Get your patient on Levonorgestrel And Ethinyl Estradiol - Levonorgestrel And Ethinyl Estradiol (Levonorgestrel And Ethinyl Estradiol)
Levonorgestrel And Ethinyl Estradiol - Levonorgestrel And Ethinyl Estradiol prescribing information
Cigarette smoking increases the risk of serious cardiovascular side effects from oral- contraceptive use. This risk increases with age and with heavy smoking (15 or more cigarettes per day) and is quite marked in women over 35 years of age. Women who use oral contraceptives should be strongly advised not to smoke.
Indications and usage
Oral contraceptives are indicated for the prevention of pregnancy in women who elect to use this product as a method of contraception. Oral contraceptives are highly effective. Table I lists the typical accidental pregnancy rates for users of combination oral contraceptives and other methods of contraception. The efficacy of these contraceptive methods, except sterilization and the IUD, depends upon the reliability with which they are used. Correct and consistent use of methods can result in lower failure rates.
TABLE I: PERCENTAGE OF WOMEN EXPERIENCING AN UNINTENDED PREGNANCY DURING THE FIRST YEAR OF USE OF A CONTRACEPTIVE METHOD
NA - not available | ||
| Method | Perfect use | Typical use |
| Levonorgestrel implants | 0.05 | 0.05 |
| Male sterilization | 0.1 | 0.15 |
| Female sterilization | 0.5 | 0.5 |
| Depo-Provera ® | ||
| (injectable progestogen) | 0.3 | 0.3 |
| Oral contraceptives | 5 | |
| Combined | 0.1 | NA |
| Progestin only | 0.5 | NA |
| IUD | ||
| Progesterone | 1.5 | 2.0 |
| Copper T 380A | 0.6 | 0.8 |
| Condom | ||
| (male) without spermicide | 3 | 14 |
| (female) without spermicide | 5 | 21 |
| Cervical cap | ||
| Nulliparous women | 9 | 20 |
| Parous women | 26 | 40 |
| Vaginal sponge | ||
| Nulliparous women | 9 | 20 |
| Parous women | 20 | 40 |
| Diaphragm with | ||
| spermicidal cream or jelly | 6 | 20 |
| Spermicides alone | ||
| (foam, creams, jellies, and vaginal suppositories) | 6 | 26 |
| Periodic abstinence (all methods) | 1 to 9 • | 25 |
| Withdrawal | 4 | 19 |
| No contraception (planned pregnancy) | 85 | 85 |
Dosage and Administration
To achieve maximum contraceptive effectiveness, levonorgestrel and ethinyl estradiol (tablets triphasic regimen) must be taken exactly as directed and at intervals not exceeding 24 hours. Levonorgestrel and ethinyl estradiol tablets are a three-phase preparation plus 7 inert tablets. The dosage of levonorgestrel and ethinyl estradiol tablets is one tablet daily for 28 consecutive days per menstrual cycle in the following order; 6 light blue tablets (phase 1), followed by 5 white to off white tablets (phase 2), followed by 10 pink tablets (phase 3), plus 7 orange inert tablets, according to the prescribed schedule.
It is recommended that levonorgestrel and ethinyl estradiol tablets be taken at the same time each day, preferably after the evening meal or at bedtime. During the first cycle of medication, the patient should be instructed to take one levonorgestrel and ethinyl estradiol tablet daily in the order of 6 light blue, 5 white to off white, 10 pink tablets, and then 7 orange inert tablets for twenty-eight (28) consecutive days, beginning on day one (1) of her menstrual cycle. (The first day of menstruation is day one.) Withdrawal bleeding usually occurs within 3 days following the last pink tablet. (If levonorgestrel and ethinyl estradiol tablets are first taken later than the first day of the first menstrual cycle of medication or postpartum, contraceptive reliance should not be placed on levonorgestrel and ethinyl estradiol tablets until after the first 7 consecutive days of administration. The possibility of ovulation and conception prior to initiation of medication should be considered.)
When switching from another oral contraceptive, levonorgestrel and ethinyl estradiol tablets should be started on the first day of bleeding following the last active tablet taken of the previous oral contraceptive.
The patient begins her next and all subsequent 28-day courses of levonorgestrel and ethinyl estradiol tablets on the same day of the week that she began her first course, following the same schedule. She begins taking her light blue tablets on the next day after ingestion of the last orange tablet, regardless of whether or not a menstrual period has occurred or is still in progress. Any time a subsequent cycle of levonorgestrel and ethinyl estradiol tablets is started later than the next day, the patient should be protected by another means of contraception until she has taken a tablet daily for seven consecutive days.
