Get your patient on Metronidazole
Metronidazole prescribing information
WARNING
Metronidazole has been shown to be carcinogenic in mice and rats (see PRECAUTIONS ). Unnecessary use of the drug should be avoided. Its use should be reserved for the conditions described in the INDICATIONS AND USAGE section below.
INDICATIONS AND USAGE
Treatment of Anaerobic Bacterial Infections
Metronidazole Injection is indicated in the treatment of serious infections caused by susceptible anaerobic bacteria. Indicated surgical procedures should be performed in conjunction with Metronidazole Injection therapy. In a mixed aerobic and anaerobic infection, antibacterial drugs appropriate for the treatment of the aerobic infection should be used in addition to Metronidazole Injection.
Metronidazole Injection is effective in Bacteroides fragilis infections resistant to clindamycin, chloramphenicol and penicillin.
- Intra-Abdominal Infections, including peritonitis, intra-abdominal abscess and liver abscess, caused by Bacteroides species including the B. fragilis group ( B. fragilis , B. distasonis , B. ovatus , B. thetaiotaomicron , B. vulgatus ), Clostridium species, Eubacterium species, Peptococcus species and Peptostreptococcus species in adults and pediatric patients less than 4 months of age.
- Skin and Skin Structure Infections caused by Bacteroides species including the B. fragilis group, Clostridium species, Peptococcus species, Peptostreptococcus species and Fusobacterium species in adults.
- Gynecologic Infections , including endometritis, endomyometritis, tubo-ovarian abscess and postsurgical vaginal cuff infection, caused by Bacteroides species including the B. fragilis group, Clostridium species, Peptococcus species, Peptostreptococcus species and Fusobacterium species in adults.
- Bacterial Septicemia caused by Bacteroides species including the B. fragilis group and Clostridium species in adults.
- Bone and Joint Infections , as adjunctive therapy, caused by Bacteroides species including the B. fragilis group in adults.
- Central Nervous System (CNS) Infections , including meningitis and brain abscess, caused by Bacteroides species including the B. fragilis group in adults.
- Lower Respiratory Tract Infections , including pneumonia, empyema and lung abscess, caused by Bacteroides species including the B. fragilis group in adults.
- Endocarditis caused by Bacteroides species including the B. fragilis group in adults.
Prophylaxis Indication
The prophylactic administration of Metronidazole Injection preoperatively, intraoperatively and postoperatively may reduce the incidence of postoperative infection in adult patients undergoing elective colorectal surgery which is classified as contaminated or potentially contaminated. Prophylactic use of Metronidazole Injection should be discontinued within 12 hours after surgery. If there are signs of infection, specimens for cultures should be obtained for the identification of the causative organism(s) so that appropriate therapy may be given (see DOSAGE AND ADMINISTRATION ).
Usage
To reduce the development of drug-resistant bacteria and maintain the effectiveness of Metronidazole Injection and other antibacterial drugs, Metronidazole Injection should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
DOSAGE AND ADMINISTRATION
Dosage, rate of administration, and duration of treatment are to be individualized and depend upon the indication for use, the patient’s age, weight, clinical condition and concomitant treatment, and on the patient’s clinical and laboratory response to the treatment.
Recommended Dosage for the Treatment of Anaerobic Bacterial Infections
The recommended dosage schedule for adults for the treatment of anaerobic infections is presented in the Table 1 :
Loading Dose | 15 mg/kg infused intravenously over one hour (approximately 1 gram for a 70-kg adult). |
Maintenance Dosage | 7.5 mg/kg infused intravenously over one hour every six hours (approximately 500 mg for a 70-kg adult). The first maintenance dose should be instituted six hours following the initiation of the loading dose. |
Parenteral therapy may be changed to oral metronidazole therapy when conditions warrant, based upon the severity of the disease and the response of the patient to treatment with Metronidazole Injection. The usual adult oral dosage is 7.5 mg/kg every six hours (approximately 500 mg for a 70-kg adult).
A maximum of 4 grams should not be exceeded during a 24-hour period.
The usual duration of therapy is 7 to 10 days; however, infections of the bone and joint, lower respiratory tract and endocardium may require longer treatment.
