Get your patient on Methocarbamol - Methocarbamol tablet, Coated (Methocarbamol)
Methocarbamol - Methocarbamol tablet, Coated prescribing information
INDICATIONS AND USAGE
Methocarbamol is indicated as an adjunct to rest, physical therapy, and other measures for the relief of discomfort associated with acute, painful musculoskeletal conditions. The mode of action of methocarbamol has not been clearly identified, but may be related to its sedative properties. Methocarbamol does not directly relax tense skeletal muscles in man.
DOSAGE AND ADMINISTRATION
Methocarbamol, 1000 mg – Adults:
Initial dosage: 1 ½ tablets 4 time daily
Maintenance dosage: 1 tablet 4 times daily
Six grams a day are recommended for the first 48 to 72 hours of treatment. (For severe conditions 8 grams a day may be administered). Thereafter, the dosage can usually be reduced to approximately 4 grams a day
CONTRAINDICATIONS
Methocarbamol is contraindicated in patients hypersensitive to methocarbamol or to any of the tablet components.
ADVERSE REACTIONS
Adverse reactions reported coincident with the administration of methocarbamol include:
Body as a whole: Anaphylactic reaction, angioneurotic edema, fever, headache
Cardiovascular system: Bradycardia, flushing, hypotension, syncope, thrombophlebitis
Digestive system: Dyspepsia, jaundice (including cholestatic jaundice), nausea and vomiting
Hemic and lymphatic system: Leukopenia
Immune system: Hypersensitivity reactions
Nervous system: Amnesia, confusion, diplopia, dizziness or lightheadedness, drowsiness, insomnia, mild muscular incoordination, nystagmus, sedation, seizures (including grand mal), vertigo
Skin and special senses: Blurred vision, conjunctivitis, nasal congestion, metallic taste, pruritus, rash, urticaria
To report SUSPECTED ADVERSE REACTIONS, contact Trifluent Pharma, LLC at 1-210-944-6920 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatchDrug Interactions
See WARNINGS and PRECAUTIONS for interaction with CNS drugs and alcohol.
Methocarbamol may inhibit the effect of pyridostigmine bromide. Therefore, methocarbamol should be used with caution in patients with myasthenia gravis receiving anticholinesterase agents.
DESCRIPTION
Methocarbamol Tablets USP, 1000 mg, a carbamate derivative of gualfenesin, is a central nervous system (CNS) depressant with sedative and musculoskeletal relaxant properties.
The chemical name of methocarbamol is a 3-(2 methoxyphenoxy)-1,2-propanediol 1-carbamate and has the empirical formula C 11 H 15 NO 5 . Its molecular weight is 241.24. The structural formula is shown below.
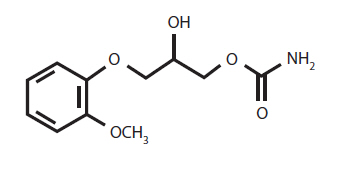
Methocarbamol is a white powder, sparingly soluble in water and chloroform, soluble in alcohol (only with heating) and propylene glycol, and insoluble in benzene and n-hexane.
Methocarbamol tablet, 1000 mg is available as an orange, film coated, oblong-shaped tablet containing 1000 mg of methocarbamol, USP for oral administration. The inactive ingredients present are microcrystalline cellulose, croscarmellose sodium, FD&C Yellow 6 aluminum lake, hydroxypropyl cellulose, hypromellose, magnesium stearate, lactose monohydrate, polyethylene glycol, triacetin, titanium dioxide.
Clinical Pharmacology
The mechanism of action of methocarbamol in humans has not been established, but may be due to general central nervous system (CNS) depression. It has no direct action on the contractile mechanism of striated muscle, the motor end plate or the nerve fiber.
Pharmacokinetics
In healthy volunteers, the plasma clearance of methocarbamol ranges between 0.20 and 0.80 L/h/kg, the mean plasma elimination half-life ranges between 1 and 2 hours, and the plasma protein binding ranges between 46% and 50%.
Methocarbamol is metabolized via dealkylation and hydroxylation. Conjugation of methocarbamol also is likely. Essentially all methocarbamol metabolites are eliminated in the urine. Small amounts of unchanged methocarbamol also are excreted in the urine.
Special populations
Elderly
The mean (± SD) elimination half-life of methocarbamol in elderly healthy volunteers (mean (± SD) age, 69 (± 4) years) was slightly prolonged compared to a younger (mean (± SD) age, 53.3 (± 8.8) years), healthy population (1.5 (± 0.4) hours versus 1.1 (±0.27) hours, respectively). The fraction of bound methocarbamol was slightly decreased in the elderly versus younger volunteers (41 to 43% versus 46 to 50%, respectively).
Preclinical studies (in male rats) show that alendronate transiently distributes to soft tissues following 1 mg/kg IV administration but is then rapidly redistributed to bone or excreted in the urine. The mean steady-state volume of distribution, exclusive of bone, is at least 28 L in humans. Concentrations of drug in plasma following therapeutic oral doses are too low (less than 5 ng/mL) for analytical detection. Protein binding in human plasma is approximately 78%.
Renally impaired
The clearance of methocarbamol in 8 renally-impaired patients on maintenance hemodialysis was reduced about 40% compared to 17 normal subjects, although the mean (± SD) elimination half-life in these two groups was similar: 1.2 (± 0.6) versus 1.1 (± 0.3) hours, respectively.
Hepatically impaired
In 8 patients with cirrhosis secondary to alcohol abuse, the mean total clearance of methocarbamol was reduced approximately 70% compared to that obtained in 8 age and weight-matched normal subjects. The mean (± SD) elimination half-life in the cirrhotic patients and the normal subjects was 3.38 (± 1.62) hours and 1.11 (± 0.27) hours, respectively. The percent of methocarbamol bound to plasma proteins was decreased to approximately 40 to 45% compared to 46 to 50% in the normal subjects.
HOW SUPPLIED
Methocarbamol Tablets USP, 1000 mg - Orange, film coated, oblong-shaped tablets, one side debossed 'AP349', the other side bisected. They are supplied as follows:
Bottles of 100, NDC 73352-510-10
Store at controlled room temperature, between 20°C and 25°C (68°F and 77°F). Dispense in tight container.