Get your patient on Norgestimate And Ethinyl Estradiol - Norgestimate And Ethinyl Estradiol (Norgestimate And Ethinyl Estradiol)
Norgestimate And Ethinyl Estradiol - Norgestimate And Ethinyl Estradiol prescribing information
WARNING: CIGARETTE SMOKING and SERIOUS CARDIOVASCULAR EVENTS
Cigarette smoking increases the risk of serious cardiovascular events from combination oral contraceptive (COC) use. This risk increases with age, particularly in women over 35 years of age, and with the number of cigarettes smoked. For this reason, COCs are contraindicated in women who are over 35 years of age and smoke [see Contraindications (4 )].
Contraindications, Pregnancy (4 ) Removed 06/2023
INDICATIONS AND USAGE
Norgestimate and ethinyl estradiol tablets are combinations of norgestimate, a progestin, and ethinyl estradiol, an estrogen, indicated for use by females of reproductive potential to prevent pregnancy. (1.1 )
Norgestimate and ethinyl estradiol tablets 0.18 mg/0.035 mg, 0.215 mg/0.035 mg, 0.25 mg/0.035 mg are indicated for the treatment of moderate acne vulgaris in females at least 15 years of age, who have no known contraindications to oral contraceptive therapy and have achieved menarche.
Norgestimate and ethinyl estradiol tablets 0.18 mg/0.035 mg, 0.215 mg/0.035 mg, 0.25 mg/0.035 mg should be used for the treatment of acne only if the patient desires an oral contraceptive for birth control. (1.2 )
Oral Contraceptive
Norgestimate and Ethinyl Estradiol Tablets, 0.18 mg/0.035 mg, 0.215 mg/0.035 mg, 0.25 mg/0.035 mg and Norgestimate and Ethinyl Estradiol Tablets, 0.25 mg/0.035 mg are indicated for use by females of reproductive potential to prevent pregnancy [see Clinical Studies (14 )].
Acne
Norgestimate and Ethinyl Estradiol Tablets, 0.18 mg/0.035 mg, 0.215 mg/0.035 mg, 0.25 mg/0.035 mg are indicated for the treatment of moderate acne vulgaris in females at least 15 years of age, who have no known contraindications to oral contraceptive therapy and have achieved menarche. Norgestimate and Ethinyl Estradiol Tablets, 0.18 mg/0.035 mg, 0.215 mg/0.035 mg, 0.25 mg/0.035 mg should be used for the treatment of acne only if the patient desires an oral contraceptive for birth control [see Clinical Studies (14 )].
DOSAGE AND ADMINISTRATION
Recommended Dosage and Administration
Take one tablet by mouth at the same time each day with or without food. Table 1 provides the recommended dosage and administration instructions for norgestimate and ethinyl estradiol tablets, 0.25 mg/0.035 mg and Norgestimate and ethinyl estradiol tablets, 0.18 mg/0.035 mg, 0.215 mg/0.035 mg, 0.25 mg/0.035 mg.
Table 1: Instructions for Administration of Norgestimate and Ethinyl Estradiol Tablets, 0.25 mg/0.035 mg, or Norgestimate and Ethinyl Estradiol Tablets, 0.18 mg/0.035 mg, 0.215 mg/0.035 mg, 0.25 mg/0.035 mg | |
Starting COCs in women not currently using hormonal contraception (Day 1 Start or Sunday Start) Important: Consider the possibility of ovulation and conception prior to initiation of this product. Tablet Color:
| Day 1 Start:
|
Sunday Start:
| |
Switching to Norgestimate and Ethinyl Estradiol Tablets, 0.25 mg/0.035 mg, or Norgestimate and Ethinyl Estradiol Tablets, 0.18 mg/0.035 mg, 0.215 mg/0.035 mg, 0.25 mg/0.035 mg from another oral contraceptive | Start on the same day that a new pack of the previous oral contraceptive would have started. |
Switching from another contraceptive method to Norgestimate and Ethinyl Estradiol Tablets, 0.25 mg/0.035 mg, or Norgestimate and Ethinyl Estradiol Tablets, 0.18 mg/0.035 mg, 0.215 mg/0.035 mg, 0.25 mg/0.035 mg | Start Norgestimate and Ethinyl Estradiol Tablets, 0.25 mg/0.035 mg, or Norgestimate and Ethinyl Estradiol Tablets, 0.18 mg/0.035 mg, 0.215 mg/0.035 mg, 0.25 mg/0.035 mg: |
|
|
|
|
|
|
|
|
|
|
Complete instructions to facilitate patient counseling on proper tablet usage are located in the FDA-Approved Patient Labeling. | |
Starting Norgestimate and Ethinyl Estradiol Tablets, 0.25 mg/0.035 mg, or Norgestimate and Ethinyl Estradiol Tablets, 0.18 mg/0.035 mg, 0.215 mg/0.035 mg, 0.25 mg/0.035 mg after Abortion or Miscarriage
First-trimester
- After a first-trimester abortion or miscarriage, Norgestimate and Ethinyl Estradiol Tablets, 0.25 mg/0.035 mg, or Norgestimate and Ethinyl Estradiol Tablets, 0.18 mg/0.035 mg, 0.215 mg/0.035 mg, 0.25 mg/0.035 mg may be started immediately. An additional method of contraception is not needed if Norgestimate and Ethinyl Estradiol Tablets, 0.25 mg/0.035 mg, or Norgestimate and Ethinyl Estradiol Tablets, 0.18 mg/0.035 mg, 0.215 mg/0.035 mg, 0.25 mg/0.035 mg are started immediately.
- If Norgestimate and Ethinyl Estradiol Tablets, 0.25 mg/0.035 mg, or Norgestimate and Ethinyl Estradiol Tablets, 0.18 mg/0.035 mg, 0.215 mg/0.035 mg, 0.25 mg/0.035 mg are not started within 5 days after termination of the pregnancy, the patient should use additional non-hormonal contraception (such as condoms and spermicide) for the first seven days of her first cycle pack of Norgestimate and Ethinyl Estradiol Tablets, 0.25 mg/0.035 mg, or Norgestimate and Ethinyl Estradiol Tablets, 0.18 mg/0.035 mg, 0.215 mg/0.035 mg, 0.25 mg/0.035 mg.
Second-trimester
- Do not start until 4 weeks after a second-trimester abortion or miscarriage, due to the increased risk of thromboembolic disease. Start Norgestimate and Ethinyl Estradiol Tablets, 0.25 mg/0.035 mg, or Norgestimate and Ethinyl Estradiol Tablets, 0.18 mg/0.035 mg, 0.215 mg/0.035 mg, 0.25 mg/0.035 mg, following the instructions in Table 1 for Day 1 or Sunday start, as desired. If using Sunday start, use additional non-hormonal contraception (such as condoms and spermicide) for the first seven days of the patient’s first cycle pack of Norgestimate and Ethinyl Estradiol Tablets, 0.25 mg/0.035 mg, or Norgestimate and Ethinyl Estradiol Tablets, 0.18 mg/0.035 mg, 0.215 mg/0.035 mg, 0.25 mg/0.035 mg. [see Contraindications (4 ) and Warnings and Precautions ( Error! Hyperlink reference not valid. )].
