Get your patient on Marplan (Isocarboxazid)
Marplan prescribing information
Suicidality and Antidepressant Drugs
Antidepressants increased the risk compared to placebo of suicidal thinking and behavior (suicidality) in children, adolescents, and young adults in short-term studies with Major Depressive Disorder (MDD) and other psychiatric disorders. Anyone considering the use of Marplan or any other antidepressant in a child, adolescent or young adult must balance this risk with the clinical need. Short-term studies did not show an increase in the risk of suicidality with antidepressants compared to placebo in adults beyond age 24; there was a reduction in risk with antidepressants compared to placebo in adults aged 65 and older. Depression and certain other psychiatric disorders are themselves associated with increases in the risk of suicide. Patients of all ages who are started on antidepressant therapy should be monitored appropriately and observed closely for clinical worsening, suicidality, or unusual changes in behavior. Families and caregivers should be advised of the need for close observation and communication with the prescriber. Marplan is not approved for use in pediatric patients (seeWarnings: Clinical Worsening and Suicide Risk ,Precautions: Information for Patients , andPrecautions: Pediatric Use ) .
Pooled analyses of short-term (4 to 16 weeks) placebo-controlled trials of 9 antidepressant drugs (SSRIs and others) in children and adolescents with major depressive disorder (MDD), obsessive compulsive disorder (OCD), or other psychiatric disorders (a total of 24 trials involving over 4400 patients) have revealed a greater risk of adverse events representing suicidal thinking or behavior (suicidality) during the first few months of treatment in those receiving antidepressants. The average risk of such events in patients receiving antidepressants was 4%, twice the placebo risk of 2%. No suicides occurred in these trials.
INDICATIONS AND USAGE
Marplan is indicated for the treatment of depression. Because of its potentially serious side effects, Marplan is not an antidepressant of first choice in the treatment of newly diagnosed depressed patients.
The efficacy of Marplan in the treatment of depression was established in 6-week controlled trials of depressed outpatients. These patients had symptoms that corresponded to the DSM-IV category of major depressive disorder; however, they often also had signs and symptoms of anxiety (anxious mood, panic, and/or phobic symptoms) ( see CLINICAL PHARMACOLOGY ).
A major depressive episode (DSM-IV) implies a prominent and relatively persistent (nearly every day for at least 2 weeks) depressed or dysphoric mood that usually interferes with daily functioning, and includes at least five of the following nine symptoms: depressed mood, loss of interest in usual activities, significant change in weight and/or appetite, insomnia or hypersomnia, psychomotor agitation or retardation, increased fatigue, feelings of guilt or worthlessness, slowed thinking or impaired concentration, and a suicide attempt or suicidal ideation.
The antidepressant effectiveness of Marplan in hospitalized depressed patients, or in endogenomorphically retarded and delusionally depressed patients, has not been adequately studied.
The effectiveness of Marplan in long-term use, that is, for more than 6 weeks, has not been systematically evaluated in controlled trials. Therefore, the physician who elects to use Marplan for extended periods should periodically evaluate the long-term usefulness of the drug for the individual patient.
DOSAGE AND ADMINISTRATION
For maximum therapeutic effect, the dosage of Marplan must be individually adjusted on the basis of careful observation of the patient. Dosage should be started with one tablet (10 mg) of Marplan twice daily. If tolerated, dosage may be increased by increments of one tablet (10 mg) every 2 to 4 days to achieve a dosage of four tablets daily (40 mg) by the end of the first week of treatment. Dosage can then be increased by increments of up to 20 mg/week, if needed and tolerated, to a maximum recommended dosage of 60 mg/day. Daily dosage should be divided into two to four dosages. After maximum clinical response is achieved, an attempt should be made to reduce the dosage slowly over a period of several weeks without jeopardizing the therapeutic response. Beneficial effect may not be seen in some patients for 3 to 6 weeks. If no response is obtained by then, continued administration is unlikely to help.
Because of the limited experience with systematically monitored patients receiving Marplan at the higher end of the currently recommended dose range of up to 60 mg/day, caution is indicated in patients for whom a dose of 40 mg/day is exceeded (see ADVERSE REACTIONS ).
