Get your patient on Minocycline Hydrochloride - Minocycline Hydrochloride tablet (Minocycline Hydrochloride)
Minocycline Hydrochloride - Minocycline Hydrochloride tablet prescribing information
INDICATIONS AND USAGE
Minocycline hydrochloride tablets are indicated in the treatment of the following infections due to susceptible strains of the designated microorganisms:
- Rocky Mountain spotted fever, typhus fever and the typhus group, Q fever, rickettsialpox and tick fevers caused by Rickettsiae.
- Respiratory tract infections caused by Mycoplasma pneumoniae.
- Lymphogranuloma venereum caused by Chlamydia trachomatis.
- Psittacosis (Ornithosis) due to Chlamydia psittaci.
- Trachoma caused by Chlamydia trachomatis, although the infectious agent is not always eliminated, as judged by immunofluorescence.
- Inclusion conjunctivitis caused by Chlamydia trachomatis.
- Nongonococcal urethritis, endocervical, or rectal infections in adults caused by Ureaplasma urealyticum or Chlamydia trachomatis.
- Relapsing fever due to Borrelia recurrentis.
- Chancroid caused by Haemophilus ducreyi
- Plague due to Yersinia pestis.
- Tularemia due to Francisella tularensis.
- Cholera caused by Vibrio cholerae.
- Campylobacter fetus infections caused by Campylobacter fetus.
- Brucellosis due to Brucella species (in conjunction with streptomycin).
- Bartonellosis due to Bartonella bacilliformis.
- Granuloma inguinale caused by Calymmatobacterium granulomatis.
Minocycline is indicated for treatment of infections caused by the following gram-negative microorganisms when bacteriologic testing indicates appropriate susceptibility to the drug:
- Escherichia coli.
- Enterobacter aerogenes.
- Shigella species.
- Acinetobacter species.
- Respiratory tract infections caused by Haemophilus influenzae.
- Respiratory tract and urinary tract infections caused by Klebsiella species.
Minocycline hydrochloride tablets are indicated for the treatment of infections caused by the following gram-positive microorganisms when bacteriologic testing indicates appropriate susceptibility to the drug:
- Upper respiratory tract infections caused by Streptococcus pneumoniae
- Skin and skin structure infections caused by Staphylococcus aureus. (Note: Minocycline is not the drug of choice in the treatment of any type of staphylococcal infection.)
When penicillin is contraindicated, minocycline is an alternative drug in the treatment of the following infections:
- Uncomplicated urethritis in men due to Neisseria gonorrhoeae and for the treatment of other gonococcal infections
- Infections in women caused by Neisseria gonorrhoeae.
- Syphilis caused by Treponema pallidum subspecies pallidum.
- Yaws caused by Treponema pallidum subspecies pertenue.
- Listeriosis due to Listeria monocytogenes.
- Anthrax due to Bacillus anthracis.
- Vincent's infection caused by Fusobacterium fusiforme.
- Actinomycosis caused by Actinomyces israelii.
- Infections caused by Clostridium species.
In acute intestinal amebiasis , minocycline may be a useful adjunct to amebicides.
In severe acne , minocycline may be useful adjunctive therapy.
Oral minocycline is indicated in the treatment of asymptomatic carriers of Neisseria meningitidis to eliminate the meningococci from the nasopharynx. In order to preserve the usefulness of minocycline in the treatment of asymptomatic meningococcal carriers, diagnostic laboratory procedures, including serotyping and susceptibility testing, should be performed to establish the carrier state and the correct treatment. It is recommended that the prophylactic use of minocycline be reserved for situations in which the risk of meningococcal meningitis is high.
Oral minocycline is not indicated for the treatment of meningococcal infection.
Although no controlled clinical efficacy studies have been conducted, limited clinical data show that oral minocycline hydrochloride has been used successfully in the treatment of infections caused by Mycobacterium marinum .