Starting Norgestimate and Ethinyl Estradiol Tablets, 0.25 mg/0.035 mg, or Norgestimate and Ethinyl Estradiol Tablets, 0.18 mg/0.035 mg, 0.215 mg/0.035 mg, 0.25 mg/0.035 mg after Childbirth
- Do not start until 4 weeks after delivery, due to the increased risk of thromboembolic disease. Start contraceptive therapy with Norgestimate and Ethinyl Estradiol Tablets, 0.25 mg/0.035 mg, or Norgestimate and Ethinyl Estradiol Tablets, 0.18 mg/0.035 mg, 0.215 mg/0.035 mg, 0.25 mg/0.035 mg following the instructions in Table 1 for women not currently using hormonal contraception.
- Norgestimate and Ethinyl Estradiol Tablets, 0.25 mg/0.035 mg, or Norgestimate and Ethinyl Estradiol Tablets, 0.18 mg/0.035 mg, 0.215 mg/0.035 mg, 0.25 mg/0.035 mg are not recommended for use in lactating women [see Use in Specific Populations ( Error! Hyperlink reference not valid. )].
- If the woman has not yet had a period postpartum, consider the possibility of ovulation and conception occurring prior to use of Norgestimate and Ethinyl Estradiol Tablets, 0.25 mg/0.035 mg, or Norgestimate and Ethinyl Estradiol Tablets, 0.18 mg/0.035 mg, 0.215 mg/0.035 mg, 0.25 mg/0.035 mg. [see Contraindications (4 ), Warnings and Precautions ( Error! Hyperlink reference not valid. ), and Use in Specific Populations (8.1 and Error! Hyperlink reference not valid. )].
Recommendations Regarding Missed Doses
Contraceptive failure may occur when active tablets are missed. Table 2 describes instructions for norgestimate and ethinyl estradiol tablets, 0.25 mg/0.035 mg, or norgestimate and ethinyl estradiol tablets, 0.18 mg/0.035 mg, 0.215 mg/0.035 mg, 0.25 mg/0.035 mg dosing and use of additional non-hormonal contraception (such as condoms) when active tablets are missed.
Table 2: Instructions for Missed Norgestimate and Ethinyl Estradiol Tablets, 0.25 mg/0.035 mg, or Norgestimate and Ethinyl Estradiol Tablets, 0.18 mg/0.035 mg, 0.215 mg/0.035 mg, 0.25 mg/0.035 mg | |
| Take the tablet as soon as possible. Continue taking one tablet a day until the pack is finished. |
| Take the two missed tablets as soon as possible and the next two active tablets the next day. Continue taking one tablet a day until the pack is finished. Additional non-hormonal contraception (such as condoms and spermicide) should be used as back-up if the patient has sex within 7 days after missing tablets. |
| Day 1 start: Throw out the rest of the pack and start a new pack that same day. Sunday start: Continue taking one tablet a day until Sunday, then throw out the rest of the pack and start a new pack that same day. Additional non-hormonal contraception (such as condoms and spermicide) should be used as back-up if the patient has sex within 7 days after missing tablets. |
Dosage Recommendations if Vomiting or Diarrhea Occurs
In case of severe vomiting or diarrhea, absorption may not be complete and additional contraceptive measures should be taken. If vomiting or diarrhea occurs within 3 to 4 hours after taking an active tablet, handle this as a missed tablet.
Norgestimate and Ethinyl Estradiol Tablets, 0.18 mg/0.035 mg, 0.215 mg/0.035 mg, 0.25 mg/0.035 mg Use for Acne
The timing of initiation of dosing with Norgestimate and Ethinyl Estradiol Tablets, 0.18 mg/0.035 mg, 0.215 mg/0.035 mg, 0.25 mg/0.035 mg for acne should follow the guidelines for use of Norgestimate and Ethinyl Estradiol Tablets, 0.18 mg/0.035 mg, 0.215 mg/0.035 mg, 0.25 mg/0.035 mg as an oral contraceptive. Consult the DOSAGE AND ADMINISTRATION section (2.1 ) for instructions.
DOSAGE FORMS AND STRENGTHS
Norgestimate and Ethinyl Estradiol Tablets, USP 0.25 mg/0.035 mg are available in blister packs containing 28 tablets, in the following order:
- 21 blue, round, flat faced beveled edged, uncoated tablets, debossed with ‘A7’ on one side and plain on the other. Each blue tablet contains 0.25 mg of norgestimate, USP and 0.035 mg of ethinyl estradiol, USP.
- 7 light green, round, flat faced beveled edged, uncoated tablets debossed with ‘A2’on one side and plain on the other. Each light green tablet contains inert ingredients.
Norgestimate and Ethinyl Estradiol Tablets, USP 0.18 mg/0.035 mg, 0.215 mg/0.035 mg, 0.25 mg/0.035 mg are available in blister cards. Each blister card contains 28 tablets in the following order:
- 7 white to off-white, round, flat faced beveled edge, uncoated tablets, debossed with ‘A9’ on one side and plain on the other. Each white to off-white tablet contains 0.18 mg of norgestimate, USP and 0.035 mg of ethinyl estradiol, USP.
- 7 light blue, round, flat faced beveled edge, uncoated tablets, debossed with ‘A8’ on one side and plain on the other. Each light blue tablet contains 0.215 mg norgestimate, USP and 0.035 mg of ethinyl estradiol, USP.
- 7 blue, round, flat faced beveled edge, uncoated tablets, debossed with ‘A7’ on one side, and plain on the other. Each blue tablet contains 0.25 mg of norgestimate, USP and 0.035 mg of ethinyl estradiol, USP.
- 7 light green, round, flat faced beveled edge, uncoated tablets, debossed with ‘A2’ on one side and plain on the other. Each light green tablet contains inert ingredients.
USE IN SPECIFIC POPULATIONS
Lactating women: Not recommended; can decrease milk production. (8.2)
Pregnancy
Risk Summary
There is no use for contraception in pregnancy, therefore, norgestimate and ethinyl estradiol tablets should be discontinued during pregnancy. Epidemiologic studies and meta-analyses have not found an increased risk of genital or non-genital birth defects (including cardiac anomalies and limb reduction defects) following exposure to CHCs before conception or during early pregnancy.
In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4 percent and 15 to 20 percent, respectively.
Lactation
Risk Summary
Contraceptive hormones and/or metabolites are present in human milk. CHCs can reduce milk production in breastfeeding females. This reduction can occur at any time but is less likely to occur once breastfeeding is well-established. When possible, advise the nursing female to use other forms of contraception until she discontinues breastfeeding. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for norgestimate and ethinyl estradiol tablets and any potential adverse effects on the breastfed child from norgestimate and ethinyl estradiol tablets or from the underlying maternal condition.