Recommended Dosage for the Treatment of Intra-Abdominal Infections in Pediatric Patients Less than 4 Months of Age:
The recommended dosage schedule for pediatric patients less than 4 months of age for the treatment of intra-abdominal infections is described in Table 2 below. These dosage schedules achieve drug exposures in pediatric patients similar to adults treated with Metronidazole Injection for this indication.
Post-menstrual age (Completed weeks) | Loading Dose (mg/kg) | Maintenance Dose The first maintenance dose is given 24 hours after the start of the loading dose. (mg/kg) | Dosing interval (hours) |
23 to <34 | 15 | 7.5 | 12 |
34 to 40 | 15 | 7.5 | 8 |
>40 to 48 | 15 | 7.5 | 6 |
Recommended Dosage in Patients with Severe Hepatic Impairment For patients with severe hepatic impairment (Child-Pugh C), the metronidazole dose should be reduced by 50% (see CLINICAL PHARMACOLOGY and PRECAUTIONS ).
Recommended Dosage in Patients Undergoing Hemodialysis
Hemodialysis removes significant amounts of metronidazole and its metabolites from systemic circulation. The clearance of metronidazole will depend on the type of dialysis membrane used, the duration of the dialysis session, and other factors. If the administration of metronidazole cannot be separated from a hemodialysis session, supplementation of metronidazole dosage following a hemodialysis session should be considered, depending on the patient’s clinical situation (see CLINICAL PHARMACOLOGY ).
Dosage Considerations in Geriatric Patients
In elderly patients the pharmacokinetics of metronidazole may be altered and, therefore, monitoring of serum levels may be necessary to adjust the metronidazole dosage accordingly.
Recommended Prophylaxis Dosage
For surgical prophylactic use, to prevent postoperative infection in contaminated or potentially contaminated colorectal surgery, the recommended dosage schedule for adults is:
- 15 mg/kg infused over 30 to 60 minutes and completed approximately one hour before surgery; followed by
- 7.5 mg/kg infused over 30 to 60 minutes at 6 and 12 hours after the initial dose.
It is important that (1) administration of the initial preoperative dose be completed approximately one hour before surgery so that adequate drug levels are present in the serum and tissues at the time of initial incision, and (2) Metronidazole Injection be administered, if necessary, at 6-hour intervals to maintain effective drug levels. Prophylactic use of Metronidazole Injection should be limited to the day of surgery only, following the above guidelines.
Important Administration and Preparation Instructions
Caution: Metronidazole Injection is to be administered by slow intravenous drip infusion only, either as a continuous or intermittent infusion. Additives should not be introduced into Metronidazole Injection, unless compatibility is known. If used with a primary intravenous fluid system, the primary solution should be discontinued during metronidazole infusion. DO NOT USE EQUIPMENT CONTAINING ALUMINUM ( e.g., NEEDLES, CANNULAE) THAT WOULD COME IN CONTACT WITH THE DRUG SOLUTION AS PRECIPITATES MAY FORM.
Metronidazole Injection is incompatible with (includes but is not limited to): Aztreonam, Cefamandole nafate, Cefoxitin, Penicillin G.
In the absence of compatibility studies, this medicinal product must not be mixed with other medicinal products (see DIRECTIONS FOR USE OF VIAFLEX PLUS PLASTIC CONTAINER ).
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. Do not administer unless the solution is clear and the seal is intact.
CONTRAINDICATIONS
- Hypersensitivity Metronidazole Injection is contraindicated in patients with a prior history of hypersensitivity to metronidazole or other nitroimidazole derivatives.
- Psychotic Reaction with Disulfiram Use of oral metronidazole is associated with psychotic reactions in alcoholic patients who were using disulfiram concurrently. Do not administer metronidazole to patients who have taken disulfiram within the last two weeks (see PRECAUTIONS-Drug Interactions ).
- Interaction with Alcohol Use of oral metronidazole is associated with a disulfiram-like reaction to alcohol, including abdominal cramps, nausea, vomiting, headaches, and flushing. Discontinue consumption of alcohol or products containing propylene glycol during and for at least three days after therapy with metronidazole (see PRECAUTIONS-Drug Interactions ).
- Cockayne Syndrome Metronidazole Injection is contraindicated in patients with Cockayne syndrome. Severe irreversible hepatotoxicity/acute liver failure with fatal outcomes have been reported after initiation of metronidazole in patients with Cockayne syndrome (see ADVERSE REACTIONS ).
