daily dose [see Dosage and Administration (2) ].
8.6 Renal Impairment
Albuterol is known to be substantially excreted by the kidney, and the risk of toxic reactions may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function.
4 CONTRAINDICATIONS
Levalbuterol Inhalation Solution, USP is contraindicated in patients with a history of hypersensitivity to levalbuterol or racemic albuterol. Reactions have included urticaria, angioedema, rash, bronchospasm, anaphylaxis, and oropharyngeal edema [see Warnings and Precautions (5.6) ].
5 WARNINGS AND PRECAUTIONS
- Life-threatening paradoxical bronchospasm may occur. Discontinue Levalbuterol Inhalation Solution, USP immediately and treat with alternative therapy. (5.1 )
- Need for more doses of Levalbuterol Inhalation Solution, USP than usual may be a sign of deterioration of asthma and requires reevaluation of treatment. (5.2 )
- Levalbuterol Inhalation Solution, USP is not a substitute for corticosteroids. (5.3 )
- Cardiovascular effects may occur. Consider discontinuation of Levalbuterol Inhalation Solution, USP if these effects occur. Use with caution in patients with underlying cardiovascular disorders. (5.4 )
- Excessive use may be fatal. Do not exceed recommended dose. (5.5 )
- Immediate hypersensitivity reactions may occur. Discontinue Levalbuterol Inhalation Solution, USP immediately. (5.6 )
- Hypokalemia and changes in blood glucose may occur. (5.7 , 5.8 )
5.1 Paradoxical Bronchospasm
Levalbuterol Inhalation Solution, USP can produce paradoxical bronchospasm, which may be life-threatening. If paradoxical bronchospasm occurs, Levalbuterol Inhalation Solution, USP should be discontinued immediately and alternative therapy instituted. It should be recognized that paradoxical bronchospasm, when associated with inhaled formulations, frequently occurs with the first use of a new vial.
5.2 Deterioration of Asthma
Asthma may deteriorate acutely over a period of hours or chronically over several days or longer. If the patient needs more doses of Levalbuterol Inhalation Solution, USP than usual, this may be a marker of destabilization of asthma and requires reevaluation of the patient and treatment regimen, giving special consideration to the possible need for anti-inflammatory treatment, e.g., corticosteroids.
5.3 Use of Anti-Inflammatory Agents
Levalbuterol Inhalation Solution, USP is not a substitute for corticosteroids. The use of beta-adrenergic agonist alone may not be adequate to control asthma in many patients. Early consideration should be given to adding anti-inflammatory agents, e.g., corticosteroids, to the therapeutic regimen.
5.4 Cardiovascular Effects
Levalbuterol Inhalation Solution, USP, like other beta-adrenergic agonists, can produce clinically significant cardiovascular effects in some patients, as measured by heart rate, blood pressure, and symptoms. Although such effects are uncommon after administration of Levalbuterol Inhalation Solution, USP at recommended doses, if they occur, the drug may need to be discontinued. In addition, beta-agonists have been reported to produce electrocardiogram (ECG) changes, such as flattening of the t-wave, prolongation of the QTc interval, and ST segment depression. The clinical significance of these findings is unknown. Therefore, Levalbuterol Inhalation Solution, USP, like all sympathomimetic amines, should be used with caution in patients with cardiovascular disorders, especially coronary insufficiency, cardiac arrhythmias, and hypertension.
5.5 Do Not Exceed Recommended Dose
Do not exceed the recommended dose. Fatalities have been reported in association with excessive use of inhaled sympathomimetic drugs in patients with asthma. The exact cause of death is unknown, but cardiac arrest following an unexpected development of a severe acute asthmatic crisis and subsequent hypoxia is suspected.
5.6 Immediate Hypersensitivity Reactions
Immediate hypersensitivity reactions may occur after administration of levalbuterol or racemic albuterol. Reactions have included urticaria, angioedema, rash, bronchospasm, anaphylaxis, and oropharyngeal edema. The potential for hypersensitivity must be considered in the clinical evaluation of patients who experience immediate hypersensitivity reactions while receiving Levalbuterol Inhalation Solution, USP.
5.7 Coexisting Conditions
Levalbuterol Inhalation Solution, USP, like all sympathomimetic amines, should be used with caution in patients with cardiovascular disorders, especially coronary insufficiency, hypertension, and cardiac arrhythmias; in patients with convulsive disorders, hyperthyroidism, or diabetes mellitus; and in patients who are unusually responsive to sympathomimetic amines. Clinically significant changes in systolic and diastolic blood pressure have been seen in individual patients and could be expected to occur in some patients after the use of any beta-adrenergic bronchodilator.
Changes in blood glucose may occur. Large doses of intravenous racemic albuterol have been reported to aggravate preexisting diabetes mellitus and ketoacidosis.
5.8 Hypokalemia
As with other beta-adrenergic agonist medications, Levalbuterol Inhalation Solution, USP may produce significant hypokalemia in some patients, possibly through intracellular shunting, which has the potential to produce adverse cardiovascular effects. The decrease is usually transient, not requiring supplementation.
