Get your patient on Octreotide Acetate - Octreotide Acetate injection, Solution (Octreotide Acetate)
Octreotide Acetate - Octreotide Acetate injection, Solution prescribing information
1 INDICATIONS AND USAGE
Octreotide acetate injection is a somatostatin analogue indicated:
- Acromegaly : To reduce blood levels of growth hormone (GH) and insulin growth factor-1 (IGF-1; somatomedin C) in acromegaly patients who have had inadequate response to or cannot be treated with surgical resection, pituitary irradiation, and bromocriptine mesylate at maximally tolerated doses. (1.1 )
- Carcinoid Tumors : For the symptomatic treatment of patients with metastatic carcinoid tumors where it suppresses or inhibits the severe diarrhea and flushing episodes associated with the disease. (1.2 )
- Vasoactive Intestinal Peptide Tumors (VIPomas) : For the treatment of profuse watery diarrhea associated with VIP-secreting tumors. (1.3 )
Limitations of Use
Improvement in clinical signs and symptoms, or reduction in tumor size or rate of growth, were not shown in clinical trials performed with octreotide acetate injection; these trials were not optimally designed to detect such effects. (1.4 )
1.1 Acromegaly
Octreotide acetate injection is indicated to reduce blood levels of growth hormone (GH) and insulin growth factor-1 (IGF-1; somatomedin C) in acromegaly patients who have had inadequate response to or cannot be treated with surgical resection, pituitary irradiation, and bromocriptine mesylate at maximally tolerated doses.
1.2 Carcinoid Tumors
Octreotide acetate injection is indicated for treatment of severe diarrhea and flushing episodes associated with metastatic carcinoid tumors.
1.3 Vasoactive Intestinal Peptide Tumors
Octreotide acetate injection is indicated for the treatment of the profuse watery diarrhea associated with vasoactive intestinal peptide tumors (VIPomas)-secreting tumors.
1.4 Important Limitations of Use
Improvement in clinical signs and symptoms, or reduction in tumor size or rate of growth, were not shown in clinical trials performed with octreotide acetate injection; these trials were not optimally designed to detect such effects.
2 DOSAGE AND ADMINISTRATION
- Octreotide acetate injection may be administered subcutaneously or intravenously. (2.1 )
- Acromegaly : Recommended initial octreotide acetate injection dosage is 50 mcg three times daily during the initial 2 weeks of therapy. Maintenance dose 100 mcg to 500 mcg three times daily. (2.2 )
- Carcinoid Tumors : Recommended dosage range of 100 mcg to 600 mcg daily in two to four divided doses during the initial 2 weeks of therapy. (2.3 )
- VIPomas : Recommended dosage range of 200 mcg to 300 mcg daily in two to four divided doses during the initial 2 weeks of therapy. (2.4 )
2.1 Dosage and Administration Overview
- Octreotide acetate injection may be administered subcutaneously or intravenously. Pain with subcutaneous administration may be reduced by using the smallest volume that will deliver the desired dose. Sites should be rotated in a systematic manner.
- Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration. Do not use if particulates and/or discoloration are observed. Octreotide acetate injection is not compatible in Total Parenteral Nutrition solutions because of the formation of a glycosyl octreotide conjugate which may decrease the efficacy of the product.
- Octreotide acetate injection may be diluted in volumes of 50 mL to 200 mL and infused intravenously over 15 to 30 minutes or administered by intravenous (IV) push over 3 minutes. In emergency situations (e.g., carcinoid crisis), it may be given by rapid bolus.
- Assess total and/or free T4 levels at baseline and periodically during chronic octreotide acetate injection therapy.
2.2 Recommended Dosage and Monitoring for Acromegaly
The recommended initial dosage of octreotide acetate injection is 50 mcg three times daily to be administered subcutaneously. Increase octreotide acetate injection dose based upon GH or IGF-1 levels. The goal is to achieve GH levels less than 5 ng/mL or IGF-1 levels within normal range. Monitor GH or IGF-1 every two weeks after initiating octreotide acetate injection therapy or with dosage change, and to guide titration.