CONTRAINDICATIONS
Marplan (isocarboxazid) should not be administered in combination with any of the following: MAO inhibitors or dibenzazepine derivatives; sympathomimetics (including amphetamines); some central nervous system depressants (including narcotics and alcohol); antihypertensive, diuretic, antihistaminic, sedative or anesthetic drugs, buproprion HCL, buspirone HCL, dextromethorphan, cheese or other foods with a high tyramine content; or excessive quantities of caffeine.
Marplan (isocarboxazid) should not be administered to any patient with a confirmed or suspected cerebrovascular defect or to any patient with cardiovascular disease, hypertension, or history of headache.
Contraindicated Patient Populations
Hypersensitivity Marplan should not be used in patients with known hypersensitivity to isocarboxazid.
Cerebrovascular Disorders Marplan should not be administered to any patient with a confirmed or suspected cerebrovascular defect or to any patient with cardiovascular disease or hypertension.
Pheochromocytoma Marplan should not be used in the presence of pheochromocytoma, as such tumors secrete pressor substances whose metabolism may be inhibited by Marplan.
Liver Disease Marplan should not be used in patients with a history of liver disease, or in those with abnormal liver function tests.
Renal Impairment Marplan should not be used in patients with severe impairment of renal function.
Contraindicated MAOI-Other Drug Combinations Other MAOI Inhibitors or With Dibenzazepine-Related Entities Marplan should not be administered together with, or in close proximity to, other MAO inhibitors or dibenzazepine-related entities. Hypertensive crises, severe convulsive seizures, coma, or circulatory collapse may occur in patients receiving such combinations.
In patients being transferred to Marplan from another MAO inhibitor or from a dibenzazepine-related entity, a medication-free interval of at least 1 week should be allowed, after which Marplan therapy should be started using half the normal starting dosage for at least the first week of therapy. Similarly, at least 1 week should elapse between the discontinuation of Marplan and initiation of another MAO inhibitor or dibenzazepine-related entity, or the readministration of Marplan. The following list includes some other MAO inhibitors, dibenzazepine-related entities, and tricyclic antidepressants.
| Generic Name | Trademark (Manufacturer) |
| Other MAO Inhibitors | |
| Furazolidone | Furoxone ® (Roberts Laboratories) |
| Pargyline HCL | Eutonyl ® (Abbott Laboratories) |
| Pargyline HCL and methyclothiazide | Eutron ® (Abbott Laboratories) |
| Phenelzine sulfate | Nardil ® (Parke-Davis) |
| Procarbazine | Matulane ® (Roche Laboratories) |
| Tranylcypromine sulfate | Parnate ® (SmithKline Beecham Pharmaceuticals) |
| Dibenzazepine-Related and Other Tricyclics | |
| Amitriptyline HCL | Elavil ® (Zeneca) |
| Endep ® (Roche Products) | |
| Perphenazine and amitriptyline HCL | Etrafon ® (Schering) |
| Triavil ® (Merck Sharp & Dohme) | |
| Clomipramine hydrochloride | Anafranil ® (Novartis) |
| Desipramine HCL | Norpramin ® (Hoechst Marion Roussel) |
| Pertofrane ® (Rhône-Poulenc Rorer Pharmaceuticals) | |
| Imipramine HCL | Janimine ® (Abbott Laboratories) |
| Tofranil ® (Novartis) | |
| Nortriptyline HCL | Aventyl ® (Eli Lilly & Co.) |
| Pamelor ® (Novartis) | |
| Protripyline HCL | Vivactil ® (Merck Sharp & Dohme) |
| Doxepin HCL | Adapin ® (Fisons) |
| Sinequan ® (Pfizer) | |
| Carbamazepine | Tegretol ® (Novartis) |
| Cyclobenzaprine HCL | Flexeril ® (Merck Sharp & Dohme) |
| Amoxapine | Asendin ® (Lederle) |
| Maprotiline HCL | Ludiomil ® (Novartis) |
| Trimipramine maleate | Surmontil ® (Wyeth-Ayerst Laboratories) |
Bupropion
The concurrent administration of a MAO inhibitor and buproprion hydrochloride (Wellbutrin ® , and Zyban ® , Glaxo Wellcome) is contraindicated. At least 14 days should elapse between discontinuation of an MAO inhibitor and initiation of treatment with buproprion hydrochloride.