Pediatric Use
Safety and efficacy of norgestimate and ethinyl estradiol tablets have been established in women of reproductive age. Efficacy is expected to be the same for post-pubertal adolescents under the age of 18 and for users 18 years and older. Use of this product before menarche is not indicated.
There was no significant difference between norgestimate and ethinyl estradiol tablets, 0.18 mg/0.035 mg, 0.215 mg/0.035 mg, 0.25 mg/0.035 mg, and placebo in mean change in total lumbar spine (L1-L4) and total hip bone mineral density between baseline and Cycle 13 in 123 adolescent females with anorexia nervosa in a double-blind, placebo-controlled, multicenter, one-year treatment duration clinical trial for the Intent To Treat (ITT) population.
Geriatric Use
Norgestimate and ethinyl estradiol tablets have not been studied in postmenopausal women and are not indicated in this population.
Hepatic Impairment
The pharmacokinetics of norgestimate and ethinyl estradiol tablets have not been studied in subjects with hepatic impairment. However, steroid hormones may be poorly metabolized in patients with hepatic impairment. Acute or chronic disturbances of liver function may necessitate the discontinuation of COC use until markers of liver function return to normal and COC causation has been excluded [See Contraindications (4 ) and Warnings and Precautions (5.2 )].
Renal Impairment
The pharmacokinetics of norgestimate and ethinyl estradiol tablets have not been studied in women with renal impairment.
CONTRAINDICATIONS
Norgestimate and ethinyl estradiol tablets are contraindicated in females who are known to have or develop the following conditions:
- A high risk of arterial or venous thrombotic diseases. Examples include women who are known to:
- Smoke, if over age 35 [see Boxed Warning and Warnings and Precautions (5.1 )]
- Have deep vein thrombosis or pulmonary embolism, now or in the past [see Warnings and Precautions (5.1 )]
- Have inherited or acquired hyper coagulopathies [see Warnings and Precautions (5.1 )]
- Have cerebrovascular disease [see Warnings and Precautions (5.1 )]
- Have coronary artery disease [see Warnings and Precautions (5.1 )]
- Have thrombogenic valvular or thrombogenic rhythm diseases of the heart (for example, subacute bacterial endocarditis with valvular disease, or atrial fibrillation) [see Warnings and Precautions (5.1 )]
- Have uncontrolled hypertension [see Warnings and Precautions (5.4 )]
- Have diabetes mellitus with vascular disease [see Warnings and Precautions (5.6 )]
- Have headaches with focal neurological symptoms or migraine headaches with aura [see Warnings and Precautions (5.7 )]
- Women over age 35 with any migraine headaches [see Warnings and Precautions (5.7 )]
- Liver tumors, benign or malignant, or liver disease [see Warnings and Precautions (5.2 )]
- Undiagnosed abnormal uterine bleeding [see Warnings and Precautions (5.8 )]
- Current diagnosis of, or history of, breast cancer, which may be hormone-sensitive [see Warnings and Precautions (5.10 )]
- Use of Hepatitis C drug combinations containing ombitasvir/paritaprevir/ritonavir, with or without dasabuvir, due to the potential for ALT elevations [see Warnings and Precautions(5.3 )]
WARNINGS AND PRECAUTIONS
- Thromboembolic Disorders and Other Vascular Problems: Stop norgestimate and ethinyl estradiol tablets if a thrombotic event occurs. Stop at least 4 weeks before and through 2 weeks after major surgery. Start no earlier than 4 weeks after delivery, in women who are not breastfeeding. (5.1 )
- Liver disease: Discontinue norgestimate and ethinyl estradiol tablets if jaundice occurs. (5.2 )
- High blood pressure: If used in women with well-controlled hypertension, monitor blood pressure and stop norgestimate and ethinyl estradiol tablets if blood pressure rises significantly. (5.4 )
- Carbohydrate and lipid metabolic effects: Monitor prediabetic and diabetic women taking norgestimate and ethinyl estradiol tablets. Consider an alternate contraceptive method for women with uncontrolled dyslipidemia. (5.6 )
- Headache: Evaluate significant change in headaches and discontinue norgestimate and ethinyl estradiol tablets if indicated. (5.7 )
- Bleeding Irregularities and Amenorrhea: Evaluate irregular bleeding or amenorrhea. (5.8 )
Thromboembolic Disorders and Other Vascular Problems
- Stop norgestimate and ethinyl estradiol tablets if an arterial thrombotic event or venous thromboembolic (VTE) event occurs.
- Stop norgestimate and ethinyl estradiol tablets if there is unexplained loss of vision, proptosis, diplopia, papilledema, or retinal vascular lesions. Evaluate for retinal vein thrombosis immediately [see Adverse Reactions (6.2 )] .
- If feasible, stop norgestimate and ethinyl estradiol tablets at least 4 weeks before and through 2 weeks after major surgery or other surgeries known to have an elevated risk of VTE as well as during and following prolonged immobilization.
- Start norgestimate and ethinyl estradiol tablets no earlier than 4 weeks after delivery, in women who are not breastfeeding. The risk of postpartum VTE decreases after the third postpartum week, whereas the risk of ovulation increases after the third postpartum week.
- The use of COCs increases the risk of VTE. However, pregnancy increases the risk of VTE as much or more than the use of COCs. The risk of VTE in women using COCs is 3 to 9 cases per 10,000 woman-years. The risk of VTE is highest during the first year of use of COCs and when restarting hormonal contraception after a break of 4 weeks or longer. The risk of thromboembolic disease due to COCs gradually disappears after use is discontinued.
- Use of COCs also increases the risk of arterial thromboses such as strokes and myocardial infarctions, especially in women with other risk factors for these events. COCs have been shown to increase both the relative and attributable risks of cerebrovascular events (thrombotic and hemorrhagic strokes). This risk increases with age, particularly in women over 35 years of age who smoke.
- Use COCs with caution in women with cardiovascular disease risk factors.
Liver Disease
Impaired Liver Function
Norgestimate and ethinyl estradiol tablets are contraindicated in women with liver disease, such as acute viral hepatitis or severe (decompensated) cirrhosis of liver [see Contraindications (4 )]. Acute or chronic disturbances of liver function may necessitate the discontinuation of COC use until markers of liver function return to normal and COC causation has been excluded. Discontinue norgestimate and ethinyl estradiol tablets if jaundice develops.
Liver Tumors
Norgestimate and ethinyl estradiol tablets are contraindicated in women with benign and malignant liver tumors [see Contraindications (4 )]. Hepatic adenomas are associated with COC use. An estimate of the attributable risk is 3.3 cases/100,000 COC users. Rupture of hepatic adenomas may cause death through intra-abdominal hemorrhage.
Studies have shown an increased risk of developing hepatocellular carcinoma in long-term (>8 years) COC users. However, the risk of liver cancers in COC users is less than one case per million users.