The most common dosage is 100 mcg three times daily, but some patients require up to 500 mcg three times daily for maximum effectiveness. Doses greater than 300 mcg/day seldom result in additional biochemical benefit, and if an increase in dose fails to provide additional benefit, the dose should be reduced.
Octreotide acetate injection should be withdrawn yearly for approximately 4 weeks from patients who have received irradiation to assess disease activity. If GH or IGF-1 levels increase and signs and symptoms recur, octreotide acetate injection therapy may be resumed.
2.3 Recommended Dosage and Monitoring for Carcinoid Tumors
The recommended daily dosage of octreotide acetate injection during the first 2 weeks of therapy ranges from 100 to 600 mcg/day in two to four divided doses given subcutaneously (mean daily dosage is 300 mcg). In the clinical studies, the median daily maintenance dosage was approximately 450 mcg, but clinical and biochemical benefits were obtained in some patients with as little as 50 mcg, while others required doses up to 1500 mcg/day. However, experience with doses above 750 mcg/day is limited. Measurement of urinary 5-hydroxyindole acetic acid, plasma serotonin, plasma Substance P may be useful in monitoring the progress of therapy.
2.4 Recommended Dosage and Monitoring for Vasoactive Intestinal Peptide Tumors
Daily dosages of 200 mcg to 300 mcg in two to four divided doses given subcutaneously are recommended during the initial 2 weeks of therapy (range, 150 mcg to 750 mcg) to control symptoms of the disease. On an individual basis, dosage may be adjusted to achieve a therapeutic response, but usually doses above 450 mcg/day are not required. Measurement of Plasma vasoactive intestinal peptide (VIP) may be useful in monitoring the progress of therapy.
3 DOSAGE FORMS AND STRENGTHS
Injection: 100 mcg/mL of octreotide (as acetate) as a clear solution in a single-dose vial.
8 USE IN SPECIFIC POPULATIONS
- Females and Males of Reproductive Potential : Advise premenopausal females of the potential for an unintended pregnancy. (8.3 )
8.1 Pregnancy
Risk Summary
The limited data with octreotide acetate injection in pregnant women are insufficient to inform a drug-associated risk for major birth defects and miscarriage. In animal reproduction studies, no adverse developmental-effects were observed with IV administration of octreotide to pregnant rats and rabbits during organogenesis at doses 7- and 13-times, respectively the maximum recommended human dose (MRHD) of 1.5 mg/day based on body surface area (BSA). Transient growth retardation, with no impact on postnatal development, was observed in rat offspring from a pre- and post-natal study of octreotide at IV doses below the MRHD based on BSA ( see Data ).
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Data
Human Data
In postmarketing data, a limited number of exposed pregnancies have been reported in patients with acromegaly. Most women were exposed to octreotide during the first trimester of pregnancy at doses ranging from 100 to 300 mcg/day of octreotide acetate injection or 20 mg to 30 mg once a month of octreotide acetate for injectable suspension, however some women elected to continue octreotide therapy throughout pregnancy. In cases with a known outcome, no congenital malformations were reported.
Animal Data
In embryo-fetal development studies in rats and rabbits, pregnant animals received IV doses of octreotide up to 1 mg/kg/day during the period of organogenesis. A slight reduction in body weight gain was noted in pregnant rats at 0.1 and 1 mg/kg/day. There were no maternal effects in rabbits or embryo-fetal effects in either species up to the maximum dose tested. At 1 mg/kg/day in rats and rabbits, the dose multiple was approximately 7- and 13-times, respectively, at the highest recommended human dose of 1.5 mg/day based on BSA.
In a pre- and post-natal development rat study at IV doses of 0.02-1 mg/kg/day, a transient growth retardation of the offspring was observed at all doses which was possibly a consequence of GH inhibition by octreotide. The doses attributed to the delayed growth are below the human dose of 1.5 mg/day, based on BSA.
8.2 Lactation
Risk Summary
There is no information available on the presence of octreotide acetate injection in human milk, the effects of the drug on the breastfed infant, or the effects of the drug on milk production. Studies show that octreotide administered subcutaneously passes into the milk of lactating rats; however, due to species-specific differences in lactation physiology, animal data may not reliably predict drug levels in human milk ( see Data ). The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for octreotide acetate injection, and any potential adverse effects on the breastfed child from octreotide acetate injection or from the underlying maternal condition.