Selective Serotonin Re - uptake Inhibitors (SSRIs)
Marplan should not be administered in combination with any SSRI. There have been reports of serious, sometimes fatal, reactions (including hyperthermia, rigidity, myoclonus, autonomic instability with possible rapid fluctuations of vital signs, and mental status changes that include extreme agitation and confusion progressing to delirium and coma) in patients receiving fluoxetine (Prozac ® , Lilly) in combination with a monoamine oxidase inhibitor (MAOI), and in patients who have recently discontinued fluoxetine and are then started on a MAOI. Some cases presented with features resembling neuroleptic malignant syndrome. Fluoxetine and other SSRIs should therefore not be used in combination with Marplan, or within 14 days of discontinuing therapy with Marplan. As fluoxetine and its major metabolite have very long elimination half-lives, at least 5 weeks should be allowed after stopping fluoxetine before starting Marplan. At least 2 weeks should be allowed after stopping sertraline (Zoloft ® , Pfizer) or paroxetine (Paxil ® , SmithKline Beecham Pharmaceuticals) before starting Marplan. In addition, there should be an interval of least 10 days between discontinuation of Marplan and initiation of fluoxetine or other SSRIs.
Buspirone
Marplan should not be used in combination with buspirone HCL (Buspar ® , Bristol Myers Squibb); several cases of elevated blood pressure have been reported in patients taking MAO inhibitors who were then given buspirone HCL. At least 10 days should elapse between the discontinuation of Marplan and the institution of buspirone HCL. Serious reactions may also occur when MAO inhibitors are given with serotoninergic drugs (e.g., dexfenfluramine, fluoxetine, fluvoxamine, paroxetine, sertraline, citalopram, venlafaxine).
Sympathomimetics
Marplan should not be administered in combination with sympathomimetics, including amphetamines, or with over-the-counter drugs such as cold, hay fever, or weight-reducing preparations that contain vasoconstrictors.
During Marplan therapy, it appears that some patients are particularly vulnerable to the effects of sympathomimetics when the activity of metabolizing enzymes is inhibited. Use of sympathomimetics and compounds such as guanethidine, methyldopa, methylphenidate, reserpine, epinephrine, norepinephrine, phenylalanine, dopamine, levodopa, tyrosine, and tryptophan with Marplan may precipitate hypertension, headache, and related symptoms. The combination of MAO inhibitors and tryptophan has been reported to cause behavioral and neurologic symptoms, including disorientation, confusion, amnesia, delirium, agitation, hypomanic signs, ataxia, myoclonus, hyperreflexia, shivering, ocular oscillations, and Babinski signs.
Meperidine
Meperidine should not be used concomitantly with MAO inhibitors or within 2- or 3-weeks following MAO therapy. Serious reactions have been precipitated with concomitant use, including coma, severe hypertension or hypotension, severe respiratory depression, convulsions, malignant hyperpyrexia, excitation, peripheral vascular collapse, and death. It is thought that these reactions may be mediated by accumulation of 5-HT (serotonin) consequent to MAO inhibition.
Dextromethorphan
Marplan should not be used in combination with dextromethorphan. The combination of MAO inhibitors and dextromethorphan has been reported to cause brief episodes of psychosis or bizarre behavior.
Cheese or Other Foods With a High Tyramine Content
Hypertensive crises have sometimes occurred during Marplan therapy after ingestion of foods with a high tyramine content. In general, patients should avoid protein foods in which aging or protein breakdown is used to increase flavor. In particular, patients should be instructed not to take foods such as cheese (particularly strong or aged varieties), sour cream, Chianti wine, sherry, beer (including non-alcoholic beer), liqueurs, pickled herring, anchovies, caviar, liver, canned figs, raisins, bananas or avocados (particularly if overripe), chocolate, soy sauce, sauerkraut, the pods of broad beans (fava beans), yeast extracts, yogurt, meat extracts, meat prepared with tenderizers, or dry sausage.
Anesthetic Agents
Patients taking Marplan should not undergo elective surgery requiring general anesthesia. Also, they should not be given cocaine or local anesthesia containing sympathomimetic vasoconstrictors. The possible combined hypotensive effects of Marplan and spinal anesthesia should be kept in mind. Marplan should be discontinued at least 10 days before elective surgery.
CNS Depressants
Marplan should not be used in combination with some central nervous system depressants, such as narcotics, barbiturates, or alcohol.