If spotting or breakthrough bleeding occurs, the patient is instructed to continue on the same regimen. This type of bleeding is usually transient and without significance; however, if the bleeding is persistent or prolonged, the patient is advised to consult her physician. Although the occurrence of pregnancy is highly unlikely if levonorgestrel and ethinyl estradiol tablets are taken according to directions, if withdrawal bleeding does not occur, the possibility of pregnancy must be considered. If the patient has not adhered to the prescribed schedule (missed one or more tablets or started taking them on a day later than she should have), the probability of pregnancy should be considered at the time of the first missed period and appropriate diagnostic measures taken before the medication is resumed. If the patient has adhered to the prescribed regimen and misses two consecutive periods, pregnancy should be ruled out before continuing the contraceptive regimen.
The risk of pregnancy increases with each active (light blue, white to off white, or pink) tablet missed. For additional patient instructions regarding missed pills, see the "WHAT TO DO IF YOU MISS PILLS" section in the DETAILED PATIENT LABELING below. If breakthrough bleeding occurs following missed active tablets, it will usually be transient and of no consequence. If the patient misses one or more orange tablets, she is still protected against pregnancy provided she begins taking light blue tablets on the again on the proper day.
In the nonlactating mother, Levonorgestrel and ethinyl estradiol tablets may be initiated postpartum, for contraception. When the tablets are administered in the postpartum period, the increased risk of thromboembolic disease associated with the postparturn period must be considered (see "Contraindications," "Warnings," and "Precautions" concerning thromboembolic disease). It is to be noted that early resumption of ovulation may occur if Parlodel ® (bromocriptine mesylate) has been used for the prevention of lactation.
Contraindications
Levonorgestrel and Ethinyl Estradiol Tablet is contraindicated in females who are known to have or develop the following conditions: Thrombophlebitis or thromboembolic disorders
A past history of deep-vein-thrombophlebitis or thromboembolic disorders.
Cerebral-vascular or coronary-artery disease.
Current diagnosis or history of breast cancer, which may be hormone sensitive.
Undiagnosed abnormal genital bleeding.
Cholestatic jaundice of pregnancy or jaundice with prior pill use.
Hepatic adenomas and carcinomas.
Known or suspected pregnancy.
Women who are receiving Hepatitis C drug combinations containing ombitasvir/paritaprevir/ritonavir, with or without dasabuvir, due to the potential for alanine aminotransferase (ALT) elevations (see WARNINGS, RISK OF LIVER ENZYME ELEVATIONS WITH CONCOMITANT HEPATITIS C TREATMENT ).
Adverse Reactions
Postmarketing Experience:
Five studies that compared breast cancer risk between ever-users (current or past use) of COCs and never-users of COCs reported no association between ever use of COCs and breast cancer risk, with effect estimates ranging from 0.90 - 1.12 (Figure 1).
Three studies compared breast cancer risk between current or recent COC users (<6 months since last use) and never users of COCs (Figure 1). One of these studies reported no association between breast cancer risk and COC use. The other two studies found an increased relative risk of 1.19 – 1.33 with current or recent use. Both of these studies found an increased risk of breast cancer with current use of longer duration, with relative risks ranging from 1.03 with less than one year of COC use to approximately 1.4 with more than 8-10 years of COC use.
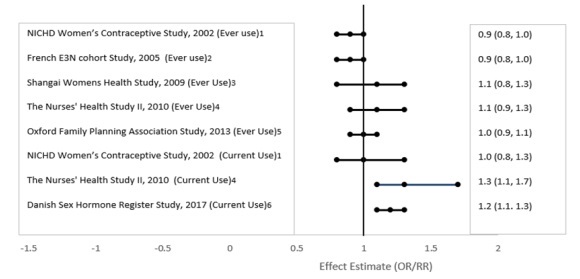
RR = relative risk; OR = odds ratio; HR = hazard ratio. "ever COC" are females with current or past COC use; "never COC use" are females that never used COCs.
An increased risk of the following serious adverse reactions has been associated with the use of oral- contraceptives (see "Warnings" section):
Thrombophlebitis.
Arterial thromboembolism.
Pulmonary embolism.
Myocardial infarction.
Cerebral hemorrhage.
Cerebral thrombosis.
Hypertension.
Gallbladder disease.
Hepatic adenomas or benign liver tumors.
There is evidence of an association between the following conditions and the use of oral contraceptives, although additional confirmatory studies are needed:
Mesenteric thrombosis.
