Get your patient on Meclizine Hydrochloride - Meclizine Hydrochloride tablet (Meclizine Hydrochloride)
Meclizine Hydrochloride - Meclizine Hydrochloride tablet prescribing information
INDICATIONS AND USAGE
Meclizine hydrochloride tablets are indicated for the treatment of vertigo associated with diseases affecting the vestibular system in adults.
DOSAGE FORMS AND STRENGTHS
- 12.5 mg, blue, modified oval-shaped tablets, de-bossed with “Є12” on one side and plain on the other side.
- 25 mg, white, modified oval-shaped tablets, de-bossed with “Є14” on one side, and plain on the other side.
USE IN SPECIFIC POPULATIONS
Pregnancy
Risk Summary
Data from epidemiological studies have not generally indicated a drug-associated risk of major birth defects with meclizine during pregnancy. However, in a published study, an increased incidence of fetal malformations was observed following oral administration of meclizine to pregnant rats during the period of organogenesis, at doses similar to those used clinically.
In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4% and 15 to 20%, respectively. The background risk of major birth defects and miscarriage for the indicated population is unknown.
Data
Human Data
Epidemiological studies reporting on pregnancies exposed to meclizine have not identified an association between the use of meclizine during pregnancy and an increased risk of major birth defects.
Animal Data
In a published study, oral administration of meclizine (25 to 250 mg/kg) to pregnant rats during the period of organogenesis resulted in a high incidence of fetal malformations. These effects occurred at doses as low as 25 mg/kg, which is approximately 2 times the maximum recommended human dose (100 mg) on a body surface area (mg/m 2 ) basis
Lactation
Risk Summary
There are no data on the presence of meclizine in human milk, the effects on the breastfed infant, or the effects on milk production. The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for meclizine and any potential adverse effects on the breastfed infant from meclizine or from the underlying maternal condition.
Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
Geriatric Use
In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
Hepatic Impairment
The effect of hepatic impairment on the pharmacokinetics of meclizine has not been evaluated. As meclizine undergoes metabolism, hepatic impairment may result in increased systemic exposure of meclizine. Treatment with meclizine hydrochloride tablets should be administered with caution in patients with hepatic impairment.
Renal Impairment
The effect of renal impairment on the pharmacokinetics of meclizine has not been evaluated. Because of a potential for drug/metabolite accumulation, meclizine hydrochloride tablets should be administered with caution in patients with renal impairment and in the elderly, as renal function generally declines with age.
Genetic CYP2D6 Polymorphism
The genetic polymorphism of CYP2D6 that results in poor-, intermediate-, extensive-, and ultrarapid metabolizer phenotypes could contribute to large inter-individual variability in meclizine exposure. Therefore, when meclizine hydrochloride tablets are administered to patients with CYP2D6 polymorphism, monitor for adverse reactions and clinical effect accordingly.
CONTRAINDICATIONS
Meclizine hydrochloride tablets are contraindicated in patients with a hypersensitivity to meclizine or any of the inactive ingredients [see ADVERSE REACTIONS (6) and DESCRIPTION (11) ].
ADVERSE REACTIONS
The following adverse reactions associated with the use of meclizine hydrochloride tablets were identified in clinical studies or postmarketing reports. Because some of these reactions were reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Anaphylactic reaction, drowsiness, dry mouth, headache, fatigue, and vomiting. On rare occasions blurred vision has been reported.
DRUG INTERACTIONS
- Coadministration of meclizine hydrochloride tablets with other CNS depressants, including alcohol, may result in increased CNS depression (7.1 ).
- CYP2D6 inhibitors: As meclizine is metabolized by CYP2D6, there is a potential for drug-drug interactions between meclizine hydrochloride and CYP2D6 inhibitors (7.2 ).
CNS Depressants
There may be increased CNS depression when meclizine hydrochloride tablets are administered concurrently with other CNS depressants, including alcohol [see WARNINGS AND PRECAUTIONS (5.1 )].
CYP2D6 Inhibitors
Based on in-vitro evaluation, meclizine is metabolized by CYP2D6. Therefore, there is a possibility for a drug interaction between meclizine and CYP2D6 inhibitors. Therefore, monitor for adverse reactions and clinical effect accordingly
DESCRIPTION
Meclizine hydrochloride, a histamine (H1) receptor antagonist, is a white or slightly yellowish, crystalline powder. It has the following structural formula:
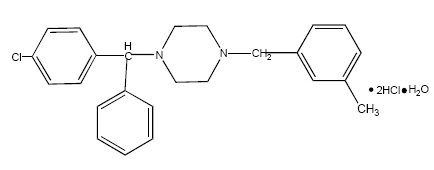
Chemically, meclizine hydrochloride is 1-(p-chloro-α-phenylbenzyl)-4-(m-methylbenzyl) piperazine dihydrochloride monohydrate.
Meclizine hydrochloride tablets, USP are available in two different strengths, 12.5 mg and 25 mg. Inactive ingredients: microcrystalline cellulose, lactose monohydrate, croscarmellose sodium and magnesium stearate. The 12.5 mg tablet also contains FD&C Blue #1 Aluminum Lake.
CLINICAL PHARMACOLOGY
Mechanism of Action
The precise mechanism by which meclizine exerts its therapeutic effect is unknown but is presumed to involve antagonism of the histamine H1 receptor.
Pharmacodynamics
There are no relevant pharmacodynamic data regarding meclizine.
Pharmacokinetics
The available pharmacokinetic information for meclizine following oral administration has been summarized from published literature.
Absorption
Meclizine is absorbed after oral administration with maximum plasma concentrations reaching at a median T max value of 3 hours post-dose (range: 1.5 to 6 hours) for the tablet dosage form.
Distribution
Drug distribution characteristics for meclizine in humans are unknown.
Elimination
Meclizine has a plasma elimination half-life of about 5-6 hours in humans.
Metabolism
In an in vitro metabolic study using human hepatic microsome and recombinant CYP enzyme, CYP2D6 was found to be the dominant enzyme for metabolism of meclizine.
NONCLINICAL TOXICOLOGY
Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
Animal studies to assess the carcinogenic potential of meclizine have not been conducted.
Mutagenesis
Genetic toxicology studies of meclizine have not been conducted.
Impairment of Fertility
Animal studies to assess the effects of meclizine on fertility and early embryonic development have not been conducted.
HOW SUPPLIED/STORAGE AND HANDLING
Meclizine Hydrochloride Tablets, USP are available as follows:
12.5 mg, blue, modified oval-shaped tablets, de-bossed with “Є12” on one side, and plain on the other side. They are supplied as follows:
NDC 42806-012-01 in bottles of 100
NDC 42806-012-10 in bottles of 1000
25 mg, white, modified oval-shaped tablets, de-bossed with “Є14” on one side, and plain on the other side. They are supplied as follows:
NDC 42806-014-01 in bottles of 100
NDC 42806-014-10 in bottles of 1000
16.2 Storage and Handling
Store at 20 o C to 25 o C (68 o F to 77 o F) [See USP Controlled Room Temperature].
Dispense in a tight, light-resistant container (USP).
Mechanism of Action
The precise mechanism by which meclizine exerts its therapeutic effect is unknown but is presumed to involve antagonism of the histamine H1 receptor.