To reduce the development of drug-resistant bacteria and maintain the effectiveness of, minocycline hydrochloride tablets and other antibacterial drugs, minocycline hydrochloride tablets should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
DOSAGE AND ADMINISTRATION
THE USUAL DOSAGE AND FREQUENCY OF ADMINISTRATION OF MINOCYCLINE DIFFERS FROM THAT OF THE OTHER TETRACYCLINES. EXCEEDING THE RECOMMENDED DOSAGE MAY RESULT IN AN INCREASED INCIDENCE OF SIDE EFFECTS.
Minocycline hydrochloride tablets may be taken with or without food (see CLINICAL PHARMACOLOGY ).
Ingestion of adequate amounts of fluids along with capsule and tablet forms of drugs in the tetracycline-class is recommended to reduce the risk of esophageal irritation and ulceration. The tablets should be swallowed whole.
For Pediatric Patients Above 8 Years of Age
Usual pediatric dose: 4 mg/kg initially followed by 2 mg/kg every 12 hours, not to exceed the usual adult dose.
Adults
The usual dosage of minocycline hydrochloride tablets is 200 mg initially followed by 100 mg every 12 hours. Alternatively, if more frequent doses are preferred, two or four 50 mg tablets may be given initially followed by one 50 mg tablet four times daily.
Uncomplicated gonococcal infections other than urethritis and anorectal infections in men: 200 mg initially, followed by 100 mg every 12 hours for a minimum of four days, with post-therapy cultures within 2 to 3 days.
In the treatment of uncomplicated gonococcal urethritis in men, 100 mg every 12 hours for 5 days is recommended.
For the treatment of syphilis, the usual dosage of minocycline hydrochloride should be administered over a period of 10 to 15 days. Close follow-up, including laboratory tests, is recommended.
In the treatment of meningococcal carrier state, the recommended dosage is 100 mg every 12 hours for five days.
Mycobacterium marinum infections: Although optimal doses have not been established, 100 mg every 12 hours for 6 to 8 weeks have been used successfully in a limited number of cases.
Uncomplicated urethral, endocervical, or rectal infection in adults caused by Chlamydia trachomatis or Ureaplasma urealyticum : 100 mg orally, every 12 hours for at least seven days.
Ingestion of adequate amounts of fluids along with capsule and tablet forms of drugs in the tetracycline-class is recommended to reduce the risk of esophageal irritation and ulceration.
The pharmacokinetics of minocycline in patients with renal impairment (CL CR <80mL/min) have not been fully characterized. Current data are insufficient to determine if a dosage adjustment is warranted. The total daily dosage should not exceed 200 mg in 24 hours. However, due to the anti-anabolic effect of tetracyclines, BUN and creatinine should be monitored (see WARNINGS ).
CONTRAINDICATIONS
This drug is contraindicated in persons who have shown hypersensitivity to any of the tetracyclines or to any of the components of the product formulation.
ADVERSE REACTIONS
Due to oral minocycline's virtually complete absorption, side effects to the lower bowel, particularly diarrhea, have been infrequent. The following adverse reactions have been observed in patients receiving tetracyclines.
Body as a whole: Fever, and discoloration of secretions.
Gastrointestinal : Anorexia, nausea, vomiting, diarrhea, dyspepsia, stomatitis, glossitis, dysphagia, enamel hypoplasia, enterocolitis, pseudomembranous colitis, pancreatitis, inflammatory lesions (with monilial overgrowth) in the oral and anogenital regions. Instances of esophagitis and esophageal ulcerations have been reported in patients taking the tetracycline-class antibiotics in capsule and tablet form. Most of these patients took the medication immediately before going to bed (see DOSAGE AND ADMINISTRATION ).
Genitourinary: Vulvovaginitis.
Hepatic toxicity: Hyperbilirubinemia, hepatic cholestasis, increases in liver enzymes, fatal hepatic failure, and jaundice. Hepatitis, including autoimmune hepatitis, and liver failure have been reported (see PRECAUTIONS ).