6 ADVERSE REACTIONS
The following serious adverse reactions are described below and elsewhere in the labeling:
- Paradoxical bronchospasm [see Warnings and Precautions (5.1) ]
- Cardiovascular effects [see Warnings and Precautions (5.4) ]
- Immediate hypersensitivity reactions [see Warnings and Precautions (5.6) ]
- Hypokalemia [see Warnings and Precautions (5.8) ]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of the drug cannot be directly compared with rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Adults and Adolescents 12 Years of Age and Older
Adverse reaction information concerning Levalbuterol Inhalation Solution, USP in adults and adolescents is derived from one 4-week, multicenter, randomized, double-blind, active-, and placebo-controlled trial in 362 patients with asthma 12 years of age and older. Adverse reactions reported in ≥ 2% of patients receiving Levalbuterol Inhalation Solution, USP or racemic albuterol and more frequently than in patients receiving placebo are listed in Table 1.
| Percent of Patients One treatment group, racemic albuterol 1.25 mg, with 68 subjects is omitted. | ||||
|---|---|---|---|---|
| Body System Preferred Term | Placebo (n = 75) | Levalbuterol Inhalation Solution, USP 1.25 mg (n = 73) | Levalbuterol Inhalation Solution, USP 0.63 mg (n = 72) | Racemic albuterol 2.5 mg (n = 74) |
| Body as a Whole | ||||
| Allergic reaction | 1.3 | 0 | 0 | 2.7 |
| Flu syndrome | 0 | 1.4 | 4.2 | 2.7 |
| Accidental injury | 0 | 2.7 | 0 | 0 |
| Pain | 1.3 | 1.4 | 2.8 | 2.7 |
| Back pain | 0 | 0 | 0 | 2.7 |
| Cardiovascular System | ||||
| Tachycardia | 0 | 2.7 | 2.8 | 2.7 |
| Migraine | 0 | 2.7 | 0 | 0 |
| Digestive System | ||||
| Dyspepsia | 1.3 | 2.7 | 1.4 | 1.4 |
| Musculoskeletal System | ||||
| Leg cramps | 1.3 | 2.7 | 0 | 1.4 |
| Central Nervous System | ||||
| Dizziness | 1.3 | 2.7 | 1.4 | 0 |
| Hypertonia | 0 | 0 | 0 | 2.7 |
| Nervousness | 0 | 9.6 | 2.8 | 8.1 |
| Tremor | 0 | 6.8 | 0 | 2.7 |
| Anxiety | 0 | 2.7 | 0 | 0 |
| Respiratory System | ||||
| Cough increased | 2.7 | 4.1 | 1.4 | 2.7 |
| Infection viral | 9.3 | 12.3 | 6.9 | 12.2 |
| Rhinitis | 2.7 | 2.7 | 11.1 | 6.8 |
| Sinusitis | 2.7 | 1.4 | 4.2 | 2.7 |
| Turbinate edema | 0 | 1.4 | 2.8 | 0 |
The incidence of certain systemic beta-adrenergic adverse reactions (e.g., tremor, nervousness) was slightly less in the Levalbuterol Inhalation Solution, USP 0.63 mg group compared with the other active treatment groups. The clinical significance of these small differences is unknown.
Changes in heart rate 15 minutes after drug administration and in plasma glucose and potassium 1 hour after drug administration on day 1 and day 29 were clinically comparable in the Levalbuterol Inhalation Solution, USP 1.25 mg and racemic albuterol 2.5 mg groups (see Table 2 ). Changes in heart rate and plasma glucose were slightly less in the Levalbuterol Inhalation Solution, USP 0.63 mg group compared with the other active treatment groups (see Table 2 ). The clinical significance of these small differences is unknown. After 4 weeks, effects on heart rate, plasma glucose, and plasma potassium were generally diminished compared with day 1 in all active treatment groups.
| Mean Changes (Day 1) | |||
|---|---|---|---|
| Treatment | Heart Rate (bpm) | Glucose (mg/dL) | Potassium (mEq/L) |
| Levalbuterol Inhalation Solution, USP 0.63 mg, n = 72 | 2.4 | 4.6 | -0.2 |
| Levalbuterol Inhalation Solution, USP 1.25 mg, n = 73 | 6.9 | 10.3 | -0.3 |
| Racemic albuterol 2.5 mg, n = 74 | 5.7 | 8.2 | -0.3 |
| Placebo, n = 75 | -2.8 | -0.2 | -0.2 |
No other clinically relevant laboratory abnormalities related to administration of Levalbuterol Inhalation Solution, USP were observed in this study.
In the clinical trials, a slightly greater number of serious adverse events, discontinuations due to adverse events, and clinically significant ECG changes were reported in patients who received Levalbuterol Inhalation Solution, USP 1.25 mg compared with the other active treatment groups.
The following adverse reactions, considered potentially related to Levalbuterol Inhalation Solution, USP, occurred in less than 2% of the 292 subjects who received Levalbuterol Inhalation Solution, USP and more frequently than in patients who received placebo in any clinical trial:
| Body as a Whole: | chills, pain, chest pain |
| Cardiovascular System: | ECG abnormal, ECG change, hypertension, hypotension, syncope |
| Digestive System: | diarrhea, dry mouth, dry throat, dyspepsia, gastroenteritis, nausea |
| Hemic and Lymphatic System: | lymphadenopathy |
| Musculoskeletal System: | leg cramps, myalgia |
| Nervous System: | anxiety, hyperesthesia of the hand, insomnia, paresthesia, tremor |
| Special Senses: | eye itch |
The following reactions, considered potentially related to Levalbuterol Inhalation Solution, USP, occurred in less than 2% of the treated subjects but at a frequency less than in patients who received placebo: asthma exacerbation, cough increased, wheezing, sweating, and vomiting.
