Get your patient on Metolazone - Metolazone tablet (Metolazone)
Metolazone - Metolazone tablet prescribing information
DO NOT INTERCHANGE
DO NOT INTERCHANGE METOLAZONE TABLETS, USP, ZAROXOLYN ® TABLETS, AND OTHER FORMULATIONS OF METOLAZONE THAT SHARE THEIR SLOW AND INCOMPLETE BIOAVAILABILITY AND ARE NOT THERAPEUTICALLY EQUIVALENT AT THE SAME DOSES TO MYKROX ® TABLETS, A MORE RAPIDLY AVAILABLE AND COMPLETELY BIOAVAILABLE METOLAZONE PRODUCT. FORMULATIONS BIOEQUIVALENT TO ZAROXOLYN AND FORMULATIONS BIOEQUIVALENT TO MYKROX SHOULD NOT BE INTERCHANGED FOR ONE ANOTHER.
INDICATIONS AND USAGE
Metolazone tablets are indicated for the treatment of salt and water retention including:
- edema accompanying congestive heart failure;
- edema accompanying renal diseases, including the nephrotic syndrome and states of diminished renal function.
Metolazone tablets are also indicated for the treatment of hypertension, alone or in combination with other antihypertensive drugs of a different class. MYKROX Tablets, a more rapidly available form of metolazone, are intended for the treatment of new patients with mild to moderate hypertension. A dose titration is necessary if MYKROX Tablets are to be substituted for metolazone tablets in the treatment of hypertension. See package circular for MYKROX Tablets.
Usage In Pregnancy
The routine use of diuretics in an otherwise healthy woman is inappropriate and exposes mother and fetus to unnecessary hazard. Diuretics do not prevent development of toxemia of pregnancy, and there is no evidence that they are useful in the treatment of developed toxemia.
Edema during pregnancy may arise from pathologic causes or from the physiologic and mechanical consequences of pregnancy. Metolazone tablets are indicated in pregnancy when edema is due to pathologic causes, just as it is in the absence of pregnancy (see PRECAUTIONS ). Dependent edema in pregnancy resulting from restriction of venous return by the expanded uterus is properly treated through elevation of the lower extremities and use of support hose; use of diuretics to lower intravascular volume in this case is illogical and unnecessary. There is hypervolemia during normal pregnancy which is harmful to neither the fetus nor the mother (in the absence of cardiovascular disease), but which is associated with edema, including generalized edema, in the majority of pregnant women. If this edema produces discomfort, increased recumbency will often provide relief. In rare instances, this edema may cause extreme discomfort which is not relieved by rest. In these cases, a short course of diuretics may be appropriate.
DOSAGE AND ADMINISTRATION
Effective dosage of metolazone tablets should be individualized according to indication and patient response. A single daily dose is recommended. Therapy with metolazone tablets should be titrated to gain an initial therapeutic response and to determine the minimal dose possible to maintain the desired therapeutic response.
Usual Single Daily Dosage Schedules
Suitable initial dosages will usually fall in the ranges given.
Edema of cardiac failure:
Metolazone tablets 5 to 20 mg once daily.
Edema of renal disease:
Metolazone tablets 5 to 20 mg once daily.
Mild to moderate essential hypertension:
Metolazone tablets 2.5 to 5 mg once daily.
New patients – MYKROX Tablets (metolazone tablets, USP) (see MYKROX package circular). If considered desirable to switch patients currently on metolazone tablets, USP, to MYKROX, the dose should be determined by titration starting at one tablet (0.5 mg) once daily and increasing to two tablets (1 mg) once daily if needed.
Treatment Of Edematous States
The time interval required for the initial dosage to produce an effect may vary. Diuresis and saluresis usually begin within one hour and persist for 24 hours or longer. When a desired therapeutic effect has been obtained, it may be advisable to reduce the dose if possible. The daily dose depends on the severity of the patient's condition, sodium intake, and responsiveness. A decision to change the daily dose should be based on the results of thorough clinical and laboratory evaluations. If antihypertensive drugs or diuretics are given concurrently with metolazone tablets more careful dosage adjustment may be necessary. For patients who tend to experience paroxysmal nocturnal dyspnea, it may be advisable to employ a larger dose to ensure prolongation of diuresis and saluresis for a full 24-hour period.