ADVERSE REACTIONS
The following reactions have been reported during treatment with metronidazole formulations:
INFECTIONS AND INFESTATIONS: Vaginal candidiasis
BLOOD AND LYMPHATIC SYSTEM DISORDERS: Agranulocytosis, Leukopenia, Neutropenia, Thrombocytopenia, Eosinophilia
IMMUNE SYSTEM DISORDERS: Anaphylactic reaction, Hypersensitivity
METABOLISM AND NUTRITION DISORDERS: Decreased appetite
PSYCHIATRIC DISORDERS: Confusional state, Depression, Insomnia, Decreased libido
NERVOUS SYSTEM DISORDERS: Encephalopathy, Seizure, Neuropathy peripheral, Ataxia, Dizziness, Hypoesthesia, Paresthesia, Dysgeusia, Headache, Nystagmus, Aseptic meningitis, Somnolence, Dysarthria, Numbness, Syncope
EYE DISORDERS: Optic neuropathy, Saccadic eye movement
EAR AND LABYRINTH DISORDERS: Vertigo, tinnitus, hearing impairment, hearing loss
CARDIAC DISORDERS: QT prolongation has been reported, particularly when metronidazole was administered with drugs with the potential for prolonging the QT interval. Flattening of the T-wave may be seen in electrocardiographic tracings, Tachycardia, Palpitation
RESPIRATORY, THORACIC AND MEDIASTINAL DISORDERS: Dyspnea
GASTROINTESTINAL DISORDERS: Pancreatitis, Abdominal pain, Diarrhea, Nausea, Vomiting, Asthenia, Proctitis
HEPATOBILIARY DISORDERS: Hepatotoxicity/Liver Failure in patients with Cockayne syndrome (see CONTRAINDICATIONS ), Jaundice
SKIN AND SUBCUTANEOUS DISORDERS: Toxic epidermal necrolysis, Swelling face, Pruritus, Urticaria, Hyperhidrosis, Erythema, Rash; Stevens-Johnson syndrome, Fixed drug eruption, Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS), acute generalized exanthematous pustulosis (AGEP) (see WARNINGS )
MUSCULOSKELETAL AND CONNECTIVE TISSUE DISORDERS: Muscle spasms, Arthralgia, Myalgia
RENAL AND URINARY DISORDERS: Chromaturia, Dysuria
Hepatic: Cases of severe irreversible hepatotoxicity/acute liver failure, including cases with fatal outcomes with very rapid onset after initiation of systemic use of metronidazole, have been reported in patients with Cockayne Syndrome (latency from drug start to signs of liver failure as short as 2 days) (see CONTRAINDICATIONS )
REPRODUCTIVE: Dyspareunia
GENERAL DISORDERS AND ADMINISTRATION SITE CONDITIONS: Injection site reaction, Malaise, Face edema, Edema peripheral, Chest pain, Chills
INVESTIGATIONS: Hepatic enzyme increased.
Patients with Crohn's disease are known to have an increased incidence of gastrointestinal and certain extraintestinal cancers. There have been some reports in the medical literature of breast and colon cancer in Crohn's disease patients who have been treated with metronidazole at high doses for extended periods of time. A cause and effect relationship has not been established. Crohn's disease is not an approved indication for Metronidazole Injection.
Drug Interactions
- Disulfiram Psychotic reactions and confusion have been reported in alcoholic patients who are using metronidazole and disulfiram concurrently. Do not administer Metronidazole Injection to patients who have taken disulfiram within the last two weeks (see CONTRAINDICATIONS ).
- Alcoholic Beverages Abdominal cramps, nausea, vomiting, headaches, tachycardia and flushing may occur if alcoholic beverages or products containing propylene glycol are consumed during or following metronidazole therapy. Discontinue consumption of alcohol or products containing propylene glycol before, during and up to 72 hours after therapy with Metronidazole Injection (see CONTRAINDICATIONS ).
- Warfarin and other Oral Anticoagulants Metronidazole has been reported to potentiate the anticoagulant effect of warfarin and other oral coumarin anticoagulants, resulting in a prolongation of prothrombin time and increased risk of hemorrhages. When Metronidazole Injection is prescribed for patients on this type of anticoagulant therapy, prothrombin time and international normalized ratio (INR) should be carefully monitored and their anticoagulant dose adjusted accordingly. Monitor patients for signs and symptoms of bleeding.