Data
Following a subcutaneous dose (1 mg/kg) of octreotide to lactating rats, transfer of octreotide into milk was observed at a low concentration compared to plasma (milk/plasma ratio of 0.009).
8.3 Females and Males of Reproductive Potential
Discuss the potential for unintended pregnancy with premenopausal women as the therapeutic benefits of a reduction in GH levels and normalization of insulin-like growth factor 1 (IGF-1) concentration in acromegalic females treated with octreotide may lead to improved fertility.
8.4 Pediatric Use
Safety and efficacy of octreotide acetate injection in the pediatric population have not been demonstrated.
No formal controlled clinical trials have been performed to evaluate the safety and effectiveness of octreotide acetate injection in pediatric patients under age 6 years. In postmarketing reports, serious adverse events, including hypoxia, necrotizing enterocolitis, and death, have been reported with octreotide acetate injection use in children, most notably in children under 2 years of age. The relationship of these events to octreotide has not been established as the majority of these pediatric patients had serious underlying co-morbid conditions.
The efficacy and safety of octreotide acetate injection using the octreotide acetate for injectable suspension formulation was examined in a single randomized, double-blind, placebo-controlled, 6 month pharmacokinetics study in 60 pediatric patients age 6 to 17 years with hypothalamic obesity resulting from cranial insult. The mean octreotide concentration after 6 doses of 40 mg octreotide acetate for injectable suspension administered by intramuscular (IM) injection every 4 weeks was approximately 3 ng/mL. Steady-state concentrations was achieved after 3 injections of a 40-mg dose. Mean body mass index (BMI) increased 0.1 kg/m 2 in octreotide acetate for injectable suspension-treated subjects compared to 0.0 kg/m 2 in saline control-treated subjects. Efficacy was not demonstrated. Diarrhea occurred in 11 of 30 (37%) patients treated with octreotide acetate for injectable suspension. No unexpected adverse events were observed. However, with octreotide acetate for injectable suspension at 40 mg once a month, the incidence of new cholelithiasis in this pediatric population (33%) was higher than that seen in other adult indications such as acromegaly (22%) or malignant carcinoid syndrome (24%), where octreotide acetate for injectable suspension was 10 mg to 30 mg once a month.
8.5 Geriatric Use
Clinical studies of octreotide acetate injection did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
8.6 Renal Impairment
In patients with severe renal failure requiring dialysis, the half-life of octreotide acetate injection may be increased, necessitating adjustment of the maintenance dosage [see Clinical Pharmacology (12.3)] .
8.7 Hepatic Impairment-Cirrhotic Patients
In patients with liver cirrhosis, the half-life of the drug may be increased, necessitating adjustment of the maintenance dosage [see Clinical Pharmacology (12.3)] .
4 CONTRAINDICATIONS
Sensitivity to this drug or any of its components.
5 WARNINGS AND PRECAUTIONS
- Cardiac Function Abnormalities : Increased risk for higher degree of atrioventricular blocks. Consider cardiac monitoring in patients receiving octreotide acetate injection intravenously. Bradycardia, arrhythmias, or conduction abnormalities may occur. Use with caution in at-risk patients. Dosage adjustment of cardiac medications may be necessary. (5.1 )
- Cholelithiasis and Complications of Cholelithiasis : Monitor periodically. Discontinue if complications of cholelithiasis are suspected. (5.2 )
- Glucose Metabolism : Hypoglycemia or hyperglycemia may occur. Glucose monitoring is recommended and anti-diabetic treatment may need adjustment. (5.3 )
- Thyroid Function : Hypothyroidism may occur. Monitor thyroid levels periodically. (5.4 )
- Steatorrhea and Malabsorption of Dietary Fats : New onset steatorrhea, stool discoloration, loose stools, abdominal bloating, and weight loss may occur. If new occurrence or worsening of these symptoms are reported, evaluate for potential pancreatic exocrine insufficiency. (5.5 )
5.1 Cardiac Function Abnormalities
Complete Atrioventricular Block
Patients who receive octreotide acetate injection intravenously may be at increased risk for higher degree atrioventricular blocks. In postmarketing reports, complete atrioventricular block was reported in patients receiving IV octreotide acetate injection during surgical procedures. In the majority of patients, octreotide acetate injection was given at higher than recommended doses and/or as a continuous IV infusion. The safety of continuous IV infusion has not been established in patients receiving octreotide acetate injection for the approved indications. Consider cardiac monitoring in patients receiving octreotide acetate injection intravenously.