Skin: Alopecia, erythema nodosum, hyperpigmentation of nails, pruritus, toxic epidermal necrolysis, and vasculitis. Maculopapular and erythematous rashes. Exfoliative dermatitis has been reported. Fixed drug eruptions have been reported. Lesions occurring on the glans penis have caused balanitis. Erythema multiforme and Stevens-Johnson syndrome have been reported. Photosensitivity is discussed above (see WARNINGS ). Pigmentation of the skin and mucous membranes has been reported.
Respiratory: Cough, dyspnea, bronchospasm, exacerbation of asthma, and pneumonitis.
Renal toxicity: Interstitial nephritis. Elevations in BUN have been reported and are apparently dose related (see WARNINGS ). Reversible acute renal failure has been reported.
Musculoskeletal: Arthralgia, arthritis, bone discoloration, myalgia, joint stiffness, and joint swelling.
Hypersensitivity reactions: Urticaria, angioneurotic edema, polyarthralgia, anaphylaxis/ anaphylactoid reaction (including shock and fatalities) anaphylactoid purpura, myocarditis, pericarditis, exacerbation of systemic lupus erythematosus and pulmonary infiltrates with eosinophilia have been reported. A transient lupus-like syndrome and serum sickness-like reactions have also been reported.
Blood: Agranulocytosis, hemolytic anemia, thrombocytopenia, leukopenia, neutropenia, pancytopenia, and eosinophilia have been reported.
Central Nervous System: Convulsions, dizziness, hypesthesia, paresthesia, sedation, and vertigo. Bulging fontanels in infants and benign intracranial hypertension (pseudotumor cerebri) in adults have been reported (see PRECAUTIONS-General ). Headache has also been reported.
Other: Thyroid cancer has been reported in the post-marketing setting in association with minocycline products. When minocycline therapy is given over prolonged periods, monitoring for signs of thyroid cancer should be considered. When given over prolonged periods, tetracyclines have been reported to produce brown-black microscopic discoloration of the thyroid gland. Cases of abnormal thyroid function have been reported.
Tooth discoloration in children less than 8 years of age, and also in adults has been reported (see WARNINGS ).
Oral cavity discoloration (including tongue, lip, and gum) have been reported.
Tinnitus and decreased hearing have been reported in patients on minocycline hydrochloride.
The following syndromes have been reported. In some cases involving these syndromes, death has been reported. As with other serious adverse reactions, if any of these syndromes are recognized, the drug should be discontinued immediately:
Hypersensitivity syndrome consisting of cutaneous reaction (such as rash or exfoliative dermatitis), eosinophilia, and one or more of the following: hepatitis, pneumonitis, nephritis, myocarditis, and pericarditis. Fever and lymphadenopathy may be present.
Lupus-like syndrome consisting of positive antinuclear antibody; arthralgia, arthritis, joint stiffness, or joint swelling; and one or more of the following: fever, myalgia, hepatitis, rash, and vasculitis.
Serum sickness-like syndrome consisting of fever; urticaria or rash; and arthralgia, arthritis, joint stiffness, or joint swelling. Eosinophilia may be present.
To report SUSPECTED ADVERSE REACTIONS, contact Strides Pharma Inc. at 1-877-244-9825 or go to www.strides.com or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
Because tetracyclines have been shown to depress plasma prothrombin activity, patients who are on anticoagulant therapy may require downward adjustment of their anticoagulant dosage.
Since bacteriostatic drugs may interfere with the bactericidal action of penicillin, it is advisable to avoid giving tetracycline-class drugs in conjunction with penicillin.
Absorption of tetracyclines is impaired by antacids containing aluminum, calcium, or magnesium, and iron-containing preparations.
The concurrent use of tetracycline and methoxyflurane has been reported to result in fatal renal toxicity.
Concurrent use of tetracyclines with oral contraceptives may render oral contraceptives less effective.
Administration of isotretinoin should be avoided shortly before, during, and shortly after minocycline therapy. Each drug alone has been associated with pseudotumor cerebri (see PRECAUTIONS ).
Increased risk of ergotism when ergot alkaloids or their derivatives are given with tetracyclines.