- Lithium In patients stabilized on relatively high doses of lithium, short-term metronidazole therapy has been associated with elevation of serum lithium and, in a few cases, signs of lithium toxicity. Lithium toxicity may lead to renal damage. Frequent monitoring of serum lithium and serum creatinine levels is necessary.
- Busulfan Metronidazole has been reported to increase plasma concentrations of busulfan, which can result in an increased risk for serious busulfan toxicity such as sinusoidal obstruction syndrome, gastrointestinal mucositis, and hepatic veno-occlusive disease. Metronidazole Injection should not be administered concomitantly with busulfan unless the benefit outweighs the risk. If no therapeutic alternatives to metronidazole are available, and concomitant administration with busulfan is medically needed, frequent monitoring of busulfan plasma concentration should be performed and the busulfan dose should be adjusted accordingly.
- Drugs that Inhibit CYP450 Enzymes The simultaneous administration of drugs that decrease microsomal liver enzyme activity, such as cimetidine, may decrease metabolism and reduce plasma clearance of metronidazole which may result in metronidazole toxicity.
- Drugs that Induce CYP450 Enzymes The simultaneous administration of drugs that induce microsomal liver enzyme activity, such as phenytoin or phenobarbital, may accelerate the elimination of metronidazole and therefore decrease its efficacy.
- Cytochrome P450 3A4 (CYP3A4) substrates Concomitant use of Metronidazole Injection and CYP3A4 substrates (e.g., amiodarone, tacrolimus, cyclosporine, carbamazepine, phenytoin, and quinidine) may increase respective CYP3A4-substrate plasma levels. Monitoring of plasma concentrations of CYP3A4 substrates may be necessary.
- 5-Fluorouracil Metronidazole Injection decreases the clearance of 5-fluorouracil and may therefore cause 5-fluorouracil toxicity.
- Vecuronium Metronidazole Injection may potentiate the effects of vecuronium.
- Drugs that Prolong the QT interval QT prolongation has been reported, particularly when metronidazole was administered with drugs with the potential for prolonging the QT interval.
- Drug/Laboratory Test Interactions Metronidazole may interfere with certain types of determinations of serum chemistry values, such as aspartate aminotransferase (AST, SGOT), alanine aminotransferase (ALT, SGPT), lactate dehydrogenase (LDH), triglycerides and glucose hexokinase. Metronidazole causes an increase in ultraviolet absorbance at 340 nm resulting in falsely decreased values.
DESCRIPTION
Metronidazole Injection, USP, is a parenteral formulation of the synthetic nitroimidazole antibacterial agent 2-methyl-5-nitro-1 H -imidazole-1-ethanol.
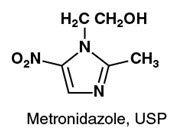
Metronidazole Injection, USP, in 100 mL VIAFLEX Plus single dose plastic container, is a sterile, nonpyrogenic, iso-osmotic, buffered solution of 500 mg Metronidazole, USP, 790 mg Sodium Chloride, USP, 47.6 mg Dibasic Sodium Phosphate Dried, USP and 22.9 mg Citric Acid Anhydrous, USP. Metronidazole Injection, USP has an osmolarity of 310 mOsmol/L (calc) and a pH of 5.5 (4.5 to 7.0). Each container contains 14 mEq of sodium.
The plastic container is fabricated from a specially formulated polyvinyl chloride plastic. Water can permeate from inside the container into the overwrap in amounts insufficient to affect the solution significantly. Solutions in contact with the plastic container can leach out certain of its chemical components in very small amounts within the expiration period, e.g. , di-2-ethylhexyl phthalate (DEHP), up to 5 parts per million. However, the safety of the plastic has been confirmed in tests in animals according to USP biological tests for plastic containers as well as by tissue culture toxicity studies.
CLINICAL PHARMACOLOGY
In patients treated with intravenous metronidazole, using a dosage regimen of 15 mg/kg loading dose followed 6 hours later by 7.5 mg/kg every 6 hours, the average peak steady-state plasma concentrations (C max ) and trough concentrations (C min ) were 25 mcg/mL and 18 mcg/mL, respectively. Plasma concentrations of metronidazole are proportional to the administered dose. An eight-hour intravenous infusion of 100 mg to 4,000 mg of metronidazole in normal subjects showed a linear relationship between dose and peak plasma concentration. The average elimination half-life of metronidazole in healthy subjects is eight hours.
