Get your patient on Nystatin - Nystatin tablet, Film Coated (Nystatin)
Nystatin - Nystatin tablet, Film Coated prescribing information
INDICATIONS AND USAGE
Nystatin Tablets are intended for the treatment of non-esophageal mucus membrane gastrointestinal candidiasis.
DOSAGE AND ADMINISTRATION
The usual therapeutic dosage is one to two tablets (500,000 to 1,000,000 units nystatin) three times daily. Treatment should generally be continued for at least 48 hours after clinical cure to prevent relapse.
CONTRAINDICATIONS
Nystatin tablets are contraindicated in patients with a history of hypersensitivity to any of their components.
ADVERSE REACTIONS
Nystatin is well tolerated even with prolonged therapy. Oral irritation and sensitization have been reported. (See PRECAUTIONS: General. )
Gastrointestinal
Diarrhea (including one case of bloody diarrhea), nausea, vomiting, gastrointestinal upset/disturbances.
Dermatologic
Rash, including urticaria has been reported rarely. Stevens-Johnson syndrome has been reported very rarely.
Other
Tachycardia, bronchospasm, facial swelling, and nonspecific myalgia have also been rarely reported.
To report SUSPECTED ADVERSE REACTIONS, contact Chartwell RX, LLC. at 1-845-232-1683 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DESCRIPTION
Nystatin, USP is an antimycotic polyene antibiotic obtained from Streptomyces noursei . Its structural formula:
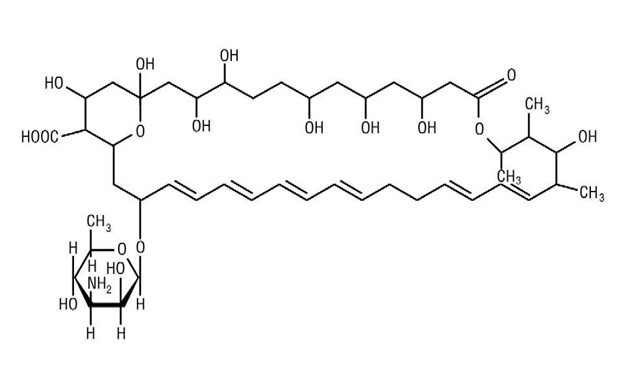
C 47 H 75 NO 17 M.W. 926.13
Nystatin tablets, USP are for oral administration and contain 500,000 units of nystatin, USP per tablet. Nystatin tablets, USP contain the inactive ingredients: corn starch, confectioner sugar, dibasic calcium phosphate, FD&C yellow #6, FD&C red #40, FD&C blue #2, hydroxypropyl cellulose, hypromellose, microcrystalline cellulose, magnesium stearate, polyethylene glycol, polysorbate 80, talc and titanium dioxide.
CLINICAL PHARMACOLOGY
Pharmacokinetics
Gastrointestinal absorption of nystatin is insignificant. Most orally administered nystatin is passed unchanged in the stool. In patients with renal insufficiency receiving oral therapy with conventional dosage forms, significant plasma concentrations of nystatin may occasionally occur.
Microbiology
Nystatin is both fungistatic and fungicidal in vitro against a wide variety of yeasts and yeast-like fungi. Candida albicans demonstrates no significant resistance to nystatin in vitro on repeated subculture in increasing levels of nystatin; other Candida species become quite resistant. Generally, resistance does not develop in vivo . Nystatin acts by binding to sterols in the cell membrane of susceptible Candida species with a resultant change in membrane permeability allowing leakage of intracellular components. Nystatin exhibits no appreciable activity against bacteria, protozoa, or viruses.
HOW SUPPLIED
Nystatin Tablets, USP 500,000 Units are round brown, film-coated tablets debossed "HP51" on one side and plain on the other side are packaged in: Bottles of 90: NDC 62135-741-90
Store at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature].
Manufactured by: Vivimed Life Sciences Private Limited Alathur, Kanchipuram – 603 110, Tamilnadu, India. M.L. No.: TN00002327
Manufactured for: Chartwell RX, LLC. Congers, NY 10920
Revised: 08/2023
L71669