Treatment Of Hypertension
The time interval required for the initial dosage regimen to show effect may vary from three or four days to three to six weeks in the treatment of elevated blood pressure. Doses should be adjusted at appropriate intervals to achieve maximum therapeutic effect.
CONTRAINDICATIONS
Anuria, hepatic coma or precoma, known allergy or hypersensitivity to metolazone.
ADVERSE REACTIONS
Metolazone tablets are usually well tolerated, and most reported adverse reactions have been mild and transient. Many of metolazone tablets related adverse reactions represent extensions of its expected pharmacologic activity and can be attributed to either its antihypertensive action or its renal/metabolic actions.
The following adverse reactions have been reported. Several are single or comparably rare occurrences. Adverse reactions are listed in decreasing order of severity within body systems.
Cardiovascular
Chest pain/discomfort, orthostatic hypotension, excessive volume depletion, hemoconcentration, venous thrombosis, palpitations.
Central And Peripheral Nervous System
Syncope, neuropathy, vertigo, paresthesias, psychotic depression, impotence, dizziness/lightheadedness, drowsiness, fatigue, weakness, restlessness (sometimes resulting in insomnia), headache.
Dermatologic/Hypersensitivity
Toxic epidermal necrolysis (TEN), Stevens-Johnson Syndrome, necrotizing angiitis (cutaneous vasculitis), skin necrosis, purpura, petechiae, dermatitis (photosensitivity), urticaria, pruritus, skin rashes.
Gastrointestinal
Hepatitis, intrahepatic cholestatic jaundice, pancreatitis, vomiting, nausea, epigastric distress, diarrhea, constipation, anorexia, abdominal bloating, abdominal pain.
Hematologic
Aplastic/hypoplastic anemia, agranulocytosis, leukopenia, thrombocytopenia.
Metabolic
Hypokalemia, hyponatremia, hyperuricemia, hypochloremia, hypochloremic alkalosis, hyperglycemia, glycosuria, increase in serum urea nitrogen (BUN) or creatinine, hypophosphatemia, hypomagnesemia, hypercalcemia.
Musculoskeletal
Joint pain, acute gouty attacks, muscle cramps or spasm.
Other
Transient blurred vision, chills, dry mouth.
In addition, adverse reactions reported with similar antihypertensive-diuretics, but which have not been reported to date for metolazone tablets include: bitter taste, sialadenitis, xanthopsia, respiratory distress (including pneumonitis), and anaphylactic reactions. These reactions should be considered as possible occurrences with clinical usage of metolazone tablets.
Whenever adverse reactions are moderate or severe, metolazone tablets dosage should be reduced or therapy withdrawn.
To report SUSPECTED ADVERSE REACTIONS, contact Micro Labs USA, Inc. at 1-855-839-8195 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
Diuretics
Furosemide and probably other loop diuretics given concomitantly with metolazone can cause unusually large or prolonged losses of fluid and electrolytes (see WARNINGS ).
Other Antihypertensives
When metolazone tablets are used with other antihypertensive drugs, care must be taken, especially during initial therapy. Dosage adjustments of other antihypertensives may be necessary.
Alcohol, Barbiturates, And Narcotics
The hypotensive effects of these drugs may be potentiated by the volume contraction that may be associated with metolazone therapy.
Digitalis Glycosides
Diuretic-induced hypokalemia can increase the sensitivity of the myocardium to digitalis. Serious arrhythmias can result.
Corticosteroids Or ACTH
May increase the risk of hypokalemia and increase salt and water retention.
Lithium
Serum lithium levels may increase (see WARNINGS ).
Curariform Drugs
Diuretic-induced hypokalemia may enhance neuromuscular blocking effects of curariform drugs (such as tubocurarine) – the most serious effect would be respiratory depression which could proceed to apnea. Accordingly, it may be advisable to discontinue metolazone tablets three days before elective surgery.
Salicylates And Other Non-Steroidal Anti-Inflammatory Drugs
May decrease the antihypertensive effects of metolazone tablets.
Sympathomimetics
Metolazone may decrease arterial responsiveness to norepinephrine, but this diminution is not sufficient to preclude effectiveness of the pressor agent for therapeutic use.
Insulin And Oral Antidiabetic Agents
See Glucose Tolerance under PRECAUTIONS, General .
Methenamine
Efficacy may be decreased due to urinary alkalizing effect of metolazone.