Distribution
Metronidazole is the major component appearing in the plasma, with lesser quantities of metabolites also being present. Less than 20% of the circulating metronidazole is bound to plasma proteins. Metronidazole appears in cerebrospinal fluid, saliva and breast milk in concentrations similar to those found in plasma. Bactericidal concentrations of metronidazole have also been detected in pus from hepatic abscesses.
Following a single intravenous dose of metronidazole 500 mg, 4 healthy subjects who underwent gastrointestinal endoscopy had peak gastric juice metronidazole concentrations of 5 to 6 mcg/mL at one hour post-dose. In patients receiving intravenous metronidazole in whom gastric secretions are continuously removed by nasogastric aspiration, sufficient metronidazole may be removed in the aspirate to cause a reduction in serum levels.
Metabolism
The metabolites of metronidazole result primarily from side-chain oxidation [1-(ß-hydroxyethyl)-2-hydroxymethyl-5-nitroimidazole and 2-methyl-5-nitroimidazole-1-yl-acetic acid] and glucuronide conjugation. Both the parent compound and the hydroxyl metabolite possess in vitro antimicrobial activity.
Excretion
The major route of elimination of metronidazole and its metabolites is via the urine (60-80% of the dose), with approximately 20% of the amount excreted appearing as unchanged metronidazole. Renal clearance of metronidazole is approximately 10 mL/min/1.73 m 2 . Fecal excretion accounts for 6-15% of the dose.
Renal Impairment
Decreased renal function does not alter the single-dose pharmacokinetics of metronidazole.
Subjects with end-stage renal disease (ESRD; CL CR = 8.1±9.1 mL/min) and who received a single intravenous infusion of metronidazole 500 mg had no significant change in metronidazole pharmacokinetics but had 2-fold higher Cmax of hydroxy-metronidazole and 5-fold higher Cmax of metronidazole acetate, compared to healthy subjects with normal renal function (CL CR = 126 ± 16 mL/min). Thus, on account of the potential accumulation of metronidazole metabolites in ESRD patients, monitoring for metronidazole associated adverse events is recommended (see PRECAUTIONS ).
Effect of Dialysis
Following a single intravenous infusion or oral dose of metronidazole 500 mg, the clearance of metronidazole was investigated in ESRD subjects undergoing hemodialysis or continuous ambulatory peritoneal dialysis (CAPD). A hemodialysis session lasting for 4 to 8 hours removed 40% to 65% of the administered metronidazole dose, depending on the type of the dialyzer membrane used and the duration of the dialysis session. If the administration of metronidazole cannot be separated from the dialysis session, supplementation of metronidazole dose following hemodialysis should be considered (see DOSAGE AND ADMINISTRATION ). A peritoneal dialysis session lasting for 7.5 hours removed approximately 10% of the administered metronidazole dose. No adjustment in metronidazole dose is needed in ESRD patients undergoing CAPD.
Hepatic Impairment
Following a single intravenous infusion of 500 mg metronidazole, the mean AUC 24 of metronidazole was higher by 114% in patients with severe (Child-Pugh C) hepatic impairment, and by 54% and 53% in patients with a mild (Child-Pugh A) and moderate (Child-Pugh B) hepatic impairment, respectively, compared to healthy control subjects. There were no significant changes in the AUC 24 of hydroxy-metronidazole in these hepatically impaired patients. A reduction in metronidazole dosage by 50% is recommended in patients with severe (Child-Pugh C) hepatic impairment (see DOSAGE AND ADMINISTRATION ). No dosage adjustment is needed for patients with mild to moderate hepatic impairment. Patients with mild to moderate hepatic impairment should be monitored for metronidazole associated adverse events (see PRECAUTIONS and DOSAGE AND ADMINISTRATION ).
Geriatric Patients
Following a single 500 mg oral or IV dose of metronidazole, subjects >70 years old with no apparent renal or hepatic dysfunction had a 40% to 80% higher mean AUC of hydroxy-metronidazole (active metabolite), with no apparent increase in the mean AUC of metronidazole (parent compound), compared to young healthy controls < 40 years old. In geriatric patients, monitoring for metronidazole associated adverse events is recommended (see PRECAUTIONS ).