DESCRIPTION
Minocycline hydrochloride, is a semisynthetic derivative of tetracycline, 4,7-Bis (dimethylamino)-1,4,4a,5,5a,6,11,12a-octahydro-3,10,12,12a-tetrahydroxy-1,11-dioxo-2-naphthacenecarboxamide monohydrochloride. Its structural formula is:
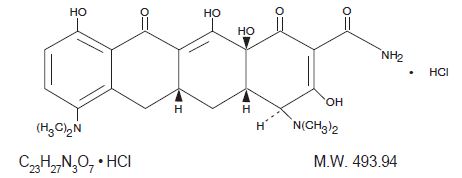
Minocycline hydrochloride tablets for oral administration contain minocycline HCl equivalent to 50 mg, 75 mg or 100 mg of minocycline. In addition, 50 mg, 75 mg and 100 mg tablets contain the following inactive ingredients: Microcrystalline Cellulose NF, Lactose Anhydrous NF, Povidone USP, Colloidal Silicon Dioxide NF, Magnesium Stearate NF, and Sodium Starch Glycolate NF. The 50 mg, 75 mg and 100 mg tablets also contain Opadry White which contains: Titanium Dioxide USP, Hypromellose Type 2910 USP, Polyethylene Glycol 400 NF, and Polysorbate 80 NF.
CLINICAL PHARMACOLOGY
Following a single dose of one 100 mg tablet of minocycline hydrochloride administered to 28 normal fasting adult volunteers, maximum serum concentrations were attained in 1 to 3 hours (average 1.71 hours) and ranged from 491.71 to 1292.70 ng/mL (average 758.29 ng/mL). The serum half-life in the normal volunteers ranged from 11.38 to 24.31 hours (average 17.03 hours).
When minocycline hydrochloride tablets were given concomitantly with a meal, which included dairy products, the extent of absorption of minocycline hydrochloride tablets was slightly decreased (6%). The peak plasma concentrations were slightly decreased (12%) and delayed by 1.09 hours when administered with food, compared to dosing under fasting conditions. Minocycline HCl may be administered with or without food.
In previous studies with other minocycline dosage forms, the minocycline serum half-life ranged from 11 to 16 hours in 7 patients with hepatic dysfunction, and from 18 to 69 hours in 5 patients with renal dysfunction. The urinary and fecal recovery of minocycline when administered to 12 normal volunteers was one-half to one-third that of other tetracyclines.
Microbiology
The tetracyclines are primarily bacteriostatic and are thought to exert their antimicrobial effect by the inhibition of protein synthesis. The tetracyclines, including minocycline, have similar antimicrobial spectra of activity against a wide range of gram-positive and gram-negative organisms. Cross-resistance of these organisms to tetracycline is common.
Minocycline has been shown to be active against most strains of the following microorganisms, both in vitro and in clinical infections as described in the INDICATIONS AND USAGE section:
AEROBIC GRAM-POSITIVE MICROORGANISMS:
Because many strains of the following gram-positive microorganisms have been shown to be resistant to tetracyclines, culture and susceptibility testing are especially recommended. Tetracycline antibiotics should not be used for streptococcal diseases unless the organism has been demonstrated to be susceptible. Tetracyclines are not the drug of choice in the treatment of any type of staphylococcal infection.
Bacillus anthracis a
Listeria monocytogenes a
Staphylococcus aureus
Streptococcus pneumoniae
AEROBIC GRAM-NEGATIVE MICROORGANISMS
Bartonella bacilliformis
Brucella species
Calymmatobacterium granulomatis
Campylobacter fetus
Francisella tularensis
Haemophilus ducreyi
Vibrio cholerae
Yersinia pestis
Because many strains of the following groups of gram-negative microorganisms have been shown to be resistant to tetracyclines, culture and susceptibility tests are especially recommended.