Pediatric Patients 6 to 11 Years of Age
Adverse reaction information concerning Levalbuterol Inhalation Solution, USP in pediatric patients is derived from one 3-week, multicenter, randomized, double-blind, active-, and placebo-controlled trial in 316 pediatric patients 6 to 11 years of age. Adverse reactions reported in ≥ 2% of patients in any treatment group and more frequently than in patients receiving placebo are listed in Table 3.
| Percent of Patients | |||||
|---|---|---|---|---|---|
| Body System Preferred Term | Placebo (n = 59) | Levalbuterol Inhalation Solution, USP 0.31 mg (n = 66) | Levalbuterol Inhalation Solution, USP 0.63 mg (n = 67) | Racemic albuterol 1.25 mg (n = 64) | Racemic albuterol 2.5 mg (n = 60) |
| Body as a Whole | |||||
| Abdominal pain | 3.4 | 0 | 1.5 | 3.1 | 6.7 |
| Accidental injury | 3.4 | 6.1 | 4.5 | 3.1 | 5.0 |
| Asthenia | 0 | 3.0 | 3.0 | 1.6 | 1.7 |
| Fever | 5.1 | 9.1 | 3.0 | 1.6 | 6.7 |
| Headache | 8.5 | 7.6 | 11.9 | 9.4 | 3.3 |
| Pain | 3.4 | 3.0 | 1.5 | 4.7 | 6.7 |
| Viral infection | 5.1 | 7.6 | 9.0 | 4.7 | 8.3 |
| Digestive System | |||||
| Diarrhea | 0 | 1.5 | 6.0 | 1.6 | 0 |
| Hemic and Lymphatic | |||||
| Lymphadenopathy | 0 | 3.0 | 0 | 1.6 | 0 |
| Musculoskeletal System | |||||
| Myalgia | 0 | 0 | 1.5 | 1.6 | 3.3 |
| Respiratory System | |||||
| Asthma | 5.1 | 9.1 | 9.0 | 6.3 | 10.0 |
| Pharyngitis | 6.8 | 3.0 | 10.4 | 0 | 6.7 |
| Rhinitis | 1.7 | 6.1 | 10.4 | 3.1 | 5.0 |
| Skin and Appendages | |||||
| Eczema | 0 | 0 | 0 | 0 | 3.3 |
| Rash | 0 | 0 | 7.5 | 1.6 | 0 |
| Urticaria | 0 | 0 | 3.0 | 0 | 0 |
| Special Senses | |||||
| Otitis media | 1.7 | 0 | 0 | 0 | 3.3 |
Note: Subjects may have more than one adverse event per body system and preferred term.
Changes in heart rate, plasma glucose, and serum potassium are shown in Table 4. The clinical significance of these small differences is unknown.
| Mean Changes (Day 1) | |||
|---|---|---|---|
| Treatment | Heart Rate (bpm) | Glucose (mg/dL) | Potassium (mEq/L) |
| Levalbuterol Inhalation Solution, USP 0.31 mg, n = 66 | 0.8 | 4.9 | -0.31 |
| Levalbuterol Inhalation Solution, USP 0.63 mg, n = 67 | 6.7 | 5.2 | -0.36 |
| Racemic albuterol 1.25 mg, n = 64 | 6.4 | 8.0 | -0.27 |
| Racemic albuterol 2.5 mg, n = 60 | 10.9 | 10.8 | -0.56 |
| Placebo, n = 59 | -1.8 | 0.6 | -0.05 |
| Mean Changes (Day 21) | |||
| Treatment | Heart Rate (bpm) | Glucose (mg/dL) | Potassium (mEq/L) |
| Levalbuterol Inhalation Solution, USP 0.31 mg, n = 60 | 0 | 2.6 | -0.32 |
| Levalbuterol Inhalation Solution, USP 0.63 mg, n = 66 | 3.8 | 5.8 | -0.34 |
| Racemic albuterol 1.25 mg, n = 62 | 5.8 | 1.7 | -0.18 |
| Racemic albuterol 2.5 mg, n = 54 | 5.7 | 11.8 | -0.26 |
| Placebo, n = 55 | -1.7 | 1.1 | -0.04 |
6.2 Post-marketing Experience
In addition to the adverse reactions reported in clinical trials, the following adverse reactions have been observed in postapproval use of Levalbuterol Inhalation Solution, USP. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. These events have been chosen for inclusion due to their seriousness, their frequency of reporting, or their likely beta-mediated mechanism: angioedema, anaphylaxis, arrhythmias (including atrial fibrillation, supraventricular tachycardia, extrasystoles), asthma, chest pain, cough increased, dysphonia, dyspnea, gastroesophageal reflux disease (GERD), metabolic acidosis, nausea, nervousness, rash, tachycardia, tremor, urticaria.
In addition, Levalbuterol Inhalation Solution, USP, like other sympathomimetic agents, can cause adverse reactions such as hypertension, angina, vertigo, central nervous system stimulation, sleeplessness, headache, and drying or irritation of the oropharynx.
7 DRUG INTERACTIONS
- Other short-acting sympathomimetic aerosol bronchodilators and adrenergic drugs: May potentiate effect. (7.1 )
- Beta-blockers: May block bronchodilatory effects of beta-agonists and produce severe bronchospasm. Patients with asthma should not normally be treated with beta-blockers. (7.2 )
- Diuretic: May worsen electrocardiographic changes or hypokalemia associated with diuretic may worsen. Consider monitoring potassium levels. (7.3 )
- Digoxin: May decrease serum digoxin levels. Consider monitoring digoxin levels. (7.4 )
- Monoamine oxidase inhibitors (MAOs) or tricyclic antidepressants: May potentiate effect of albuterol on the cardiovascular system. (7.5 )
See 17 for PATIENT COUNSELING INFORMATION and FDA-approved patient labeling.