Pediatric Patients
In one study newborn infants appeared to demonstrate diminished capacity to eliminate metronidazole. The elimination half-life, measured during the first three days of life, was inversely related to gestational age. In infants whose gestational ages were between 28 and 40 weeks, the corresponding elimination half-lives ranged from 109 to 22.5 hours.
Two other trials included infants 23-48 weeks post-menstrual age, (0-80 days postnatal age) with complicated intra-abdominal infections. Clearance increased with increasing post-menstrual age, while volume of distribution and half-life decreased ( see PRECAUTIONS , Pediatric Use and DOSAGE AND ADMINISTRATION ).
Microbiology
Mechanism of Action
Metronidazole, a nitroimidazole, exerts antibacterial effects in an anaerobic environment against most obligate anaerobes. Once metronidazole enters the organism by passive diffusion and is activated in the cytoplasm of susceptible anaerobic bacteria, it is reduced; this process includes intra-cellular electron transport proteins such as ferredoxin, transfer of an electron to the nitro group of the metronidazole, and formation of a short-lived nitroso free radical. Because of this alteration of the metronidazole molecule, a concentration gradient is created and maintained which promotes the drug’s intracellular transport. The reduced form of metronidazole and free radicals can interact with DNA leading to inhibition of DNA synthesis and DNA degradation leading to death of bacteria. The precise mechanism of action of metronidazole is unclear.
Drug Resistance
A potential for development of resistance exists against metronidazole.
Resistance may be due to multiple mechanisms that include decreased uptake of the drug, altered reduction efficiency, overexpression of the efflux pumps, inactivation of the drug, and/or increased DNA damage repair.
Metronidazole does not possess any clinically relevant activity against facultative anaerobes or obligate aerobes.
Activity In Vitro and in Clinical Infections
Metronidazole has been shown to be active against most isolates of the following bacteria both in vitro and in clinical infections as described in the INDICATIONS AND USAGE section.
Gram-positive anaerobes
Clostridium species Eubacterium species Peptococcus species Peptostreptococcus species
Gram-negative anaerobes
Bacteroides fragilis group ( B. fragilis , B. distasonis , B. ovatus , B. thetaiotaomicron , B. vulgatus ) Fusobacterium species
The following in vitro data are available, but their clinical significance is unknown.
Metronidazole exhibits in vitro minimal inhibitory concentrations (MIC’s) of 8 mcg/mL or less against most (≥ 90%) isolates of the following bacteria; however, the safety and effectiveness of metronidazole in treating clinical infections due to these bacteria have not been established in adequate and well-controlled clinical trials.
Gram-negative anaerobes
Bacteroides fragilis group ( B. caccae, B. uniformis ) Prevotella species ( P. bivia, P. buccae, P. disiens )
Susceptibility Tests
For specific information regarding susceptibility test interpretive criteria and associated test methods and quality control standards recognized by FDA for this drug, please see: http://www.fda.gov/STIC .
HOW SUPPLIED
Metronidazole Injection, USP is supplied in 100 mL single dose plastic containers, each containing an iso-osmotic, buffered solution of 500 mg metronidazole as follows:
2B3423 NDC 72572-540-24 500 mg/100 mL
Store at controlled room temperature (77°F or 25°C) and protect from light during storage. Do not remove unit from overwrap until ready for use. The overwrap is a moisture barrier. The inner bag maintains the sterility of the product. After removing overwrap, check for minute leaks by squeezing inner bag firmly. If leaks are found, discard solution as sterility may be impaired.
Mechanism of Action
Metronidazole, a nitroimidazole, exerts antibacterial effects in an anaerobic environment against most obligate anaerobes. Once metronidazole enters the organism by passive diffusion and is activated in the cytoplasm of susceptible anaerobic bacteria, it is reduced; this process includes intra-cellular electron transport proteins such as ferredoxin, transfer of an electron to the nitro group of the metronidazole, and formation of a short-lived nitroso free radical. Because of this alteration of the metronidazole molecule, a concentration gradient is created and maintained which promotes the drug’s intracellular transport. The reduced form of metronidazole and free radicals can interact with DNA leading to inhibition of DNA synthesis and DNA degradation leading to death of bacteria. The precise mechanism of action of metronidazole is unclear.