Get your patient on Methylphenidate Hcl - Methylphenidate Hcl solution (Methylphenidate Hcl)
Methylphenidate Hcl - Methylphenidate Hcl solution prescribing information
INDICATIONS AND USAGE
Attention Deficit Disorders, Narcolepsy
Attention Deficit Disorders (previously known as Minimal Brain Dysfunction in Children). Other terms being used to describe the behavioral syndrome below include: Hyperkinetic Child Syndrome, Minimal Brain Damage, Minimal Cerebral Dysfunction, Minor Cerebral Dysfunction.
Methylphenidate hcl oral solution is indicated as an integral part of a total treatment program which typically includes other remedial measures (psychological, educational, social) for a stabilizing effect in children with a behavioral syndrome characterized by the following group of developmentally inappropriate symptoms: moderate-to-severe distractibility, short attention span, hyperactivity, emotional lability, and impulsivity. The diagnosis of this syndrome should not be made with finality when these symptoms are only of comparatively recent origin. Nonlocalizing (soft) neurological signs, learning disability, and abnormal EEG may or may not be present, and a diagnosis of central nervous system dysfunction may or may not be warranted.
Special Diagnostic Considerations
Specific etiology of this syndrome is unknown, and there is no single diagnostic test. Adequate diagnosis requires the use not only of medical but of special psychological, educational, and social resources.
Characteristics commonly reported include: chronic history of short attention span, distractibility, emotional lability, impulsivity, and moderate-to -severe hyperactivity; minor neurological signs and abnormal EEG. Learning may or may not be impaired. The diagnosis must be based upon a complete history and evaluation of the child and not solely on the presence of one or more of these characteristics.
Drug treatment is not indicated for all children with this syndrome. Stimulants are not intended for use in the child who exhibits symptoms secondary to environmental factors and/or primary psychiatric disorders, including psychosis. Appropriate educational placement is essential and psychosocial intervention is generally necessary. When remedial measures alone are insufficient, the decision to prescribe stimulant medication will depend upon the physician's assessment of the chronicity and severity of the child's symptoms.
DOSAGE AND ADMINISTRATION
Dosage should be individualized according to the needs and responses of the patient.
Adults
Administer in divided doses 2 or 3 times daily, preferably 30 to 45 minutes before meals. Average dosage is 20 to 30 mg daily. Some patients may require 40 to 60 mg daily. In others, 10 to 15 mg daily will be adequate. Patients who are unable to sleep if medication is taken late in the day should take the last dose before 6 p.m.
Children (6 years and over)
Methylphenidate hcl should be initiated in small doses, with gradual weekly increments. Daily dosage above 60 mg is not recommended.
If improvement is not observed after appropriate dosage adjustment over a one-month period, the drug should be discontinued.
Start with 5 mg twice daily (before breakfast and lunch) with gradual increments of 5 to 10 mg weekly. If paradoxical aggravation of symptoms or other adverse effects occur, reduce dosage, or, if necessary, discontinue the drug.
Methylphenidate hcl should be periodically discontinued to assess the child's condition. Improvement may be sustained when the drug is either temporarily or permanently discontinued.
Drug treatment should not and need not be indefinite and usually may be discontinued after puberty.
CONTRAINDICATIONS
Marked anxiety, tension, and agitation are contraindications to methylphenidate hcl, since the drug may aggravate these symptoms. Methylphenidate hcl is contraindicated also in patients known to be hypersensitive to the drug, in patients with glaucoma, and in patients with motor tics or with a family history or diagnosis of Tourette's syndrome.
Methylphenidate hcl is contraindicated during treatment with monoamine oxidase inhibitors, and also within a minimum of 14 days following discontinuation of a monoamine oxidase inhibitor (hypertensive crises may result).
ADVERSE REACTIONS
Nervousness and insomnia are the most common adverse reactions but are usually controlled by reducing dosage and omitting the drug in the afternoon or evening. Other reactions include hypersensitivity (including skin rash, urticaria, fever, arthralgia, exfoliative dermatitis, erythema multiforme with histopathological findings of necrotizing vasculitis, and thrombocytopenic purpura); anorexia; nausea; dizziness; palpitations; headache; dyskinesia; drowsiness; blood pressure and pulse changes, both up and down; tachycardia; angina; cardiac arrhythmia; abdominal pain; weight loss during prolonged therapy; libido changes; and rhabdomyolysis. There have been rare reports of Tourette’s syndrome. Toxic psychosis has been reported. Although a definite causal relationship has not been established, the following have been reported in patients taking this drug: instances of abnormal liver function, ranging from transaminase elevation to severe hepatic injury; isolated cases of cerebral arteritis and/or occlusion; leukopenia and/or anemia; transient depressed mood; a few instances of scalp hair loss; serotonin syndrome in combination with serotonergic drugs. Very rare reports of neuroleptic malignant syndrome (NMS) have been received, and, in most of these, patients were concurrently receiving therapies associated with NMS. In a single report, a ten year old boy who had been taking methylphenidate for approximately 18 months experienced an NMS-like event within 45 minutes of ingesting his first dose of venlafaxine. It is uncertain whether this case represented a drug-drug interaction, a response to either drug alone, or some other cause.
In children, loss of appetite, abdominal pain, weight loss during prolonged therapy, insomnia, and tachycardia may occur more frequently; however, any of the other adverse reactions listed above may also occur.
Drug Interactions
Methylphenidate hcl may decrease the hypotensive effect of guanethidine. Use cautiously with pressor agents.