Acinetobacter species
Enterobacter aerogenes
Escherichia coli
Haemophilus influenzae
Klebsiella species
Neisseria gonorrhoeae a
Neisseria meningitidis a
Shigella species
"OTHER" MICROORGANISMS
Actinomyces species a
Borrelia recurrentis
Chlamydia psittaci
Chlamydia trachomatis
Clostridium species a
Entamoeba species
Fusobacterium nucleatum subspecies fusiforme a
Mycobacterium marinum
Mycoplasma pneumoniae
Propionibacterium acnes
Rickettsiae
Treponema pallidum subspecies pallidum a
Treponema pallidum subspecies pertenue a
Ureaplasma urealyticum
a. When penicillin is contraindicated, tetracyclines are alternative drugs in the treatment of infections caused by the cited microorganisms.
Susceptibility Tests
Susceptibility testing should be performed with tetracycline since it predicts susceptibility to minocycline. However, certain organisms (e.g., some staphylococci, and Acinetobacter species) may be more susceptible to minocycline and doxycycline than tetracycline.
Dilution techniques:
Quantitative methods are used to determine antimicrobial minimal inhibitory concentrations (MICs). These MICs provide estimates of the susceptibility of bacteria to antimicrobial compounds. The MICs should be determined using a standardized procedure. Standardized procedures are based on a dilution method 1,3 (broth or agar) or equivalent with standardized inoculum concentrations and standardized concentrations of tetracycline powder. The MIC values should be interpreted according to the following criteria:
For testing aerobic gram-negative microorganisms (Enterobacteriaceae), Acinetobacter species and Staphylococcus aureus:
| MIC (mcg/mL) | Interpretation |
| ≤ 4 | Susceptible (S) |
| 8 | Intermediate (I) |
| ≥ 16 | Resistant (R) |
For testing Haemophilus influenzae b and Streptococcus pneumoniae c :
| MIC (mcg/mL) | Interpretation |
| ≤ 2 | Susceptible (S) |
| 4 | Intermediate (I) |
| ≥ 8 | Resistant (R) |
b. These interpretative standards are applicable only to broth microdilution susceptibility testing with Haemophilus influenzae using Haemophilus Test Medium. 1
c. These interpretative standards are applicable only to broth microdilution susceptibility testing using cation-adjusted Muller-Hinton broth with 2 to 5% lysed horse blood. 1
For testing Neisseria gonorrhoeae d :
| MIC (mcg/mL) | Interpretation |
| ≤ 0.25 | Susceptible (S) |
| 0.5-1 | Intermediate (I) |
| ≥ 2 | Resistant (R) |
d. These interpretative standards are applicable only to agar dilution susceptibility testing using GC agar base and 1% defined growth supplements. 1
A report of "Susceptible" indicates that the pathogen is likely to be inhibited if the antimicrobial compound in the blood reaches the concentrations usually achievable. A report of "Intermediate" indicates that the result should be considered equivocal, and, if the microorganism is not fully susceptible to alternative, clinically feasible drugs, the test should be repeated. This category implies possible clinical applicability in body sites where the drug is physiologically concentrated or in situations where high dosage of drug can be used. This category also provides a buffer zone which prevents small uncontrolled technical factors from causing major discrepancies in interpretation. A report of "Resistant" indicates that the pathogen is not likely to be inhibited if the antimicrobial compound in the blood reaches the concentrations usually achievable; other therapy should be selected.
Standardized susceptibility test procedures require the use of laboratory control microorganisms to control the technical aspects of the laboratory procedures. Standard tetracycline powder should provide the following MIC values:
| Microorganism | MIC (mcg/mL) | |
| Escherichia coli | ATCC 25922 | 0.5-2 |
| Enterococcus faecalis | ATCC 29212 | 8-32 |
| Staphylococcus aureus | ATCC 29213 | 0.25-1 |
| Haemophilus influenzae | ATCC 49247 | 4-32 |
| Streptococcus pneumoniae | ATCC 49619 | 0.12-0.5 |
| Neisseria gonorrhoeae | ATCC 49226 | 0.25-1 |
Diffusion techniques:
Quantitative methods that require measurement of zone diameters also provide reproducible estimates of the susceptibility of bacteria to antimicrobial compounds. One such standardized procedure 2,3 requires the use of standardized inoculum concentrations. This procedure uses paper disks impregnated with 30 mcg tetracycline (class disk) or 30 mcg minocycline to test the susceptibility of microorganisms to minocycline.