Revised: 02/2020
7.1 Short-Acting Bronchodilators
Avoid concomitant use of other short-acting sympathomimetic bronchodilators or epinephrine in patients being treated with Levalbuterol Inhalation Solution, USP. If additional adrenergic drugs are to be administered by any route, they should be used with caution to avoid deleterious cardiovascular effects.
7.2 Beta-blockers
Beta-adrenergic receptor blocking agents not only block the pulmonary effect of beta-adrenergic agonists such as Levalbuterol Inhalation Solution, USP, but may produce severe bronchospasm in asthmatic patients. Therefore, patients with asthma should not normally be treated with beta-blockers. However, under certain circumstances, e.g., prophylaxis after myocardial infarction, there may be no acceptable alternatives to the use of beta-adrenergic blocking agents in patients with asthma. In this setting, cardioselective beta-blockers should be considered, although they should be administered with caution.
7.3 Diuretics
The ECG changes or hypokalemia that may result from the administration of non-potassium-sparing diuretics (such as loop and thiazide diuretics) can be acutely worsened by beta-agonists, especially when the recommended dose of the beta-agonist is exceeded. Although the clinical significance of these effects is not known, caution is advised in the coadministration of beta-agonists with non-potassium-sparing diuretics. Consider monitoring potassium levels.
7.4 Digoxin
Mean decreases of 16% and 22% in serum digoxin levels were demonstrated after single-dose intravenous and oral administration of racemic albuterol, respectively, to normal volunteers who had received digoxin for 10 days. The clinical significance of these findings for patients with obstructive airway disease who are receiving Levalbuterol Inhalation Solution, USP and digoxin on a chronic basis is unclear. Nevertheless, it would be prudent to carefully evaluate the serum digoxin levels in patients who are currently receiving digoxin and Levalbuterol Inhalation Solution, USP.
7.5 Monoamine Oxidase Inhibitors or Tricyclic Antidepressants
Levalbuterol Inhalation Solution, USP should be administered with extreme caution to patients being treated with monoamine oxidase inhibitors or tricyclic antidepressants, or within 2 weeks of discontinuation of such agents, because the action of levalbuterol on the vascular system may be potentiated. Consider alternative therapy in patients taking MAO inhibitors or tricyclic antidepressants.
11 DESCRIPTION
Levalbuterol Inhalation Solution, USP is a sterile, clear, colorless, preservative-free solution of the hydrochloride salt of levalbuterol, the (R)-enantiomer of the drug substance racemic albuterol. Levalbuterol HCl is a relatively selective beta 2 -adrenergic receptor agonist [see Clinical Pharmacology (12) ]. The chemical name for levalbuterol HCl is (R)-α 1 -[[(1,1-dimethylethyl)amino]methyl]-4-hydroxy-1,3-benzenedimethanol hydrochloride, and its established chemical structure is as follows:
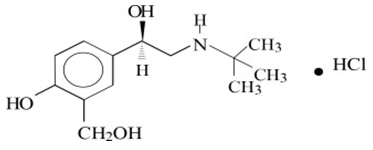
The molecular weight of levalbuterol HCl is 275.8, and its empirical formula is C 13 H 21 NO 3 ∙HCl. It is a white to off-white, crystalline solid, with a melting point of approximately 187°C and solubility of approximately 180 mg/mL in water.
Levalbuterol HCl is the USAN modified name for (R)-albuterol HCl in the United States.
Levalbuterol Inhalation Solution, USP is supplied in unit-dose vials and requires no dilution before administration by nebulization. Each 3 mL unit-dose vial contains 0.31 mg/3 mL (0.0103%) of levalbuterol (as 0.36 mg/3 mL of levalbuterol HCl) or 0.63 mg/3 mL (0.021%) of levalbuterol (as 0.73 mg/3 mL of levalbuterol HCl) or 1.25 mg/3 mL (0.042%) of levalbuterol (as 1.44 mg/3 mL of levalbuterol HCl), sodium chloride to adjust tonicity, edetate disodium (EDTA) as a stabilizer for the active pharmaceutical ingredient, and sulfuric acid to adjust the pH to 4.0 (3.3 to 4.5).
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Activation of beta 2 -adrenergic receptors on airway smooth muscle leads to the activation of adenylate cyclase and to an increase in the intracellular concentration of cyclic-3′, 5′-adenosine monophosphate (cyclic AMP). The increase in cyclic AMP is associated with the activation of protein kinase A, which in turn inhibits the phosphorylation of myosin and lowers intracellular ionic calcium concentrations, resulting in muscle relaxation. Levalbuterol relaxes the smooth muscles of all airways, from the trachea to the terminal bronchioles. Increased cyclic AMP concentrations are also associated with the inhibition of release of mediators from mast cells in the airway. Levalbuterol acts as a functional antagonist to relax the airway irrespective of the spasmogen involved, thus protecting against all bronchoconstrictor challenges. While it is recognized that beta 2 -adrenergic receptors are the predominant receptors on bronchial smooth muscle, data indicate that there are beta-receptors in the human heart, 10% to 50% of which are beta 2 -adrenergic receptors. The precise function of these receptors has not been established [see Warnings and Precautions (5.4) ]. However, all beta-adrenergic agonist drugs can produce a significant cardiovascular effect in some patients, as measured by pulse rate, blood pressure, symptoms, and/or electrocardiographic changes.