Retinal thrombosis.
The following adverse reactions have been reported in patients receiving oral contraceptives and are believed to be drug related:
Nausea. Vomiting.
Gastrointestinal symptoms (such as abdominal cramps and bloating).
Breakthrough bleeding.
Spotting.
Change in menstrual flow.
Amenorrhea.
Temporary infertility after discontinuation of treatment.
Edema.
Melasma which may persist.
Breast changes: tenderness, enlargement, secretion.
Change in weight (increase or decrease).
Change in cervical erosion and secretion.
Diminution in lactation when given immediately postpartum.
Cholestatic jaundice.
Migraine.
Rash (allergic).
Mental depression.
Reduced tolerance to carbohydrates.
Vaginal candidiasis.
Change in corneal curvature (steepening).
Intolerance to contact lenses.
The following adverse reactions have been reported in users of oral contraceptives, and the association has been neither confirmed nor refuted:
Congenital anomalies.
Premenstrual syndrome.
Cataracts.
Optic neuritis.
Changes in appetite.
Cystitis-like syndrome.
Headache.
Nervousness.
Dizziness.
Hirsutism.
Loss of scalp hair.
Erythema multiforme.
Erythema nodosum.
Hemorrhagic eruption.
Vaginitis.
Porphyria.
Impaired renal function.
Hemolytic uremic syndrome.
Budd-Chiari syndrome.
Acne.
Changes in libido.
Colitis.
Sickle-cell disease.
Cerebral-vascular disease with mitral valve prolapse.
Lupus-like syndromes.
Description
Each levonorgestrel and ethinyl estradiol tablets USP cycle of 28 tablets consists of three different drug phases as follows: Phase 1 comprised of 6 light blue tablets, each containing 0.05 mg of levonorgestrel (d(-)-13 beta-ethyl-17-alpha-ethinyl-17-beta-hydroxygon-4-en-3-one), a totally synthetic progestogen, and 0.03 mg of ethinyl estradiol (19-nor-17α-pregna-1,3,5(10)-trien-20-yne-3, 17-diol); phase 2 comprised of 5 white to off white tablets, each containing 0.075 mg levonorgestrel and 0.04 mg ethinyl estradiol; and phase 3 comprised of 10 pink tablets, each containing 0.125 mg levonorgestrel and 0.03 mg ethinyl estradiol; then followed by 7 orange inert tablets. The inactive ingredients present in the light blue, white to off white and pink tablets are corn starch, lactose monohydrate, magnesium stearate and povidone. Each light blue tablet also contains FD & C Blue #1. Each pink tablet also contains FD & C Red #40. Each inactive orange tablet contains the following inactive ingredients: croscarmellose sodium, FD&C Yellow #6, lactose monohydrate, magnesium stearate and microcrystalline cellulose.
Clinical Pharmacology
Mechanism of Action
Combination oral contraceptives prevent pregnancy primarily by suppressing ovulation.
How Supplied
Levonorgestrel and ethinyl estradiol tablets USP (tablets-triphasic regimen) are available in a blister containing 28 tablets (NDC 68180-857-71). Three such blisters are packed in a carton (NDC 68180-857-73). Each cycle contains 28 tablets as follows:
- Six light blue tablets containing 0.05 mg of levonorgestrel and 0.03 mg of ethinyl estradiol. The light blue tablets are round, uncoated, debossed with "LU" on one side and "W31" on the other side.
- Five white to off white tablets containing 0.075 mg of levonorgestrel and 0.04 mg of ethinyl estradiol. The white to off white tablets are round, uncoated, debossed with "LU" on one side and "W32" on the other side.
- Ten pink tablets containing 0.125 mg of levonorgestrel and 0.03 mg of ethinyl estradiol. The pink tablets are round, uncoated, debossed with "LU" on one side and "W33" on the other side.
- Seven orange inert tablets. The orange inert tablets are round, bevel edged tablets debossed with "LU" on one side and "T22" on the other side.
Storage
Store at 25°C (77°F); excursions permitted to 15 to 30°C (59 to 86°F) [see USP Controlled Room Temperature].
References available upon request.
The brands listed are trademarks of their respective owners and are not trademarks of Lupin Pharmaceuticals, Inc. The makers of these brands are not affiliated with and do not endorse Lupin Pharmaceuticals, Inc. or its products.

Distributed by:
Lupin Pharmaceuticals, Inc.
Naples, FL 34108
United States
Manufactured by:
Lupin Limited
Pithampur (M.P.) – 454 775
INDIA
Revised: June 2025