Other Cardiac Conduction Abnormalities
Other cardiac conduction abnormalities have occurred during treatment with octreotide acetate injection. In acromegalic patients, bradycardia (< 50 bpm) developed in 25%; conduction abnormalities occurred in 10% and arrhythmias occurred in 9% of patients during octreotide acetate injection therapy [see Adverse Reactions (6)] . Other electrocardiogram (ECG) changes observed included QT prolongation, axis shifts, early repolarization, low voltage, R/S transition, and early R-wave progression. These ECG changes are not uncommon in acromegalic patients. Dose adjustments in drugs such as beta-blockers that have bradycardia effects may be necessary. In one acromegalic patient with severe congestive heart failure (CHF), initiation of octreotide acetate injection therapy resulted in worsening of CHF with improvement when drug was discontinued. Confirmation of a drug effect was obtained with a positive rechallenge.
5.2 Cholelithiasis and Complications of Cholelithiasis
Octreotide acetate injection may inhibit gallbladder contractility and decrease bile secretion, which may lead to gallbladder abnormalities or sludge. Acute cholecystitis, ascending cholangitis, biliary obstruction, cholestatic hepatitis, or pancreatitis have been reported with octreotide acetate injection therapy. In clinical trials (primarily patients with acromegaly or psoriasis), the incidence of biliary tract abnormalities was 63% (27% gallstones, 24% sludge without stones, 12% biliary duct dilatation). The incidence of stones or sludge in patients who received octreotide acetate injection for 12 months or longer was 52%. Less than 2% of patients treated with octreotide acetate injection for 1 month or less developed gallstones. One patient developed ascending cholangitis during octreotide acetate injection therapy and died. If complications of cholelithiasis are suspected, discontinue octreotide acetate injection and treat appropriately.
5.3 Hyperglycemia and Hypoglycemia
Octreotide acetate injection alters the balance between the counter-regulatory hormones, insulin, glucagon and GH, which may result in hypoglycemia or hyperglycemia. The hypoglycemia or hyperglycemia which occurs during octreotide acetate injection therapy is usually mild but may result in overt diabetes mellitus or necessitate dose changes in insulin or other anti-diabetic agents. Hypoglycemia and hyperglycemia occurred on octreotide acetate injection in 3% and 16% of acromegalic patients, respectively [see Adverse Reactions (6)] . Severe hyperglycemia, subsequent pneumonia, and death following initiation of octreotide acetate injection therapy was reported in one patient with no history of hyperglycemia.
Monitor glucose levels during octreotide acetate injection therapy. Adjust dosing of insulin or other anti-diabetic therapy accordingly.
5.4 Thyroid Function Abnormalities
Octreotide suppresses secretion of thyroid stimulating hormone (TSH), which may result in hypothyroidism. Baseline and periodic assessment of thyroid function (TSH, total, and/or free T 4 ) is recommended during chronic therapy [see Adverse Reactions (6)] .
5.5 Steatorrhea and Malabsorption of Dietary Fats
New onset steatorrhea, stool discoloration and loose stools have been reported in patients receiving somatostatin analogs, including octreotide acetate injection. Somatostatin analogs reversibly inhibit secretion of pancreatic enzymes and bile acids, which may result in malabsorption of dietary fats and subsequent symptoms of steatorrhea, loose stools, abdominal bloating, and weight loss. If new occurrence or worsening of these symptoms are reported in patients receiving octreotide acetate injection, evaluate patients for potential pancreatic exocrine insufficiency and manage accordingly.
5.6 Changes in Vitamin B12 Levels
Depressed vitamin B 12 levels and abnormal Schilling’s tests have been observed in some patients receiving octreotide acetate injection therapy, and monitoring of vitamin B 12 levels is recommended during octreotide acetate injection therapy.