12.2 Pharmacodynamics
Adults and Adolescents ≥ 12 Years Old
In a randomized, double-blind, placebo-controlled, cross-over study, 20 adults with mild-to-moderate asthma received single doses of Levalbuterol Inhalation Solution, USP (0.31 mg, 0.63 mg, and 1.25 mg) and racemic albuterol sulfate inhalation solution (2.5 mg). All doses of active treatment produced a significantly greater degree of bronchodilation (as measured by percent change from pre-dose mean FEV 1 ) than placebo, and there were no significant differences between any of the active treatment arms. The bronchodilator responses to 1.25 mg of Levalbuterol Inhalation Solution, USP and 2.5 mg of racemic albuterol sulfate inhalation solution were clinically comparable over the 6-hour evaluation period, except for a slightly longer duration of action (> 15% increase in FEV 1 from baseline) after administration of 1.25 mg of Levalbuterol Inhalation Solution, USP. Systemic beta-adrenergic adverse effects were observed with all active doses and were generally dose-related for (R)-albuterol. Levalbuterol Inhalation Solution, USP at a dose of 1.25 mg produced a slightly higher rate of systemic beta-adrenergic adverse effects than the 2.5 mg dose of racemic albuterol sulfate inhalation solution.
In a randomized, double-blind, placebo-controlled, cross-over study, 12 adults with mild-to-moderate asthma were challenged with inhaled methacholine chloride 20 and 180 minutes following administration of a single dose of 2.5 mg of racemic albuterol sulfate, 1.25 mg of Levalbuterol Inhalation Solution, USP, 1.25 mg of (S)-albuterol, or placebo using a Pari LC Jet™ nebulizer. Racemic albuterol sulfate, Levalbuterol Inhalation Solution, USP, and (S)-albuterol had a protective effect against methacholine-induced bronchoconstriction 20 minutes after administration, although the effect of (S)-albuterol was minimal. At 180 minutes after administration, the bronchoprotective effect of 1.25 mg of Levalbuterol Inhalation Solution, USP was comparable to that of 2.5 mg of racemic albuterol sulfate. At 180 minutes after administration, 1.25 mg of (S)-albuterol had no bronchoprotective effect.
In a clinical study in adults with mild-to-moderate asthma, comparable efficacy (as measured by change from baseline FEV 1 ) and safety (as measured by heart rate, blood pressure, ECG, serum potassium, and tremor) were demonstrated after a cumulative dose of 5 mg of Levalbuterol Inhalation Solution, USP (four consecutive doses of 1.25 mg administered every 30 minutes) and 10 mg of racemic albuterol sulfate inhalation solution (four consecutive doses of 2.5 mg administered every 30 minutes).
12.3 Pharmacokinetics
Adults and Adolescents ≥ 12 Years Old
The inhalation pharmacokinetics of Levalbuterol Inhalation Solution, USP were investigated in a randomized cross-over study in 30 healthy adults following administration of a single dose of 1.25 mg and a cumulative dose of 5 mg of Levalbuterol Inhalation Solution, USP and a single dose of 2.5 mg and a cumulative dose of 10 mg of racemic albuterol sulfate inhalation solution by nebulization using a PARI LC Jet™ nebulizer with a Dura-Neb ® 2000 compressor.
Following administration of a single 1.25 mg dose of Levalbuterol Inhalation Solution, USP, exposure to (R)-albuterol (AUC of 3.3 ng∙hr/mL) was approximately 2-fold higher than following administration of a single 2.5 mg dose of racemic albuterol inhalation solution (AUC of 1.7 ng∙hr/mL) (see Table 6 ). Following administration of a cumulative 5 mg dose of Levalbuterol Inhalation Solution, USP (1.25 mg given every 30 minutes for a total of four doses) or a cumulative 10 mg dose of racemic albuterol inhalation solution (2.5 mg given every 30 minutes for a total of four doses), C max and AUC of (R)-albuterol were comparable (see Table 6 ).
| Single Dose | Cumulative Dose | |||
|---|---|---|---|---|
| Levalbuterol Inhalation Solution, USP 1.25 mg | Racemic albuterol sulfate 2.5 mg | Levalbuterol Inhalation Solution, USP 5 mg | Racemic albuterol sulfate 10 mg | |
| C max (ng/mL) (R)-albuterol | 1.1 (0.45) | 0.8 (0.41) Values reflect only (R)-albuterol and do not include (S)-albuterol. | 4.5 (2.20) | 4.2 (1.51) |
| T max (h) Median (Min, Max) reported for Tmax. (R)-albuterol | 0.2 (0.17, 0.37) | 0.2 (0.17, 1.50) | 0.2 (-0.18 A negative Tmax indicates Cmax occurred between first and last nebulizations. , 1.25) | 0.2 (-0.28, 1.00) |
| AUC (ng∙h/mL) (R)-albuterol | 3.3 (1.58) | 1.7 (0.99) | 17.4 (8.56) | 16.0 (7.12) |
| T ½ (h) (R)-albuterol | 3.3 (2.48) | 1.5 (0.61) | 4.0 (1.05) | 4.1 (0.97) |
Children 6-11 Years Old
The pharmacokinetic parameters of (R)-and (S)-albuterol in children with asthma were obtained using population pharmacokinetic analysis. These data are presented in Table 7. For comparison, adult data obtained by conventional pharmacokinetic analysis from a different study also are presented in Table 7.
In children, AUC and C max of (R)-albuterol following administration of 0.63 mg Levalbuterol Inhalation Solution, USP were comparable to those following administration of 1.25 mg racemic albuterol sulfate inhalation solution.