Antihypertensives
Marplan should not be used in combination with antihypertensive agents, including thiazide diuretics. A marked potentiating effect on these drugs has been reported, resulting in hypotension.
Caffeine
Excessive use of caffeine in any form should be avoided in patients receiving Marplan.
ADVERSE REACTIONS
Adverse Findings Observed in Short-Term, Placebo-Controlled Trials
Systematically collected data are available from only 86 patients exposed to Marplan, of whom only 52 received doses of ≥50 mg/day, including only 11 who were dosed at ≥60 mg/day. Because of the limited experience with systematically monitored patients receiving Marplan at the higher end of the currently recommended dose range of up to 60 mg/day, caution is indicated in patients for whom a dose of 40 mg/day is exceeded ( see WARNINGS ).
The table that follows enumerates the incidence, rounded to the nearest percent, of treatment emergent adverse events that occurred among 86 depressed patients who received Marplan at doses ranging from 20 to 80 mg/day in placebo-controlled trials of 6 weeks in duration. Events included are those occurring in 1% or more of patients treated with Marplan and for which the incidence in patients treated with Marplan was greater than the incidence in placebo-treated patients.
The prescriber should be aware that these figures cannot be used to predict the incidence of adverse events in the course of usual medical practice where patient characteristics and other factors differ from those which prevailed in clinical trials. Similarly, the cited frequencies cannot be compared with figures obtained from other clinical investigations involving different treatments, uses, and investigators. The cited figures, however, do provide the prescribing physician with some basis for estimating the relative contribution of drug and non-drug factors to the adverse event incidence rate in the population studied.
The commonly observed adverse event that occurred in Marplan patients with an incidence of 5% or greater and at least twice the incidence in placebo patients were nausea, dry mouth, and dizziness (see Table).
In three clinical trials for which the data were pooled, 4 of 85 (5%) patients who received placebo, 10 of 86 (12%) who received <50 mg of Marplan per day, and 1 of 52 (2%) who received ≥50 mg of Marplan per day prematurely discontinued treatment. The most common reasons for discontinuation were dizziness, orthostatic hypotension, syncope, and dry mouth.
| BODY SYSTEM/ ADVERSE EVENT | PLACEBO (N=85) | MARPLAN <50 mg (N=86) | MARPLAN ≥ 50 mg (N=52) 2 |
| MISCELLANEOUS | |||
| Drowsy | 0 | 4% | 0% |
| Anxiety | 1 | 2% | 0% |
| Chills | 0% | 2% | 0% |
| Forgetful | 1% | 2% | 2% |
| Hyperactive | 0% | 2% | 0% |
| Lethargy | 0% | 2% | 2% |
| Sedation | 1% | 2% | 0% |
| Syncope | 0% | 2% | 0% |
| INTEGUMENTARY | |||
| Sweating | 0% | 2% | 2% |
| MUSCULOSKELETAL | |||
| Heavy feeling | 0% | 2% | 0% |
| CARDIOVASCULAR | |||
| Orthostatic hypotension | 1% | 4% | 4% |
| Palpitations | 1% | 2% | 0% |
| GASTROINTESTINAL | |||
| Dry mouth | 4% | 9% | 6% |
| Constipation | 6% | 7% | 4% |
| Nausea | 2% | 6% | 4% |
| Diarrhea | 1% | 2% | 0% |
| UROGENITAL | |||
| Impotence | 0% | 2% | 0% |
| Urinary frequency | 1% | 2% | 0% |
| Urinary hesitancy | 0% | 1% | 4% |
| CENTRAL NERVOUS SYSTEM | |||
| Headache | 13% | 15% | 6% |
| Insomnia | 4% | 4% | 6% |
| Sleep disturbance | 0% | 5% | 2% |
| Tremor | 0% | 4% | 4% |
| Myoclonic jerks | 0% | 2% | 0% |
| Paresthesia | 1% | 2% | 0% |
| SPECIAL SENSES | |||
| Dizziness | 14% | 29% | 15% |
1 Events reported by at least 1% of patients treated with Marplan are presented, except for those that had an incidence on placebo greater than or equal to that on Marplan.