Anticoagulants
Metolazone, as well as other thiazide-like diuretics, may affect the hypoprothrombinemic response to anticoagulants; dosage adjustments may be necessary.
DESCRIPTION
Metolazone tablets, USP, for oral administration contain 2.5 mg, 5 mg, or 10 mg of metolazone, USP, a diuretic/saluretic/antihypertensive drug of the quinazoline class.
Metolazone has the molecular formula C 16 H 16 ClN 3 O 3 S, the chemical name 7-chloro-1, 2, 3, 4-tetrahydro-2- methyl-3-(2-methylphenyl)-4-oxo-6-quinazolinesulfonamide, and a molecular weight of 365.83. The structural formula is:
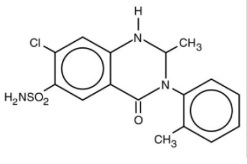
Metolazone is only sparingly soluble in water, but more soluble in plasma, blood, alkali, and organic solvents.
Inactive Ingredients: colloidal silicon dioxide, magnesium stearate, microcrystalline cellulose and dye: 2.5 mg - D&C Red No. 30 and FD&C Blue No. 1; 5 mg - FD&C Blue No. 2; 10 mg - D&C Yellow No. 10 and FD&C Yellow No. 6.
CLINICAL PHARMACOLOGY
Metolazone is a quinazoline diuretic, with properties generally similar to the thiazide diuretics. The actions of metolazone result from interference with the renal tubular mechanism of electrolyte reabsorption. Metolazone acts primarily to inhibit sodium reabsorption at the cortical diluting site and to a lesser extent in the proximal convoluted tubule. Sodium and chloride ions are excreted in approximately equivalent amounts. The increased delivery of sodium to the distal tubular exchange site results in increased potassium excretion. Metolazone does not inhibit carbonic anhydrase. A proximal action of metolazone has been shown in humans by increased excretion of phosphate and magnesium ions and by a markedly increased fractional excretion of sodium in patients with severely compromised glomerular filtration. This action has been demonstrated in animals by micropuncture studies.
When metolazone tablets are given, diuresis and saluresis usually begin within one hour and may persist for 24 hours or more. For most patients, the duration of effect can be varied by adjusting the daily dose. High doses may prolong the effect. A single daily dose is recommended. When a desired therapeutic effect has been obtained, it may be possible to reduce dosage to a lower maintenance level.
The diuretic potency of metolazone tablets at maximum therapeutic dosage is approximately equal to thiazide diuretics. However, unlike thiazides, metolazone tablets may produce diuresis in patients with glomerular filtration rates below 20 mL/min.
Metolazone tablets and furosemide administered concurrently have produced marked diuresis in some patients where edema or ascites was refractory to treatment with maximum recommended doses of these or other diuretics administered alone. The mechanism of this interaction is unknown (see WARNINGS and PRECAUTIONS , Drug Interactions ).
Maximum blood levels of metolazone are found approximately eight hours after dosing. A small fraction of metolazone is metabolized. Most of the drug is excreted in the unconverted form in the urine.
HOW SUPPLIED
Metolazone tablets, USP, are biconvex, round tablets, and are available in three strengths:
2.5 mg – pink colored mottled tablets debossed with “M1” on one side and plain on other side, free from physical defects. It is available as follows:
Bottles of 100 with child-resistant closure: NDC 42571-416-01
Bottles of 1000: NDC 42571-416-10
5 mg – blue colored mottled tablets debossed with “M2” on one side and plain on other side, free from physical defects. It is available as follows:
Bottles of 100 with child-resistant closure: NDC 42571-417-01
Bottles of 1000: NDC 42571-417-10
10 mg – yellow colored tablets debossed with “M3” on one side and plain on other side, free from physical defects. It is available as follows:
Bottles of 100 with child-resistant closure: NDC 42571-418-01
Bottles of 1000: NDC 42571-418-10
Storage
Store at 25°C (77°F); excursions permitted to 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature].
Protect from light. Keep out of the reach of children. Dispense in a tight, light-resistant container as defined in the USP using a child-resistant closure.
For more information about Metolazone Tablets, USP call 1-855-839-8195.
All brand names are the trademarks of their respective owners. Manufactured for: Micro Labs USA, Inc. Somerset, NJ 08873 Rev. 04/2023-01