Reports from the laboratory providing results of the standard single-disk susceptibility test with a 30 mcg tetracycline or minocycline disk should be interpreted according to the following criteria:
For testing aerobic gram-negative microorganisms (Enterobacteriaceae), Acinetobacter species and Staphylococcus aureus :
| Zone Diameter (mm) | Interpretation |
| ≥ 19 | Susceptible (S) |
| 15-18 | Intermediate (I) |
| ≤ 14 | Resistant (R) |
For testing Haemophilus influenzae e :
| Zone Diameter (mm) | Interpretation |
| ≥ 29 | Susceptible (S) |
| 26-28 | Intermediate (I) |
| ≤ 25 | Resistant (R) |
e. These zone diameter standards are applicable only to susceptibility testing with Haemophilus influenzae using Haemophilus Test Medium and a 30 mcg tetracycline disk. 2
For testing Neisseria gonorrhoeae f :
| Zone Diameter (mm) | Interpretation |
| ≥ 38 | Susceptible (S) |
| 31-37 | Intermediate (I) |
| ≤ 30 | Resistant (R) |
f. These interpretative standards are applicable only to disk diffusion testing using GC agar and 1% growth supplements, and a 30 mcg tetracycline disk. 2
For testing Streptococcus pneumoniae g :
| Zone Diameter (mm) | Interpretation |
| ≥ 23 | Susceptible (S) |
| 19-22 | Intermediate (I) |
| ≤ 18 | Resistant (R) |
g. These interpretative standards are applicable only to disk diffusion testing using Muller-Hinton agar adjusted with 5% sheep blood and a 30 mcg tetracycline class disk. 2
For testing Vibrio cholerae h :
| Zone Diameter (mm) | Interpretation |
| ≥ 19 | Susceptible (S) |
| 15-18 | Intermediate (I) |
| ≤ 14 | Resistant (R) |
h. These interpretative standards are applicable only to disk diffusion testing performed with a 30 mcg tetracycline class disk. 2
Interpretation should be as stated above for results using dilution techniques. Interpretation involves correlation of the diameter obtained in the disk test with the MIC for tetracycline.
As with standardized dilution techniques, diffusion methods require the use of laboratory control microorganisms that are used to control the technical aspects of the laboratory procedures. For the diffusion technique, the 30 mcg tetracycline or minocycline disk should provide the following zone diameters in these laboratory test quality control strains:
| Microorganism | Zone Diameter Range (mm) | ||
| Tetracycline | Minocycline | ||
| Escherichia coli | ATCC25922 | 18-25 | 19-25 |
| Staphylococcus aureus | ATCC 29213 | 24-30 | 25-30 |
| Haemophilus influenzae | ATCC 49247 | 14-22 | --- |
| Neisseria gonorrhoeae | ATCC 49226 | 30-42 | --- |
| Streptococcus pneumoniae | ATCC 49619 | 27-31 | --- |
HOW SUPPLIED
Minocycline hydrochloride tablets are supplied as aqueous film coated tablets containing minocycline hydrochloride equivalent to 50 mg, 75 mg and 100 mg minocycline.
The 50 mg tablets are white, unscored, modified capsule shaped, coated tablet debossed "Par" on one side and "511" on the other. Each tablet contains minocycline hydrochloride equivalent to 50 mg minocycline, supplied as follows:
NDC 64380-154-01 Bottle of 100
The 75 mg tablets are white, unscored, modified capsule shaped, coated tablet debossed "Par" on one side and "512" on the other. Each tablet contains minocycline hydrochloride equivalent to 75 mg minocycline, supplied as follows:
NDC 64380-155-01 Bottle of 100
The 100 mg tablets are white, unscored, modified capsule shaped, coated tablet debossed "Par" on one side and "513" on the other. Each tablet contains minocycline hydrochloride equivalent to 100 mg minocycline, supplied as follows:
NDC 64380-156-01 Bottle of 50
Store at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature].
Protect from light, moisture and excessive heat.
Dispense in a tight, light-resistant container as defined in the USP.