When the same dose of 0.63 mg of Levalbuterol Inhalation Solution, USP was given to children and adults, the predicted C max of (R)-albuterol in children was similar to that in adults (0.52 vs. 0.56 ng/mL), while predicted AUC in children (2.55 ng∙hr/mL) was about 1.5-fold higher than that in adults (1.65 ng∙hr/mL). These data support lower doses for children 6-11 years old compared with the adult doses [see Dosage and Administration (2) ].
| Children 6-11 years | Adults ≥ 12 years | |||||
|---|---|---|---|---|---|---|
| Treatment | Levalbuterol Inhalation Solution, USP 0.31 mg | Levalbuterol Inhalation Solution, USP 0.63 mg | Racemic albuterol 1.25 mg | Racemic albuterol 2.5 mg | Levalbuterol Inhalation Solution, USP 0.63 mg | Levalbuterol Inhalation Solution, USP 1.25 mg |
| AUC 0-∞ (ng∙hr/mL) Area under the plasma concentration curve from time 0 to infinity | 1.36 | 2.55 | 2.65 | 5.02 | 1.65 The values are predicted by assuming linear pharmacokinetics | 3.3 The data obtained from Table 6 |
| C max (ng/mL) Maximum plasma concentration | 0.303 | 0.521 | 0.553 | 1.08 | 0.56 | 1.1 |
Metabolism and Elimination
Information available in the published literature suggests that the primary enzyme responsible for the metabolism of albuterol enantiomers in humans is SULT1A3 (sulfotransferase). When racemic albuterol was administered either intravenously or via inhalation after oral charcoal administration, there was a 3- to 4-fold difference in the area under the concentration-time curves between the (R)-and (S)-albuterol enantiomers, with (S)-albuterol concentrations being consistently higher. However, without charcoal pretreatment, after either oral or inhalation administration the differences were 8- to 24-fold, suggesting that (R)-albuterol is preferentially metabolized in the gastrointestinal tract, presumably by SULT1A3.
The primary route of elimination of albuterol enantiomers is through renal excretion (80% to 100%) of either the parent compound or the primary metabolite. Less than 20% of the drug is detected in the feces. Following intravenous administration of racemic albuterol, between 25% and 46% of the (R)-albuterol fraction of the dose was excreted as unchanged (R)-albuterol in the urine.
Special Populations
Hepatic Impairment
The effect of hepatic impairment on the pharmacokinetics of Levalbuterol Inhalation Solution, USP has not been evaluated.
Renal Impairment
The effect of renal impairment on the pharmacokinetics of racemic albuterol was evaluated in 5 subjects with creatinine clearance of 7 to 53 mL/min, and the results were compared with those from healthy volunteers. Renal disease had no effect on the half-life, but there was a 67% decline in racemic albuterol clearance. Caution should be used when administering high doses of Levalbuterol Inhalation Solution, USP to patients with renal impairment [see Use in Specific Populations (8.6) ].
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Although there have been no carcinogenesis studies with levalbuterol HCl, racemic albuterol sulfate has been evaluated for its carcinogenic potential.
In a 2-year study in Sprague-Dawley rats, dietary administration of racemic albuterol sulfate resulted in a significant dose-related increase in the incidence of benign leiomyomas of the mesovarium at doses of 2 mg/kg/day and greater (approximately 4 times the MRHDID of levalbuterol HCl for adults and approximately 5 times the MRHDID of levalbuterol HCl for children on a mg/m 2 basis). In an 18-month study in CD-1 mice and a 22-month study in the golden hamster, dietary administration of racemic albuterol sulfate showed no evidence of tumorigenicity. Dietary doses in CD-1 mice were up to 500 mg/kg/day (approximately 540 times the MRHDID of levalbuterol HCl for adults and approximately 630 times the MRHDID of levalbuterol HCl for children on a mg/m 2 basis) and doses in the golden hamster study were up to 50 mg/kg/day (approximately 90 times the MRHDID of levalbuterol HCl for adults on a mg/m 2 basis and approximately 105 times the MRHDID of levalbuterol HCl for children on a mg/m 2 basis).
Levalbuterol HCl was not mutagenic in the Ames test or the CHO/HPRT Mammalian Forward Gene Mutation Assay. Levalbuterol HCl was not clastogenic in the in vivo micronucleus test in mouse bone marrow. Racemic albuterol sulfate was not clastogenic in an in vitro chromosomal aberration assay in CHO cell cultures.
No fertility studies have been conducted with levalbuterol hydrochloride. Reproduction studies in rats using racemic albuterol sulfate demonstrated no evidence of impaired fertility at oral doses up to 50 mg/kg/day (approximately 108 times the maximum recommended daily inhalation dose of levalbuterol HCl for adults on a mg/m 2 basis).
14 CLINICAL STUDIES
Adults and Adolescents ≥ 12 Years Old
The safety and efficacy of Levalbuterol Inhalation Solution, USP were evaluated in a 4-week, multicenter, randomized, double-blind, placebo-controlled, parallel-group study in 362 adult and adolescent patients 12 years of age and older, with mild-to-moderate asthma (mean baseline FEV 1 60% of predicted). Approximately half of the patients were also receiving inhaled corticosteroids. Patients were randomized to receive Levalbuterol Inhalation Solution, USP 0.63 mg, Levalbuterol Inhalation Solution, USP 1.25 mg, racemic albuterol sulfate 1.25 mg, racemic albuterol sulfate 2.5 mg, or placebo three times a day administered via a PARI LC Plus™ nebulizer and a Dura-Neb ® portable compressor. Racemic albuterol delivered by a chlorofluorocarbon (CFC) metered-dose inhaler (MDI) was used on an as-needed basis as the rescue medication.