6 ADVERSE REACTIONS
Most common adverse reactions (incidence > 10%) in patients with acromegaly are gallbladder abnormalities, sinus bradycardia, diarrhea, loose stools, nausea, abdominal discomfort, hyperglycemia, and hypothyroidism. In other patients, most common adverse reactions (incidence > 10%) are gallbladder abnormalities. (6.1 )
To report SUSPECTED ADVERSE REACTIONS, contact Hainan Shuangcheng Pharmaceuticals Co., Ltd. at 86-0898-6856-1216 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch .
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Complete Atrioventricular Block [see Warnings and Precautions (5.1)]
- Cholelithiasis and Complications of Cholelithiasis [see Warnings and Precautions (5.2)]
- Hyperglycemia and Hypoglycemia [see Warnings and Precautions (5.3)]
- Thyroid Function Abnormalities [see Warnings and Precautions (5.4)]
- Steatorrhea and Malabsorption of Dietary Fats [see Warnings and Precautions (5.5)]
- Changes in Vitamin B12 Levels [see Warnings and Precautions (5.6)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Gallbladder Abnormalities
Gallbladder abnormalities, especially stones and/or biliary sludge, frequently develop in patients on chronic octreotide acetate injection therapy [see Warnings and Precautions (5.1)] . In clinical trials (primarily patients with acromegaly or psoriasis), the incidence of biliary tract abnormalities was 63% (27% gallstones, 24% sludge without stones, 12% biliary duct dilatation). The incidence of stones or sludge in patients who received octreotide acetate injection for 12 months or longer was 52%. Less than 2% of patients treated with octreotide acetate injection for 1 month or less developed gallstones.
Cardiac
In acromegalics, sinus bradycardia (< 50 bpm) developed in 25%; conduction abnormalities occurred in 10% and arrhythmias developed in 9% of patients during octreotide acetate injection therapy [see Warnings and Precautions (5.1)] .
Gastrointestinal
Diarrhea, loose stools, nausea, and abdominal discomfort were each seen in 34% to 61% of acromegalic patients in U.S. studies. 2.6% of the patients discontinued therapy due to these symptoms. These symptoms were seen in 5% to 10% of patients with carcinoid tumors and VIPomas.
The frequency of these symptoms was not dose related, but diarrhea and abdominal discomfort generally resolved more quickly in patients treated with 300 mcg/day than in those treated with 750 mcg/day. Vomiting, flatulence, abnormal stools, abdominal distention, and constipation were each seen in less than 10% of patients.
In rare instances, gastrointestinal side effects may resemble acute intestinal obstruction, with progressive abdominal distension, severe epigastric pain, abdominal tenderness, and guarding.
Hypo/Hyperglycemia
Hypoglycemia and hyperglycemia occurred in 3% and 16% of acromegalic patients, respectively, but only in about 1.5% of other patients. Symptoms of hypoglycemia were noted in approximately 2% of patients.
Hypothyroidism
In acromegalics, biochemical hypothyroidism alone occurred in 12% while goiter occurred in 8% and 4% required initiation of thyroid replacement therapy during octreotide acetate injection therapy [see Warnings and Precautions (5.4)] . In patients without acromegaly, hypothyroidism has only been reported in several isolated patients and goiter has not been reported.
Other Adverse Events
Pain on injection was reported in 7.7%, headache in 6%, and dizziness in 5%. Pancreatitis was also observed [see Warnings and Precautions (5.2)] .
Other Adverse Events 1% to 4%
Other events, each observed in 1% to 4% of patients, included fatigue, weakness, pruritus, joint pain, backache, urinary tract infection, cold symptoms, flu symptoms, injection site hematoma, bruise, edema, flushing, blurred vision, pollakiuria, fat malabsorption, hair loss, visual disturbance, and depression.
Anaphylactoid reactions, including anaphylactic shock, have been reported in several patients receiving octreotide acetate injection.
