Get your patient on Lidocaine Hydrochloride - Lidocaine Hydrochloride solution (Lidocaine Hydrochloride)
Lidocaine Hydrochloride - Lidocaine Hydrochloride solution prescribing information
WARNING: Life-threatening and fatal events in infants and young children
Postmarketing cases of seizures, cardiopulmonary arrest, and death in patients under the age of 3 years have been reported with use of Lidocaine Hydrochloride Oral Topical Solution 2% (Viscous) when it was not administered in strict adherence to the dosing and administration recommendations. In the setting of teething pain, Lidocaine Hydrochloride Oral Topical Solution 2% (Viscous) should generally not be used. For other conditions, the use of the product in patients less than 3 years of age should be limited to those situations where safer alternatives are not available or have been tried but failed.
To decrease the risk of serious adverse events with use of Lidocaine Hydrochloride Oral Topical Solution 2% (Viscous), instruct caregivers to strictly adhere to the prescribed dose and frequency of administration and store the prescription bottle safely out of reach of children.
INDICATIONS AND USAGE
Lidocaine Hydrochloride Oral Topical Solution 2% (Viscous) is indicated for the production of topical anesthesia of irritated or inflamed mucous membranes of the mouth and pharynx. It is also useful for reducing gagging during the taking of X-ray pictures and dental impressions.
DOSAGE AND ADMINISTRATION
Adult:
The maximum recommended single dose of Lidocaine Hydrochloride Oral Topical Solution 2% (Viscous) for healthy adults should be such that the dose of lidocaine does not exceed 4.5 mg/kg or 2 mg/lb body weight and does not in any case exceed a total of 300 mg.
For symptomatic treatment of irritated or inflamed mucous membranes of the mouth and pharynx, the usual adult dose is one 15 mL undiluted. For use in the mouth, the solution should be swished around in the mouth and spit out. For use in the pharynx, the undiluted solution should be gargled and may be swallowed. This dose should not be administered at intervals of less than three hours, and not more than eight doses should be given in a 24-hour period. The dosage should be adjusted commensurate with the patient's age, weight and physical condition (see PRECAUTIONS ).
Pediatric:
Care must be taken to ensure correct dosage in all pediatric patients as there have been cases of overdose due to inappropriate dosing.
It is difficult to recommend a maximum dose of any drug for children since this varies as a function of age and weight. For children over 3 years of age who have a normal lean body mass and normal body development, the maximum dose is determined by the child's weight or age. For example: in a child of 5 years weighing 50 lbs., the dose of lidocaine hydrochloride should not exceed 75 to 100 mg (3.7 to 5 mL of Lidocaine Hydrochloride Oral Topical Solution 2% Viscous).
For infants and in children under 3 years of age, the solution should be accurately measured and no more than 1.2 mL be applied to the immediate area with a cotton-tipped applicator. Wait at least 3 hours before giving the next dose; a maximum of four doses may be given in a 12-hour period. Lidocaine Hydrochloride Oral Topical Solution 2% (Viscous) should only be used if the underlying condition requires treatment with a volume of product that is less than or equal to 1.2 mL.
CONTRAINDICATIONS
Lidocaine is contraindicated in patients with a known history of hypersensitivity to local anesthetics of the amide type, or to other components of the solution.
ADVERSE REACTIONS
Adverse experiences following the administration of lidocaine are similar in nature to those observed with other amide local anesthetic agents. These adverse experiences are, in general, dose-related and may result from high plasma levels caused by excessive dosage or rapid absorption, or may result from a hypersensitivity, idiosyncrasy or diminished tolerance on the part of the patient. Serious adverse experiences are generally systemic in nature. The following types are those most commonly reported:
Drug Interactions:
Patients who are administered local anesthetics are at increased risk of developing methemoglobinemia when concurrently exposed to the following drugs, which could include other local anesthetics:
Examples of Drugs Associated with Methemoglobinemia:
| Class | Examples |
| Nitrates/Nitrites | nitric oxide, nitroglycerin, nitroprusside, nitrous oxide |
| Local anesthetics | articaine, benzocaine, bupivacaine, lidocaine, mepivacaine, prilocaine, procaine, ropivacaine, tetracaine |
| Antineoplastic agents | cyclophosphamide, flutamide, hydroxyurea, ifosfamide, rasburicase |
| Antibiotics | dapsone, nitrofurantoin, para-aminosalicylic acid, sulfonamides |
| Antimalarials | chloroquine, primaquine |
| Anticonvulsants | phenobarbital, phenytoin, sodium valproate |
| Other drugs | acetaminophen, metoclopramide, quinine, sulfasalazine |
DESCRIPTION
Lidocaine Hydrochloride Oral Topical Solution USP, 2% (Viscous) contains a local anesthetic agent and is administered topically. Lidocaine Hydrochloride Oral Topical Solution USP, 2% (Viscous) contains lidocaine hydrochloride, which is chemically designated as acetamide, 2-(diethylamino)-N-(2,6-dimethylphenyl)-, monohydrochloride and has the following structural formula:
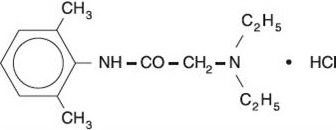
The molecular formula of lidocaine is C 14 H 22 N 2 O. The molecular weight is 234.34.