Risk of Liver Enzyme Elevations with Concomitant Hepatitis C Treatment
During clinical trials with the Hepatitis C combination drug regimen that contains ombitasvir/paritaprevir/ritonavir, with or without dasabuvir, ALT elevations greater than 5 times the upper limit of normal (ULN), including some cases greater than 20 times the ULN, were significantly more frequent in women using ethinyl estradiol-containing medications, such as COCs. Discontinue norgestimate and ethinyl estradiol tablets prior to starting therapy with the combination drug regimen ombitasvir/paritaprevir/ritonavir, with or without dasabuvir [see Contraindications (4 )]. Norgestimate and ethinyl estradiol tablets can be restarted approximately 2 weeks following completion of treatment with the Hepatitis C combination drug regimen.
High Blood Pressure
Norgestimate and ethinyl estradiol tablets are contraindicated in women with uncontrolled hypertension or hypertension with vascular disease [see Contraindications (4 )]. For women with well-controlled hypertension, monitor blood pressure and stop norgestimate and ethinyl estradiol tablets if blood pressure rises significantly.
An increase in blood pressure has been reported in women taking COCs, and this increase is more likely in older women with extended duration of use. The incidence of hypertension increases with increasing concentrations of progestin.
Gallbladder Disease
Studies suggest a small increased relative risk of developing gallbladder disease among COC users. Use of COCs may worsen existing gallbladder disease. A past history of COC-related cholestasis predicts an increased risk with subsequent COC use. Women with a history of pregnancy-related cholestasis may be at an increased risk for COC related cholestasis.
Carbohydrate and Lipid Metabolic Effects
Carefully monitor prediabetic and diabetic women who take norgestimate and ethinyl estradiol tablets. COCs may decrease glucose tolerance.
Consider alternative contraception for women with uncontrolled dyslipidemia. A small proportion of women will have adverse lipid changes while on COCs.
Women with hypertriglyceridemia, or a family history thereof, may be at an increased risk of pancreatitis when using COCs.
Headache
If a woman taking norgestimate and ethinyl estradiol tablets develops new headaches that are recurrent, persistent, or severe, evaluate the cause and discontinue norgestimate and ethinyl estradiol tablets if indicated.
Consider discontinuation of norgestimate and ethinyl estradiol tablets in the case of increased frequency or severity of migraine during COC use (which may be prodromal of a cerebrovascular event).
Bleeding Irregularities and Amenorrhea
Unscheduled Bleeding and Spotting
Unscheduled (breakthrough or intracyclic) bleeding and spotting sometimes occur in patients on COCs, especially during the first three months of use. If bleeding persists or occurs after previously regular cycles, check for causes such as pregnancy or malignancy. If pathology and pregnancy are excluded, bleeding irregularities may resolve over time or with a change to a different contraceptive product.
In clinical trials of norgestimate and ethinyl estradiol tablets, 0.25 mg/0.035 mg, and norgestimate and ethinyl estradiol tablets, 0.18 mg/0.035 mg, 0.215 mg/0.035 mg, 0.25 mg/0.035 mg, the frequency and duration of breakthrough bleeding and/or spotting was assessed in 1,647 patients (21,275 evaluable cycles) and 4,826 patients (35,546 evaluable cycles), respectively. A total of 100 (7.5%) women discontinued norgestimate and ethinyl estradiol tablets, 0.25 mg/0.035 mg and 231 (4.8%) women discontinued norgestimate and ethinyl estradiol tablets, 0.18 mg/0.035 mg, 0.215 mg/0.035 mg, 0.25 mg/0.035 mg, at least in part, due to bleeding or spotting. Based on data from the clinical trials, 14 to 34% of women using norgestimate and ethinyl estradiol tablets, 0.25 mg/0.035 mg, experienced unscheduled bleeding per cycle in the first year; for norgestimate and ethinyl estradiol tablets, 0.18 mg/0.035 mg, 0.215 mg/0.035 mg, 0.25 mg/0.035 mg, the respective numbers were 13 to 38%. The percent of women who experienced breakthrough/unscheduled bleeding tended to decrease over time.
Amenorrhea and Oligomenorrhea
Women who use norgestimate and ethinyl estradiol tablets may experience amenorrhea. Some women may experience amenorrhea or oligomenorrhea after discontinuation of COCs, especially when such a condition was pre-existent.
If scheduled (withdrawal) bleeding does not occur, consider the possibility of pregnancy. If the patient has not adhered to the prescribed dosing schedule (missed one or more active tablets or started taking them on a day later than she should have), consider the possibility of pregnancy at the time of the first missed period and take appropriate diagnostic measures. If the patient has adhered to the prescribed regimen and misses two consecutive periods, rule out pregnancy.
Depression
Carefully observe women with a history of depression and discontinue norgestimate and ethinyl estradiol tablets if depression recurs to a serious degree.
Malignant Neoplasms
Breast Cancer
Norgestimate and ethinyl estradiol is contraindicated in females who currently have or have had breast cancer because breast cancer may be hormonally sensitive [see Contraindications (4 )] .
Epidemiology studies have not found a consistent association between use of combined oral contraceptives (COCs) and breast cancer risk. Studies do not show an association between ever (current or past) use of COCs and risk of breast cancer. However, some studies report a small increase in the risk of breast cancer among current or recent users (<6 months since last use) and current users with longer duration of COC use [see Postmarketing Experience (6.2 )] .
- Cervical Cancer
- Some studies suggest that COC use has been associated with an increase in the risk of cervical cancer or intraepithelial neoplasia. However, there continues to be controversy about the extent to which such findings may be due to differences in sexual behavior and other factors.
Effect on Binding Globulins
The estrogen component of COCs may raise the serum concentrations of thyroxine-binding globulin, sex hormone-binding globulin, and cortisol-binding globulin. The dose of replacement thyroid hormone or cortisol therapy may need to be increased.
Monitoring
A woman who is taking COCs should have a yearly visit with her healthcare provider for a blood pressure check and for other indicated healthcare.
Hereditary Angioedema
In women with hereditary angioedema, exogenous estrogens may induce or exacerbate symptoms of angioedema.
Chloasma
Chloasma may occasionally occur, especially in women with a history of chloasma gravidarum. Women with a tendency to chloasma should avoid exposure to the sun or ultraviolet radiation while taking norgestimate and ethinyl estradiol tablets.
ADVERSE REACTIONS
The following serious adverse reactions with the use of COCs are discussed elsewhere in labeling:
Clinical Trial Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
Norgestimate and Ethinyl Estradiol Tablets, 0.25 mg/0.035 mg
The safety of norgestimate and ethinyl estradiol tablets, 0.25 mg/0.035 mg, was evaluated in 1,647 healthy women of child-bearing potential who participated in 3 clinical trials and received at least 1 dose of norgestimate and ethinyl estradiol tablets, 0.25 mg/0.035 mg, for contraception. Two trials were randomized active-controlled trials and 1 was an uncontrolled open-label trial. In all 3 trials, subjects were followed for up to 24 cycles.