Human pharmacologic studies have shown that methylphenidate hcl may inhibit the metabolism of coumarin anticoagulants, anticonvulsants (phenobarbital, diphenylhydantoin, primidone), phenylbutazone, and tricyclic drugs (imipramine, clomipramine, desipramine). Downward dosage adjustments of these drugs may be required when given concomitantly with methylphenidate hcl.
DESCRIPTION
Methylphenidate hcl oral solution is a mild central nervous system (CNS) stimulant available as 5 mg /5 mL and 10 mg /5 mL oral solutions for oral administration. Methylphenidate hydrochloride is methyl α-phenyl-2-piperidineacetate hydrochloride, and its structural formula is
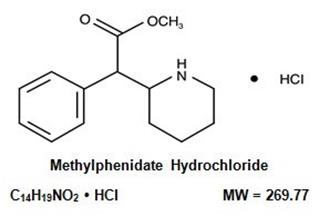
Methylphenidate hydrochloride USP is a white, odorless, fine crystalline powder. Its solutions are acid to litmus. It is freely soluble in water and in methanol, soluble in alcohol, and slightly soluble in chloroform and in acetone.
Each mL of methylphenidate hcl oral solution 5 mg /5 mL contains 1 mg of methylphenidate hydrochloride USP.
Each mL of methylphenidate hcl oral solution 10 mg /5 mL contains 2 mg of methylphenidate hydrochloride USP.
In addition, methylphenidate hcl oral solution also contains the following inactive ingredients: phosphoric acid, glycerin, N&A grape flavor, PEG 1450, and purified water.
CLINICAL PHARMACOLOGY
Methylphenidate is a racemic mixture comprised of the d- and l-threo enantiomers. The d-threo enantiomer is more pharmacologically active than the l-threo enantiomer. Methylphenidate hcl is a central nervous system (CNS) stimulant.
The mode of therapeutic action in humans is not completely understood, but methylphenidate presumably activates the brain stem arousal system and cortex to produce its stimulant effect. Methylphenidate is thought to block the reuptake of norepinephrine and dopamine into the presynaptic neuron and increase the release of these monoamines into the extraneuronal space.
There is neither specific evidence which clearly establishes the mechanism whereby methylphenidate hcl produces its mental and behavioral effects in children, nor conclusive evidence regarding ho w these effects relate to the condition of the central nervous system.
Pharmacokinetics
Absorption
Methylphenidate hcl oral solution is readily absorbed. Following oral administration of methylphenidate hcl oral solution, peak plasma methylphenidate concentrations are achieved at 1 to 2 hours. Methylphenidate hcl oral solution has been shown to be bioequivalent to Ritalin® tablet. The mean C max following a 20 mg dose is approximately 9 ng/mL.
Food Effect
In a study in adult volunteers to investigate the effects of a high-fat meal on the bioavailability of methylphenidate hcl oral solution at a dose of 20 mg, the presence of food delayed the peak by approximately 1 hour (1.7 hours, fasted and 2.7 hours, fed). Overall, a high-fat meal increased the C max of methylphenidate hcl oral solution by about 13% and the AUC by about 25%, on average. Through a cross-study comparison, the magnitude of increase in C max and AUC is found to be comparable between the methylphenidate hcl oral solution and Ritalin, the immediate release tablet.
Metabolism and Excretion
In humans, methylphenidate is metabolized primarily via deesterification to alpha-phenylpiperidine acetic acid (PPA, ritalinic acid). The metabolite has little or no pharmacologic activity.
After oral dosing of radiolabeled methylphenidate in humans, about 90% of the radioactivity was recovered in urine. The main urinary metabolite was PPA, accounting for approximately 80% of the dose.
The pharmacokinetics of the methylphenidate hcl oral solution have been studied in healthy adult volunteers. The mean terminal half-life (t ½ ) of methylphenidate following administration of 20 mg methylphenidate hcl oral solution (t ½ = 2.7 hours) is comparable to the mean terminal t ½ following administration of Ritalin (methylphenidate hydrochloride immediate-release tablets) (t ½ = 2.8h) in healthy adult volunteers.
Special Populations
Gender – The effect of gender on the pharmacokinetics of methylphenidate after methylphenidate hcl oral solution administration has not been studied.
Race – The influence of race on the pharmacokinetics of methylphenidate after methylphenidate hcl oral solution administration has not been studied.
Age – The pharmacokinetics of methylphenidate after methylphenidate hcl oral solution administration have not been studied in pediatrics.
Renal Insufficiency
There is no experience with the use of methylphenidate hcl oral solution in patients with renal insufficiency. After oral administration of radiolabeled methylphenidate in humans, methylphenidate was extensively metabolized and approximately 80% of the radioactivity was excreted in the urine in the form of ritalinic acid. Since renal clearance is not an important route of methylphenidate clearance, renal insufficiency is expected to have little effect on the pharmacokinetics of methylphenidate hcl oral solution.
Hepatic Insufficiency
There is no experience with the use of methylphenidate hcl oral solution in patients with hepatic insufficiency.
HOW SUPPLIED
Methylphenidate HCl Oral Solution 5 mg per 5 mL is available as a colorless, grape flavored liquid.
Bottles of 500 mL . . . . . . . . NDC 39328-054-50
Methylphenidate HCl Oral Solution 10 mg per 5 mL is available as a colorless, grape flavored liquid.
Bottles of 500 mL . . . . . . . . NDC 39328-154-50
Dispense in tight container with child-resistant closure and a medication guide.
Storage: Store at 20° to 25°C (68 ° to 77°F) [see USP Controlled Room Temperature].
Ritalin is a registered trademark of Novartis Corporation.
Manufactured for: Patrin Pharma, Inc. Skokie, IL 60077 USA
Rev 07/2017