2 All patients also received Marplan at doses <50 mg.
Other Events Observed During the Post - marketing Evaluation of Marplan
Isolated cases of akathisia, ataxia, black tongue, coma, dysuria, euphoria, hematologic changes, incontinence, neuritis, photosensitivity, sexual disturbances, spider telangiectases, and urinary retention have been reported. These side effects sometimes necessitate discontinuation of therapy. In rare instances, hallucinations have been reported with high dosages, but they have disappeared upon reduction of dosage or discontinuation of therapy. Toxic amblyopia was reported in one psychiatric patient who had received isocarboxazid for about a year; no causal relationship to isocarboxazid was established. Impaired water excretion compatible with the syndrome of inappropriate secretion of antidiuretic hormone (SIADH) has been reported.
To report SUSPECTED ADVERSE REACTIONS, contact Lifsa Drugs LLC at 1-833-627-0070 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
See CONTRAINDICATIONS , WARNINGS , and PRECAUTIONS sections for information on drug interactions.
Marplan should be administered with caution to patients receiving Antabuse ® (disulfiram, Wyeth-Ayerst Laboratories). In a single study, rats given high intraperitoneal doses of an MAO inhibitor plus disulfiram experienced severe toxicity, including convulsions and death.
Concomitant use of Marplan and other psychotropic agents is generally not recommended because of possible potentiating effects. This is especially true in patients who may subject themselves to an overdosage of drugs. If combination therapy is needed, careful consideration should be given to the pharmacology of all agents to be used. The monoamine oxidase inhibitory effects of Marplan may persist for a substantial period after discontinuation of the drug, and this should be borne in mind when another drug is prescribed following Marplan. To avoid potentiation, the physician wishing to terminate treatment with Marplan and begin therapy with another agent should allow for an interval of 10 days.
DESCRIPTION
Marplan (isocarboxazid), a monoamine oxidase inhibitor, is available for oral administration in 10-mg tablets. Each tablet also contains lactose, corn starch, povidone, D&C Red No. 27, FD&C Yellow No. 6, and magnesium stearate. Chemically, isocarboxazid is 5-methyl-3-isoxazolecarboxylic acid 2-benzylhydrazide. The structural formula is:
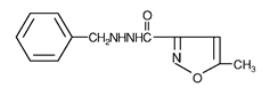
Isocarboxazid is a colorless, crystalline substance with very little taste.
CLINICAL PHARMACOLOGY
Pharmacodynamics
Isocarboxazid is a non-selective hydrazine monoamine oxidase (MAO) inhibitor. In vivo and in vitro studies demonstrated inhibition of MAO in the brain, heart, and liver. The mechanism by which MAO inhibitors act as antidepressants is not fully understood, but it is thought to involve the elevation of brain levels of biogenic amines. However, MAO is a complex enzyme system, widely distributed throughout the body, and drugs that inhibit MAO in the laboratory are associated with a number of clinical effects. Thus, it is unknown whether MAO inhibition per se, other pharmacologic actions, or an interaction of both is responsible for the antidepressant effects observed.
Pharmacokinetics
Marplan pharmacokinetic information is not available.
Clinical Efficacy Data
The effectiveness of Marplan was demonstrated in two 6-week placebo-controlled studies conducted in adult outpatients with depressive symptoms that corresponded to the DSM-IV category of major depressive disorder. The patients often also had signs and symptoms of anxiety (anxious mood, panic, and/or phobic symptoms). Patients were initiated with a dose of 10 mg bid, with increases every 2 to 4 days, as tolerated, until a therapeutic effect was achieved, up to a maximum dose of 80 mg/day. Doses were administered on a divided schedule ranging from 2 to 4 times a day. The mean dose overall for both studies was approximately 40 mg/day, with very few patients receiving doses greater than 60 mg/day. In both studies at the end of 6 weeks, patients receiving Marplan had significantly greater reduction in signs and symptoms of depression evaluated by the Hamilton Depression Scale, for both the Total Score and the Depressed Mood Score, than patients who received placebo.
HOW SUPPLIED
Marplan tablets 10 mg are peach colored, round and scored with "MARPLAN 10" imprinted on one side and scored on the other. Supplied in bottles of 100 (NDC 72336-032-01).
Storage: Store at 77ºF (25ºC); excursions permitted to 59º to 86ºF (15º to 30ºC) [See USP Controlled Room Temperature].
Dispense in a tight, light-resistant container as defined in USP.
Rx Only
© 2025 Lifsa Drugs LLC All rights reserved
LD-0001.01 DEC 2025