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of octreotide acetate injection. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Hepatobiliary : cholelithiasis, cholecystitis, cholangitis and pancreatitis, which have sometimes required cholecystectomy
Gastrointestinal : intestinal obstruction, pancreatic exocrine insufficiency
Hematologic : thrombocytopenia
7 DRUG INTERACTIONS
- The following drugs require monitoring and possible dose adjustment when used with octreotide acetate injection : cyclosporine, insulin, oral hypoglycemic agents, beta-blockers, and bromocriptine. (7 )
- Lutetium Lu 177 Dotatate Injection : Discontinue octreotide acetate injection at least 24 hours prior to each lutetium Lu 177 dotatate dose. (7.6 )
7.1 Cyclosporine
Octreotide has been associated with alterations in nutrient absorption, so it may have an effect on absorption of orally administered drugs. Concomitant administration of octreotide acetate injection with cyclosporine may decrease blood levels of cyclosporine and result in transplant rejection.
7.2 Insulin and Oral Hypoglycemic Drugs
Octreotide inhibits the secretion of insulin and glucagon. Therefore, blood glucose levels should be monitored when octreotide acetate injection treatment is initiated or when the dose is altered and anti-diabetic treatment should be adjusted accordingly.
7.3 Bromocriptine
Concomitant administration of octreotide and bromocriptine increases the availability of bromocriptine.
7.4 Other Concomitant Drug Therapy
Concomitant administration of bradycardia-inducing drugs (e.g., beta-blockers) may have an additive effect on the reduction of heart rate associated with octreotide. Dose adjustments of concomitant medication may be necessary.
Octreotide has been associated with alterations in nutrient absorption, so it may have an effect on absorption of orally administered drugs.
7.5 Drug Metabolism Interactions
Limited published data indicate that somatostatin analogs might decrease the metabolic clearance of compounds known to be metabolized by cytochrome P450 enzymes, which may be due to the suppression of GH. Since it cannot be excluded that octreotide may have this effect, other drugs mainly metabolized by CYP3A4 and which have a low therapeutic index (e.g., quinidine, terfenadine) should therefore be used with caution.
7.6 Lutetium Lu 177 Dotatate Injection
Octreotide competitively binds to somatostatin receptors and may interfere with the efficacy of lutetium Lu 177 dotatate. Discontinue octreotide acetate injection at least 24 hours prior to each lutetium Lu 177 dotatate dose.
11 DESCRIPTION
Octreotide acetate injection, a cyclic octapeptide prepared as a clear sterile solution of octreotide, acetate salt, in a buffered lactic acid solution for administration by deep subcutaneous or IV injection. Octreotide acetate, known chemically as L-Cysteinamide, D-phenylalanyl-L-cysteinyl-L-phenylalanyl-D-tryptophyl-L-lysyl-L-threonyl-N-[2-hydroxy-1-(hydroxymethyl)propyl]-,cyclic (2 → 7)-disulfide; [R-(R•, R•)] acetate salt, is a long-acting octapeptide with pharmacologic actions mimicking those of the natural hormone somatostatin.
Octreotide acetate injection is available as sterile 1-mL single-dose vials in one strength, containing 100 mcg octreotide (as acetate). Each single-dose vial also contains L-lactic acid, USP (3.4 mg), mannitol, USP (45 mg), sodium bicarbonate, USP (quantity sufficient to pH 4.2 ± 0.3), water for injection, USP (quantity sufficient to 1 mL).
Lactic acid and sodium bicarbonate are added to provide a buffered solution, pH to 4.2 ± 0.3.
The molecular weight of octreotide acetate is 1019.3 g/mol (free peptide, C 49 H 66 N 10 O 10 S 2 ) and its amino acid sequence is:
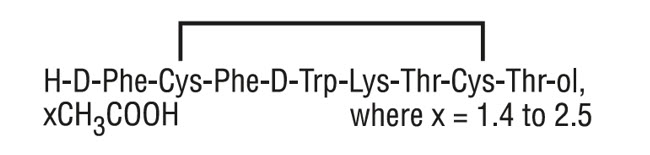
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Octreotide acetate injection exerts pharmacologic actions similar to the natural hormone, somatostatin. It is an even more potent inhibitor of GH, glucagon, and insulin than somatostatin. Like somatostatin, it also suppresses luteinizing hormone (LH) response to gonadotropin releasing hormone (GnRH), decreases splanchnic blood flow, and inhibits release of serotonin, gastrin, VIP, secretin, motilin, and pancreatic polypeptide.