Common Adverse Reactions (≥ 2% of subjects): The most common adverse reactions reported by at least 2% of the 1,647 women were the following in order of decreasing incidence: headache/migraine (32.9%), abdominal/gastrointestinal pain (7.8%), vaginal infection (8.4%), genital discharge (6.8%), breast issues (including breast pain, discharge, and enlargement) (6.3%), mood disorders (including depression and mood altered) (5.0%), flatulence (3.2%), nervousness (2.9%), and rash (2.6%).
Adverse Reactions Leading to Study Discontinuation: Over the three trials, between 11 to 21% of subjects discontinued the trial due to an adverse reaction. The most common adverse reactions (≥1%) leading to discontinuation were: metrorrhagia (6.9%), nausea/vomiting (5.0%), headache (4.1%), mood disorders (including depression and mood altered) (2.4%), premenstrual syndrome (1.7%), hypertension (1.4%), breast pain (1.4%), nervousness (1.3%), amenorrhea (1.1%), dysmenorrhea (1.1%), weight increased (1.1%), and flatulence (1.1%).
Serious Adverse Reactions: breast cancer (1 subject), mood disorders including depression, irritability, and mood swings (1 subject), myocardial infarction (1 subject), and venous thromboembolic events including pulmonary embolism (1 subject) and deep vein thrombosis (DVT) (1 subject).
Norgestimate and Ethinyl Estradiol Tablets, 0.18 mg/0.035 mg, 0.215 mg/0.035 mg, 0.25 mg/0.035 mg
The safety of norgestimate and ethinyl estradiol tablets, 0.18 mg/0.035 mg, 0.215 mg/0.035 mg, 0.25 mg/0.035 mg, was evaluated in 4,826 healthy women of child-bearing potential who participated in 6 clinical trials and received at least 1 dose of norgestimate and ethinyl estradiol tablets, 0.18 mg/0.035 mg, 0.215 mg/0.035 mg, 0.25 mg/0.035 mg, for contraception. Two trials were randomized active-controlled trials and 4 were uncontrolled open-label trials. In 3 trials, subjects were followed for up to 24 cycles; in 2 trials, subjects were followed for up to 12 cycles; and in 1 trial, subjects were followed for up to 6 cycles.
Common Adverse Reactions (≥ 2% of subjects): The most common adverse reactions reported by at least 2% of the 4,826 women were the following in order of decreasing incidence: headache/migraine (33.6%), breast issues (including breast pain, enlargement, and discharge) (8.0%), vaginal infection (7.1%), abdominal/gastrointestinal pain (5.6%), mood disorders (including mood alteration and depression) (3.8%), genital discharge (3.2%), and changes in weight (including weight fluctuation, increased or decreased) (2.5%).
Adverse Reactions Leading to Study Discontinuation: Over the trials, between 9 to 27% of subjects discontinued the trial due to an adverse reaction. The most common adverse reactions (≥1%) leading to discontinuation were: metrorrhagia (4.3%), nausea/vomiting (2.8%), headache/migraine (2.4%), mood disorders (including depression and mood altered) (1.1%), and weight increased (1.1%).
Serious Adverse Reactions: breast cancer (1 subject), carcinoma of the cervix in situ (1 subject), hypertension (1 subject), and migraine (2 subjects).
Postmarketing Experience
Five studies that compared breast cancer risk between ever-users (current or past use) of COCs and never-users of COCs reported no association between ever use of COCs and breast cancer risk, with effect estimates ranging from 0.90 to 1.12 (Figure 1).
Three studies compared breast cancer risk between current or recent COC users (<6 months since last use) and never users of COCs (Figure 1). One of these studies reported no association between breast cancer risk and COC use. The other two studies found an increased relative risk of 1.19 to 1.33 with current or recent use. Both of these studies found an increased risk of breast cancer with current use of longer duration, with relative risks ranging from 1.03 with less than one year of COC use to approximately 1.4 with more than 8 to 10 years of COC use.
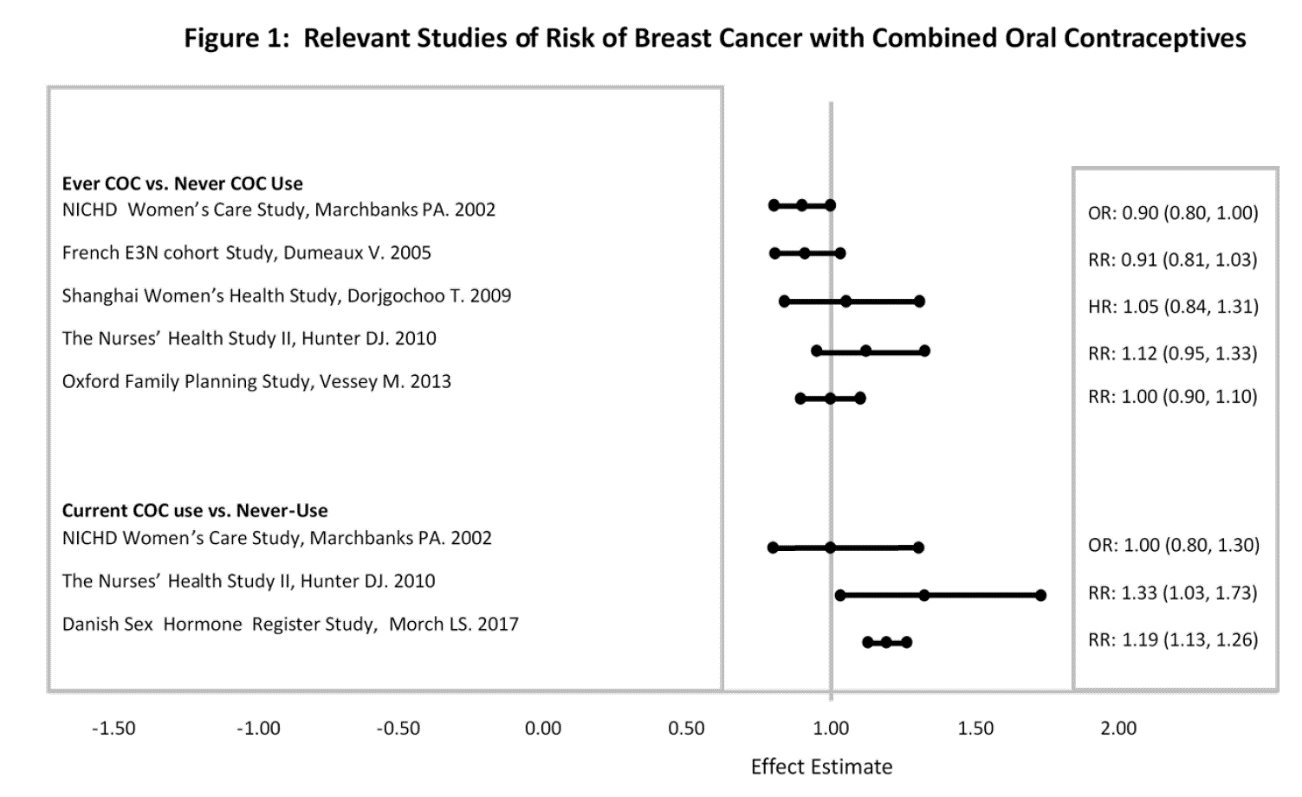
RR = relative risk; OR = odds ratio; HR = hazard ratio. “ ever COC ” are females with current or past COC use; “ never COC use ” are females that never used COCs.