Efficacy, as measured by the mean percent change from baseline FEV 1 , was demonstrated for all active treatment regimens compared with placebo on day 1 and day 29. On both day 1 (see Figure 1 ) and day 29 (see Figure 2 ), 1.25 mg of Levalbuterol Inhalation Solution, USP demonstrated the largest mean percent change from baseline FEV 1 compared with the other active treatments. A dose of 0.63 mg of Levalbuterol Inhalation Solution, USP and 2.5 mg of racemic albuterol sulfate produced a clinically comparable mean percent change from baseline FEV 1 on both day 1 and day 29.
| Figure 1: Mean Percent Change from Baseline FEV 1 on Day 1, Adults and Adolescents ≥ 12 years old |
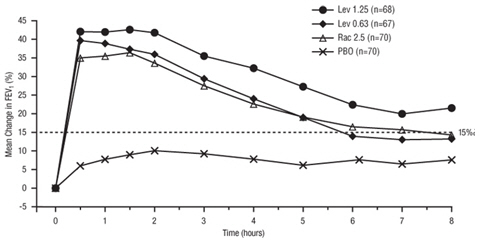 |
| Figure 2: Mean Percent Change from Baseline FEV 1 on Day 29, Adults and Adolescents ≥ 12 years old |
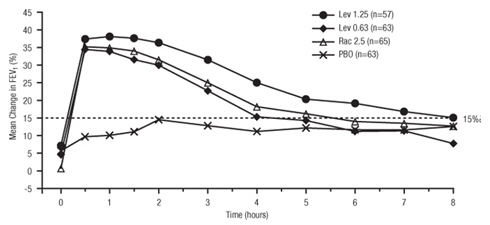 |
The mean time to onset of a 15% increase in FEV 1 over baseline for levalbuterol at doses of 0.63 mg and 1.25 mg was approximately 17 minutes and 10 minutes, respectively, and the mean time to peak effect for both doses was approximately 1.5 hours after 4 weeks of treatment. The mean duration of effect, as measured by a > 15% increase from baseline FEV 1 , was approximately 5 hours after administration of 0.63 mg of levalbuterol and approximately 6 hours after administration of 1.25 mg of levalbuterol after 4 weeks of treatment. In some patients, the duration of effect was as long as 8 hours.
Children 6-11 Years Old
A multicenter, randomized, double-blind, placebo-and active-controlled study was conducted in children with mild-to-moderate asthma (mean baseline FEV 1 73% of predicted) (n = 316). Following a 1-week placebo run-in, subjects were randomized to Levalbuterol Inhalation Solution, USP (0.31 mg or 0.63 mg), racemic albuterol (1.25 mg or 2.5 mg), or placebo, which were delivered three times a day for 3 weeks using a PARI LC Plus™ nebulizer and a Dura-Neb ® 3000 compressor.
Efficacy, as measured by mean peak percent change from baseline FEV 1 , was demonstrated for all active treatment regimens compared with placebo on day 1 and day 21. Time profile FEV 1 curves for day 1 and day 21 are shown in Figure 3 and Figure 4, respectively. The onset of effect (time to a 15% increase in FEV 1 over test-day baseline) and duration of effect (maintenance of a > 15% increase in FEV 1 over test-day baseline) of levalbuterol were clinically comparable to those of racemic albuterol.
| Figure 3: Mean Percent Change from Baseline FEV 1 on Day 1, Children 6-11 Years of Age |
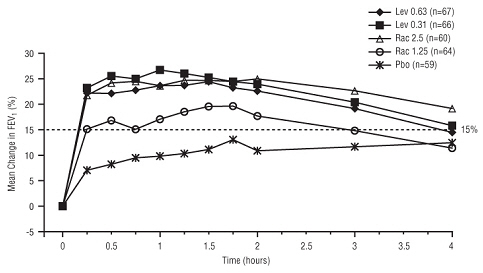 |
| Figure 4: Mean Percent Change from Baseline FEV 1 on Day 21, Children 6-11 Years of Age |
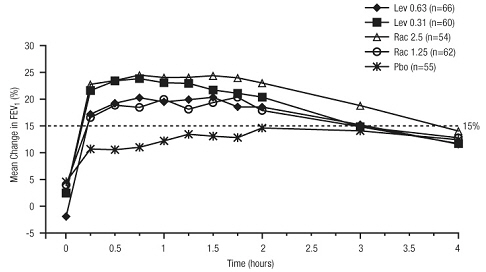 |
16 HOW SUPPLIED/STORAGE AND HANDLING
Levalbuterol Inhalation Solution, USP is supplied in 3 mL unit-dose, low-density polyethylene (LDPE) vials as a clear, colorless, sterile, preservative-free, aqueous solution, in three different strengths of levalbuterol (0.31 mg, 0.63 mg, 1.25 mg). Each strength of Levalbuterol Inhalation Solution, USP is available in a shelf-carton containing one or more foil pouches, each pouch containing one or more unit-dose LDPE vials.
Levalbuterol Inhalation Solution, USP, 0.31 mg/3 mL ( foil pouch label color green ) contains 0.31 mg/3 mL (0.0103%) of levalbuterol (as 0.36 mg/3 mL of levalbuterol HCl) and is available in cartons as listed below.