CLINICAL PHARMACOLOGY
Mechanism of Action:
Lidocaine stabilizes the neuronal membrane by inhibiting the ionic fluxes required for the initiation and conduction of impulses, thereby effecting local anesthetic action.
Hemodynamics:
Excessive blood levels may cause changes in cardiac output, total peripheral resistance, and mean arterial pressure. These changes may be attributable to a direct depressant effect of the local anesthetic agent on various components of the cardiovascular system. The net effect is normally a modest hypotension when the recommended dosages are not exceeded.
Pharmacokinetics and Metabolism:
Lidocaine is absorbed following topical administration to mucous membranes, its rate and extent of absorption being dependent upon concentration and total dose administered, the specific site of application, and duration of exposure. In general, the rate of absorption of local anestheticagents following topical application occurs most rapidly after intratracheal administration.
Lidocaine is also well-absorbed from the gastrointestinal tract, but little intact drug appears inthe circulation because of biotransformation in the liver. The plasma binding of lidocaine is dependent on drug concentration, and the fraction bound decreases with increasing concentration. At concentrations of 1 to 4 mcg of free base per mL, 60 to 80 percent of lidocaine is protein bound. Binding is also dependent on the plasma concentration of the alpha-1-acid glycoprotein.
Lidocaine crosses the blood-brain and placental barriers, presumably by passive diffusion.
Lidocaine is metabolized rapidly by the liver, and metabolites and unchanged drug are excreted by the kidneys. Biotransformation includes oxidative N-dealkylation, ring hydroxylation, cleavage of the amide linkage, and conjugation. N-dealkylation, a major pathway of biotransformation, yields the metabolites monoethylglycinexylidide and glycinexylidide. The pharmacological/toxicological actions of these metabolites are similar to, but less potent than, those of lidocaine. Approximately 90% of lidocaine administered is excreted in the form of various metabolites, and less than 10% is excreted unchanged. The primary metabolite in urine is a conjugate of 4-hydroxy-2, 6-dimethylaniline.
The elimination half-life of lidocaine following an intravenous bolus injection is typically 1.5 to 2 hours. Because of the rapid rate at which lidocaine is metabolized, any condition that affects liver function may alter lidocaine kinetics. The half-life may be prolonged two-fold or more in patients with liver dysfunction. Renal dysfunction does not affect lidocaine kinetics but may increase the accumulation of metabolites.
Factors such as acidosis and the use of CNS stimulants and depressants affect the CNS levels of lidocaine required to produce overt systemic effects. Objective adverse manifestations become increasingly apparent with increasing venous plasma levels above 6.0 mcg free baseper mL. In the rhesus monkey arterial blood levels of 18 to 21 mcg/mL have been shown to be threshold for convulsive activity.
HOW SUPPLIED
Lidocaine Hydrochloride Oral Topical Solution USP, 2% (Viscous) is a clear, colorless , viscous solution supplied in 100 mL low density polyethylene squeeze bottles. Supplied with press-in bottle adapter.
NDC 72888-125-26
Store at 20º to 25ºC (68º to 77ºF); excursions permitted to 15° to 30°C (59° to 86°F) [See USP Controlled Room Temperature].
SHAKE WELL BEFORE USE.
Lidocaine Hydrochloride Oral Topical Solution, USP Each ml contains 2% Lidocaine Instructions for Use Read these instructions carefully to learn how to use the medicine dispensing system correctly. | |
The Medicine Dispensing System | |
There are 2 parts to the dispensing system: | |
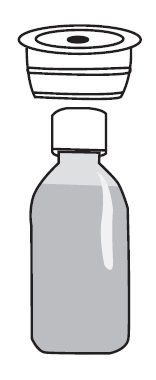 |
|
Preparing the Bottle | |
|
Note: Save the cap so you can close the bottle after each use.
Note: You may not be able to push the adapter fully down, but it will be forced into the bottle when you screw the cap back on. The adapter must always stay in the bottle. The child-resistant cap should seal the bottle in between use. |
Taking the Medicine | |
|
Note: Always replace the cap after use.
|
Distributed by: Advagen Pharma Ltd., East Windsor, NJ 08520, USA
Revised: 00, 10/2024
Mechanism of Action:
Lidocaine stabilizes the neuronal membrane by inhibiting the ionic fluxes required for the initiation and conduction of impulses, thereby effecting local anesthetic action.
Lidocaine Hydrochloride - Lidocaine Hydrochloride solution PubMed™ news
- Journal Article • 2026 MaySynthesis, spectroscopic analysis and physicochemical characterization of TOPO and lidocaine-based deep eutectic solvents with thermal stability evaluation via TGA/DTG.
- Journal Article • 2026 MayEfficacy of 0.5M mannitol as an adjuvant to lidocaine and epinephrine for intra-oral nerve blocks - a split-mouth, randomized controlled trial.
- Journal Article • 2026 MayHow to minimize pain with local anesthesia and improve patient experiences: a review.
- Journal Article • 2026 MayLocal anaesthesia with lidocaine is insufficient for ovine tail docking: A randomised clinical trial.
- Journal Article • 2026 MayLidocaine alleviates diabetic neuropathic pain by inhibiting spinal astrocyte activation through modulation of the CXCL13/CXCR5 axis.