The following additional adverse reactions have been reported from worldwide postmarketing experience with norgestimate/ethinyl estradiol. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Infections and Infestations : Urinary tract infection;
Neoplasms Benign, Malignant and Unspecified (Incl. Cysts and Polyps) : Breast cancer, benign breast neoplasm, hepatic adenoma, focal nodular hyperplasia, breast cyst;
Immune System Disorders : Anaphylactic reaction, hypersensitivity;
Metabolism and Nutrition Disorders : Dyslipidemia;
Psychiatric Disorders : Anxiety, insomnia;
Nervous System Disorders :Syncope, convulsion, paresthesia, dizziness;
Eye Disorders :Visual impairment, dry eye, contact lens intolerance;
Ear and Labyrinth Disorders :Vertigo;
Cardiac Disorders : Tachycardia, palpitations;
Vascular Events :Deep vein thrombosis, pulmonary embolism, retinal vascular thrombosis, hot flush, venous thrombosis (including Budd Chiari Syndrome and hepatic vein thrombosis);
Arterial Events :Arterial thromboembolism, myocardial infarction, cerebrovascular accident;
Respiratory, Thoracic and Mediastinal Disorders :Dyspnea;
Gastrointestinal Disorders : Pancreatitis, abdominal distension, diarrhea, constipation;
Hepatobiliary Disorders : Hepatitis;
Skin and Subcutaneous Tissue Disorders : Angioedema, erythema nodosum, hirsutism, night sweats, hyperhidrosis, photosensitivity reaction, urticaria, pruritus, acne;
Musculoskeletal, Connective Tissue, and Bone Disorders :Muscle spasms, pain in extremity, myalgia, back pain;
Reproductive System and Breast Disorders :Ovarian cyst, suppressed lactation, vulvovaginal dryness;
General Disorders and Administration Site Conditions : Chest pain, asthenic conditions.
DRUG INTERACTIONS
Consult the labeling of concurrently used drugs to obtain further information about interactions with hormonal contraceptives or the potential for enzyme alterations.
No drug-drug interaction studies were conducted with norgestimate and ethinyl estradiol tablets.
Effects of Other Drugs on Combined Oral Contraceptives
Substances decreasing the plasma concentrations of COCs
Drugs or herbal products that induce certain enzymes, including cytochrome P450 3A4 (CYP3A4), may decrease the plasma concentrations of COCs and potentially diminish the effectiveness of COCs or increase breakthrough bleeding. Some drugs or herbal products that may decrease the effectiveness of hormonal contraceptives include phenytoin, barbiturates, carbamazepine, bosentan, felbamate, griseofulvin, oxcarbazepine, rifampicin, topiramate, rifabutin, rufinamide, aprepitant, and products containing St. John’s wort. Interactions between hormonal contraceptives and other drugs may lead to breakthrough bleeding and/or contraceptive failure. Counsel women to use an alternative method of contraception or a back-up method when enzyme inducers are used with COCs, and to continue back-up contraception for 28 days after discontinuing the enzyme inducer to ensure contraceptive reliability.
Colesevelam: Colesevelam, a bile acid sequestrant, given together with a COC, has been shown to significantly decrease the AUC of EE. The drug interaction between the contraceptive and colesevelam was decreased when the two drug products were given 4 hours apart.
Substances increasing the plasma concentrations of COCs:
Co-administration of atorvastatin or rosuvastatin and certain COCs containing ethinyl estradiol (EE) increase AUC values for EE by approximately 20 to 25%. Ascorbic acid and acetaminophen may increase plasma EE concentrations, possibly by inhibition of conjugation. CYP3A4 inhibitors such as itraconazole, voriconazole, fluconazole, grapefruit juice, or ketoconazole may increase plasma hormone concentrations.
Human immunodeficiency virus (HIV)/Hepatitis C virus (HCV) protease inhibitors, non-nucleoside reverse transcriptase inhibitors, and HIV/AIDS medications containing strong inhibitors or inducers of CYP3A
Significant changes (increase or decrease) in the plasma concentrations of estrogen and/or progestin have been noted in some cases of co-administration with HIV protease inhibitors (decrease [e.g., nelfinavir, ritonavir, darunavir/ritonavir, (fos)amprenavir/ritonavir, lopinavir/ritonavir, and tipranavir/ritonavir] or increase [e.g., indinavir and atazanavir/ritonavir])/HCV protease inhibitors (decrease [e.g., boceprevir and telaprevir]) or with non-nucleoside reverse transcriptase inhibitors (decrease [e.g., nevirapine] or increase [e.g., etravirine]) or with HIV/AIDS medications containing strong inhibitors (e.g., cobicistat and ritonavir) or inducers of CYP3A.
Effects of Combined Oral Contraceptives on Other Drugs
- COCs containing EE may inhibit the metabolism of other compounds (e.g., cyclosporine, prednisolone, theophylline, tizanidine, and voriconazole) and increase their plasma concentrations.
- COCs have been shown to decrease plasma concentrations of acetaminophen, clofibric acid, morphine, salicylic acid, temazepam and lamotrigine. Significant decrease in plasma concentration of lamotrigine has been shown, likely due to induction of lamotrigine glucuronidation. This may reduce seizure control; therefore, dosage adjustments of lamotrigine may be necessary. Women on thyroid hormone replacement therapy may need increased doses of thyroid hormone because the serum concentration of thyroid-binding globulin increases with use of COCs.
Interference with Laboratory Tests
The use of contraceptive steroids may influence the results of certain laboratory tests, such as coagulation factors, lipids, glucose tolerance, and binding proteins.
Concomitant Use with HCV Combination Therapy – Liver Enzyme Elevation
Do not co-administer norgestimate and ethinyl estradiol tablets with HCV drug combinations containing ombitasvir/paritaprevir/ritonavir, with or without dasabuvir, due to potential for ALT elevations [see Warnings and Precautions (5.3 )].
DESCRIPTION
Each of the following products is a combination oral contraceptive containing the progestational compound norgestimate, USP and the estrogenic compound ethinyl estradiol, USP. Norgestimate, USP is designated as (18,19-Dinor-17-pregn-4-en-20-yn-3-one,17-(acetyloxy)-13-ethyl-,oxime,(17α)-(+)-) and ethinyl estradiol, USP is designated as (19-nor-17α-pregna,1,3,5(10)-trien-20-yne-3,17-diol).
Norgestimate and Ethinyl Estradiol Tablets, USP 0.25 mg/0.035 mg
- Each blue tablet contains 0.25 mg of norgestimate, USP and 0.035 mg of ethinyl estradiol, USP. Inactive ingredients include lactose monohydrate, povidone, FD&C Blue No. 2, colloidal silicon dioxide, talc, magnesium stearate, and pregelatinized starch.