NDC 76204-700-01 30 vials per carton / 1 vial per foil pouch
NDC 76204-700-55 25 vials per carton / 5 vials per foil pouch
NDC 76204-700-25 25 vials per carton / 25 vials per foil pouch
Levalbuterol Inhalation Solution, USP, 0.63 mg/3 mL ( foil pouch label color yellow ) contains 0.63 mg/3 mL (0.021%) of levalbuterol (as 0.73 mg/3 mL of levalbuterol HCl) and is available in cartons as listed below.
NDC 76204-800-01 30 vials per carton / 1 vial per foil pouch
NDC 76204-800-55 25 vials per carton / 5 vials per foil pouch
NDC 76204-800-25 25 vials per carton / 25 vials per foil pouch
Levalbuterol Inhalation Solution, USP, 1.25 mg/3 mL ( foil pouch label color red ) contains 1.25 mg/3 mL (0.042%) of levalbuterol (as 1.44 mg/3 mL of levalbuterol HCl) and is available in cartons as listed below.
NDC 76204-900-01 30 vials per carton / 1 vial per foil pouch
NDC 76204-900-55 25 vials per carton / 5 vials per foil pouch
NDC 76204-900-25 25 vials per carton / 25 vials per foil pouch
Store Levalbuterol Inhalation Solution, USP in the protective foil pouch at 20°- 25°C (68°- 77°F) [see USP Controlled Room Temperature]. Protect from light and excessive heat. Keep unopened vials in the foil pouch. Once the foil pouch is opened, the vials should be used within 2 weeks. Vials removed from the pouch, if not used immediately, should be protected from light and used within 1 week. Discard any vial if the solution is not colorless.
Rx only
Instructions for Using Levalbuterol Inhalation Solution, USP
Levalbuterol Inhalation Solution, USP vial (see Figure A ):
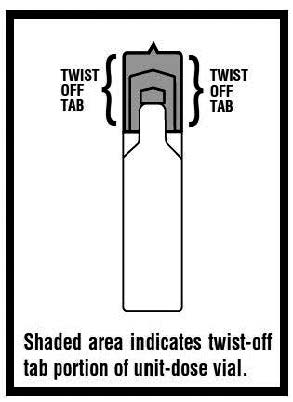
Using your Levalbuterol Inhalation Solution, USP:
Read the following Steps before using your Levalbuterol Inhalation Solution, USP. If you have any questions, ask your doctor or pharmacist.
Step 1. Open the foil pouch by tearing the notched edge along the seam of the pouch ( see Figure B ). Remove 1 vial to be used right away. Keep the rest of the unused vials in the foil pouch to protect them from light and heat.
Step 2. Hold the vial in your hands. Make sure your thumb and finger cover the twist-off tabs below the X-top ( see Figure C ).

Step 3. While holding the top firmly between your thumb and finger, twist the body of the vial to open the vial ( see Figure C ).
Step 4. Throw away the top of the vial and squeeze the entire contents of the vial into the nebulizer reservoir ( see Figure D ).
Step 5. Connect the nebulizer reservoir to the mouthpiece ( see Figure E.1 ) or face mask ( see Figure E.2 ).
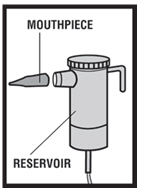 | 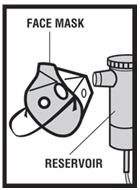 |
| Figure E.1 | Figure E.2. |
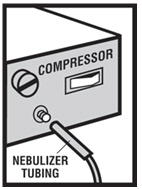
Step 6. Connect the nebulizer to the compressor ( see Figure F ).
Step 7. Sit in a comfortable, upright position. Place the mouthpiece in your mouth ( see Figure G.1 ) or put on your face mask ( see Figure G.2 ). Turn on the compressor.
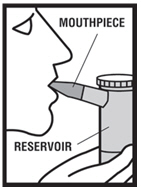 | 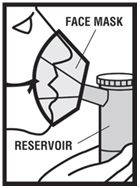 |
| Figure G.1 | Figure G.2 |
Step 8. Breathe as calmly, deeply, and evenly as possible until no more mist is seen in the nebulizer reservoir. Your treatment will take about 5 to 15 minutes. When you do not see any mist in the nebulizer reservoir, your treatment is finished.
Step 9. Clean and store your nebulizer. See the manufacturer's instructions that come with your nebulizer for how to clean and store your nebulizer.
This Patient Information Leaflet and Instructions for Use have been approved by the U.S. Food and Drug Administration.
12.1 Mechanism of Action
Activation of beta 2 -adrenergic receptors on airway smooth muscle leads to the activation of adenylate cyclase and to an increase in the intracellular concentration of cyclic-3′, 5′-adenosine monophosphate (cyclic AMP). The increase in cyclic AMP is associated with the activation of protein kinase A, which in turn inhibits the phosphorylation of myosin and lowers intracellular ionic calcium concentrations, resulting in muscle relaxation. Levalbuterol relaxes the smooth muscles of all airways, from the trachea to the terminal bronchioles. Increased cyclic AMP concentrations are also associated with the inhibition of release of mediators from mast cells in the airway. Levalbuterol acts as a functional antagonist to relax the airway irrespective of the spasmogen involved, thus protecting against all bronchoconstrictor challenges. While it is recognized that beta 2 -adrenergic receptors are the predominant receptors on bronchial smooth muscle, data indicate that there are beta-receptors in the human heart, 10% to 50% of which are beta 2 -adrenergic receptors. The precise function of these receptors has not been established [see Warnings and Precautions (5.4) ]. However, all beta-adrenergic agonist drugs can produce a significant cardiovascular effect in some patients, as measured by pulse rate, blood pressure, symptoms, and/or electrocardiographic changes.