- Each light green tablet contains only inert ingredients, as follows: D&C Yellow No. 10, FD&C Blue No. 2, lactose monohydrate, magnesium stearate, microcrystalline cellulose, pregelatinized starch and talc.
Norgestimate and Ethinyl Estradiol Tablets, USP 0.18 mg/0.035 mg, 0.215 mg/0.035 mg, 0.25 mg/0.035 mg
- Each white to off-white tablet contains 0.18 mg of norgestimate, USP and 0.035 mg of ethinyl estradiol, USP. Inactive ingredients include lactose monohydrate, povidone, magnesium stearate, colloidal silicon dioxide, talc and pregelatinized starch.
- Each light blue tablet contains 0.215 mg of norgestimate, USP and 0.035 mg of ethinyl estradiol, USP. Inactive ingredients include FD & C Blue No. 2, lactose monohydrate, povidone, magnesium stearate, colloidal silicon dioxide, talc and pregelatinized starch.
- Each blue tablet contains 0.25 mg of norgestimate, USP and 0.035 mg of ethinyl estradiol, USP. Inactive ingredients include FD & C Blue No. 2, lactose monohydrate, povidone, magnesium stearate, colloidal silicon dioxide, talc and pregelatinized starch.
- Each light green tablet contains only inert ingredients, as follows: D&C Yellow No. 10, FD&C Blue No. 2, lactose monohydrate, magnesium stearate, microcrystalline cellulose, pregelatinized starch and talc.
CLINICAL PHARMACOLOGY
Mechanism of Action
- Oral Contraception COCs lower the risk of becoming pregnant primarily by suppressing ovulation.
- Acne Acne is a skin condition with a multifactorial etiology, including androgen stimulation of sebum production. While the combination of ethinyl estradiol and norgestimate increases sex hormone-binding globulin (SHBG) and decreases free testosterone, the relationship between these changes and a decrease in the severity of facial acne in otherwise healthy women with this skin condition has not been established.
Pharmacodynamics
No specific pharmacodynamic studies were conducted with norgestimate and ethinyl estradiol tablets.
Pharmacokinetics
Absorption
Norgestimate (NGM) and EE are rapidly absorbed following oral administration. NGM is rapidly and completely metabolized by first pass (intestinal and/or hepatic) mechanisms to norelgestromin (NGMN) and norgestrel (NG), which are the major active metabolites of norgestimate.
Peak serum concentrations of NGMN and EE are generally reached by 2 hours after administration of norgestimate and ethinyl estradiol tablets. Accumulation following multiple dosing of the 250 mcg NGM / 35 mcg EE dose is approximately 2-fold for NGMN and EE compared with single dose administration. The pharmacokinetics of NGMN is dose-proportional following NGM doses of 180 mcg to 250 mcg. Steady-state concentration of EE is achieved by Day 7 of each dosing cycle. Steady-state concentrations of NGMN and NG are achieved by Day 21. Non-linear accumulation (approximately 8 fold) of NG is observed as a result of high-affinity binding to SHBG, which limits its biological activity (Table 3).
Table 3: Summary of NGMN, NG and EE pharmacokinetic parameters.
Mean (SD) Pharmacokinetic Parameters of Norgestimate and Ethinyl Estradiol Tablets, 0.18 mg/0.035 mg, 0.215 mg/0.035 mg, 0.25 mg/0.035 mg During a Three Cycle Study | ||||||
Analyte | Cycle | Day | C max | t max (h) | AUC 0-24h | t 1/2 (h) |
NGMN | 3 | 7 | 1.80 (0.46) | 1.42 (0.73) | 15 (3.88) | NC |
14 | 2.12 (0.56) | 1.21 (0.26) | 16.1 (4.97) | NC | ||
21 | 2.66 (0.47) | 1.29 (0.26) | 21.4 (3.46) | 22.3 (6.54) | ||
NG | 3 | 7 | 1.94 (0.82) | 3.15 (4.05) | 34.8 (16.5) | NC |
14 | 3 (1.04) | 2.21 (2.03) | 55.2 (23.5) | NC | ||
21 | 3.66 (1.15) | 2.58 (2.97) | 69.3 (23.8) | 40.2 (15.4) | ||
EE | 3 | 7 | 124 (39.5) | 1.27 (0.26) | 1130 (420) | NC |
14 | 128 (38.4) | 1.32 (0.25) | 1130 (324) | NC | ||
21 | 126 (34.7) | 1.31 (0.56) | 1090 (359) | 15.9 (4.39) | ||
Mean (SD) Pharmacokinetic Parameters of Norgestimate and Ethinyl Estradiol Tablets, 0.25 mg/0.035 mg During a Three Cycle Study | ||||||
Analyte | Cycle | Day | C max | t max (h) | AUC 0-24h | t 1/2 (h) |
NGMN | 1 | 1 | 1.78 (0.397) | 1.19 (0.25) | 9.90 (3.25) | 18.4 (5.91) |
3 | 21 | 2.19 (0.655) | 1.43 (0.68) | 18.1 (5.53) | 24.9 (9.04) | |
NG | 1 | 1 | 0.649 (0.49) | 1.42 (0.69) | 6.22 (2.46) | 37.8 (14) |
3 | 21 | 2.65 (1.11) | 1.67 (1.32) | 48.2 (20.5) | 45 (20.4) | |
EE | 1 | 1 | 92.2 (24.5) | 1.2 (0.26) | 629 (138) | 10.1 (1.9) |
3 | 21 | 147 (41.5) | 1.13 (0.23) | 1210 (294) | 15 (2.36) | |
C max = peak serum concentration, t max = time to reach peak serum concentration, AUC 0-24h = area under serum concentration vs time curve from 0 to 24 hours, t 1/2 = elimination half-life, NC = not calculated. NGMN and NG: C max = ng/mL, AUC 0-24h =h•ng/mL EE: C max =pg/mL, AUC 0-24h =h•pg/mL
Food Effect
The effect of food on the pharmacokinetics of norgestimate and ethinyl estradiol tablets has not been studied.
Distribution
NGMN and NG are highly bound (>97%) to serum proteins. NGMN is bound to albumin and not to SHBG, while NG is bound primarily to SHBG. EE is extensively bound (>97%) to serum albumin and induces an increase in the serum concentrations of SHBG.
Metabolism
NGM is extensively metabolized by first-pass mechanisms in the gastrointestinal tract and/or liver. NGM’s primary active metabolite is NGMN. Subsequent hepatic metabolism of NGMN occurs and metabolites include NG, which is also active, and various hydroxylated and conjugated metabolites. Although NGMN and its metabolites inhibit a variety of P450 enzymes in human liver microsomes, under the recommended dosing regimen, the in vivo concentrations of NGMN and its metabolites, even at the peak serum levels, are relatively low compared to the inhibitory constant (K i ). EE is also metabolized to various hydroxylated products and their glucuronide and sulfate conjugates.
Excretion
The metabolites of NGMN and EE are eliminated by renal and fecal pathways. Following administration of 14 C-norgestimate, 47% (45 to 49%) and 37% (16 to 49%) of the administered radioactivity was eliminated in the urine and feces, respectively. Unchanged NGM was not detected in the urine. In addition to 17-deacetyl norgestimate, a number of metabolites of NGM have been identified in human urine following administration of radiolabeled NGM. These include 18, 19-Dinor-17-pregn-4-en-20-yn-3-one, 17-hydroxy-13-ethyl,(17α)-(-);18,19-Dinor-5β-17-pregnan-20-yn,3α,17β-dihydroxy-13-ethyl,(17α), various hydroxylated metabolites and conjugates of these metabolites.
CLINICAL STUDIES
Contraception
In three US clinical trials with norgestimate and ethinyl estradiol tablets, 0.25 mg/0.035 mg, 1,651 women aged 18 to 38 years were studied for up to 24 cycles, proving a total of 24,272 cycles of exposure. The racial demographic was about 73 to 86% Caucasian, 8 to 13% African-American, 6 to 14% Hispanic with the remainder Asian or Other (≤1%). There were no exclusions on the basis of weight; the weight range for women treated was 82 to 303 lbs, with a mean weight of about 135 lbs. The pregnancy rate was approximately 1 pregnancy per 100 women-years.
In four clinical trials with norgestimate and ethinyl estradiol tablets, 0.18 mg/0.035 mg, 0.215 mg/0.035 mg, 0.25 mg/0.035 mg, 4,756 women aged 15 to 41 years were studied for 24 cycles, providing a total of 45,244 cycles of exposure. The racial demographic was about 87 to 90% Caucasian, 6 to 10% African-American, with the remainder Asian (≤1%) or Other (2 to 5%). There were no exclusions on the basis of weight; the weight range for women treated was 80 to 310 lbs, with a mean weight of about 132 lbs. The pregnancy rate was approximately 1 pregnancy per 100 women-years.
Acne
Norgestimate and ethinyl estradiol tablets, 0.18 mg/0.035 mg, 0.215 mg/0.035 mg, 0.25 mg/0.035 mg, was evaluated for the treatment of acne vulgaris in two randomized, double-blind, placebo-controlled, multicenter, six- (28 day) cycle studies. Two hundred twenty-one patients received norgestimate and ethinyl estradiol tablets, 0.18 mg/0.035 mg, 0.215 mg/0.035 mg, 0.25 mg/0.035 mg, and 234 patients received placebo. Mean age at enrollment for both groups was 28 years. At the end of 6 months, the mean total lesion count changed from 55 to 31 (42% reduction) in patients treated with norgestimate and ethinyl estradiol tablets, 0.18 mg/0.035 mg, 0.215 mg/0.035 mg, 0.25 mg/0.035 mg, and from 54 to 38 (27% reduction) in patients similarly treated with placebo. Table 4 summarizes the changes in lesion count for each type of lesion. Based on the investigator’s global assessment conducted at the final visit, patients treated with norgestimate and ethinyl estradiol tablets, 0.18 mg/0.035 mg, 0.215 mg/0.035 mg, 0.25 mg/0.035 mg, showed a statistically significant improvement in total lesions compared to those treated with placebo.
Table 4: Acne Vulgaris Indication. Combined Results: Two Multicenter, Placebo-Controlled Trials. Observed Means at Six Months (LOCF) • and at Baseline. Intent-to-Treat Population.
| Norgestimate and Ethinyl Estradiol Tablets, 0.18 mg/0.035 mg, 0.215 mg/0.035 mg, 0.25 mg/0.035 mg (N=221) | Placebo (N=234) | Difference in Counts between Norgestimate and Ethinyl Estradiol Tablets, 0.18 mg/0.035 mg, 0.215 mg/0.035 mg, 0.25 mg/0.035 mg and Placebo at 6 Months | |||
|---|---|---|---|---|---|
| # of Lesions | Counts | % Reduction | Counts | % Reduction | |
INFLAMMATORY LESIONS | |||||
Baseline Mean | 19 | 19 | |||
Sixth Month Mean | 10 | 48% | 13 | 30% | 3 (95% CI: -1.2, 5.1) |
NON-INFLAMMATORY LESIONS | |||||
Baseline Mean | 36 | 35 | |||
Sixth Month Mean | 22 | 34% | 25 | 21% | 3 (95% CI: -0.2, 7.8) |
TOTAL LESIONS | |||||
Baseline Mean | 55 | 54 | 7 (95% CI: 2, 11.9) | ||
Sixth Month Mean | 31 | 42% | 38 | 27% | |
• LOCF: Last Observation Carried Forward
HOW SUPPLIED/STORAGE AND HANDLING
How Supplied
Norgestimate and Ethinyl Estradiol Tablets, USP 0.25 mg/0.035 mg are available in blister packs containing 28 tablets, as follows:
- 21 blue, round, flat faced beveled edged, uncoated tablets, debossed with ‘A7’ on one side and plain on the other. Each blue tablet contains 0.25 mg of norgestimate, USP and 0.035 mg of ethinyl estradiol, USP.
- 7 light green, round, flat faced beveled edged, uncoated tablets debossed with ‘A2’on one side and plain on the other. Each light green tablet contains inert ingredients.
NDC 68462-309-29 1 carton containing 3 blister cards of 28 tablets
Norgestimate and Ethinyl Estradiol Tablets, USP 0.18 mg/0.035 mg, 0.215 mg/0.035 mg, 0.25 mg/0.035 mg are available in blister packs containing 28 tablets, as follows:
- 7 white to off-white, round, flat faced beveled edge, uncoated tablets, debossed with ‘A9’ on one side and plain on the other. Each white to off-white tablet contains 0.18 mg of norgestimate, USP and 0.035 mg of ethinyl estradiol, USP.
- 7 light blue, round, flat faced beveled edge, uncoated tablets, debossed with ‘A8’ on one side and plain on the other. Each light blue tablet contains 0.215 mg norgestimate, USP and 0.035 mg of ethinyl estradiol, USP.
- 7 blue, round, flat faced beveled edge, uncoated tablets, debossed with ‘A7’ on one side, and plain on the other. Each blue tablet contains 0.25 mg of norgestimate, USP and 0.035 mg of ethinyl estradiol, USP.
- 7 light green, round, flat faced beveled edge, uncoated tablets, debossed with ‘A2’ on one side and plain on the other. Each light green tablet contains inert ingredients.
NDC 68462-565-29 1 carton containing 3 blister cards of 28 tablets
Storage Conditions
- Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
- Protect from light.
- Keep out of the reach of children.
Mechanism of Action
- Oral Contraception COCs lower the risk of becoming pregnant primarily by suppressing ovulation.
- Acne Acne is a skin condition with a multifactorial etiology, including androgen stimulation of sebum production. While the combination of ethinyl estradiol and norgestimate increases sex hormone-binding globulin (SHBG) and decreases free testosterone, the relationship between these changes and a decrease in the severity of facial acne in otherwise healthy women with this skin condition has not been established.