By virtue of these pharmacological actions, octreotide has been used to treat the symptoms associated with metastatic carcinoid tumors (flushing and diarrhea), and VIP secreting adenomas (watery diarrhea).
12.2 Pharmacodynamics
Octreotide substantially reduces GH and/or IGF-1 (somatomedin C) levels in patients with acromegaly.
Single doses of octreotide have been shown to inhibit gallbladder contractility and to decrease bile secretion in normal volunteers. In controlled clinical trials, the incidence of gallstone or biliary sludge formation was markedly increased [see Warnings and Precautions (5.2)] .
Octreotide suppresses secretion of TSH.
12.3 Pharmacokinetics
Absorption
After subcutaneous injection, octreotide is absorbed rapidly and completely from the injection site. Peak concentrations of 5.2 ng/mL (100-mcg dose) were reached 0.4 hours after dosing. Using a specific radioimmunoassay, IV and subcutaneous doses were found to be bioequivalent. Peak concentrations and area under the curve (AUC) values were dose proportional after IV single doses up to 200 mcg and subcutaneous single doses up to 500 mcg and after subcutaneous multiple doses up to 500 mcg 3 times a day (1,500 mcg/day). In patients with acromegaly, a mean peak concentration of 2.8 ng/mL (100-mcg dose) was reached in 0.7 hours after subcutaneous dosing.
Distribution
In healthy volunteers, the distribution of octreotide from plasma was rapid (tα½ = 0.2 h), the volume of distribution (V dss ) was estimated to be 13.6 L, and the total body clearance ranged from 7 L/hr to 10 L/hr. In blood, the distribution into the erythrocytes was found to be negligible and about 65% was bound in the plasma in a concentration-independent manner. Binding was mainly to lipoprotein and, to a lesser extent, to albumin. In patients with acromegaly, the volume of distribution (V dss ) was estimated to be 21.6 ± 8.5 L, and the total body clearance was increased to 18 L/h. The mean percent of the drug bound was 41.2%.
Elimination
The elimination of octreotide from plasma had an apparent half-life of 1.7 to 1.9 hours compared with 1 to 3 minutes with the natural hormone. The duration of action of octreotide acetate injection is variable but extends up to 12 hours depending upon the type of tumor. About 32% of the dose is excreted unchanged into the urine. In an elderly population, dose adjustments may be necessary due to a significant increase in the half-life (46%) and a significant decrease in the clearance (26%) of the drug.
In patients with acromegaly, the disposition and elimination half-lives were similar to normal subjects.
Specific Populations
Renal Impairment
In patients with mild renal impairment (CL CR 40 to 60 mL/min), octreotide t 1/2 was 2.4 hours and total body clearance was 8.8 L/hr, in moderate impairment (CL CR 10 to 39 mL/min) t 1/2 was 3.0 hours and total body clearance 7.3 L/hr. In patients with severe renal impairment not requiring dialysis (CL CR < 10 mL/min), octreotide t 1/2 was 3.1 hours and total body clearance was 7.6 L/hr. In patients with severe renal failure requiring dialysis, total body clearance was reduced to about half that found in healthy subjects (from approximately 10 L/hr to 4.5 L/hr).
Hepatic Impairment
Patients with liver cirrhosis showed prolonged elimination of drug, with octreotide t 1/2 increasing to 3.7 hr and total body clearance decreasing to 5.9 L/hr, whereas patients with fatty liver disease showed t 1/2 increased to 3.4 hr and total body clearance of 8.2 L/hr.
12.6 Immunogenicity
Evaluation of 20 patients treated for at least 6 months has failed to demonstrate titers of antibodies exceeding background levels. However, antibody titers to octreotide acetate injection were subsequently reported in 3 patients and resulted in prolonged duration of drug action in 2 patients.
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Studies in laboratory animals have demonstrated no mutagenic potential of octreotide acetate injection.
No carcinogenic potential was demonstrated in mice treated subcutaneously for 85 to 99 weeks at doses up to 2,000 mcg/kg/day (8 x the human exposure based on BSA). In a 116-week subcutaneous study in rats, a 27% and 12% incidence of injection-site sarcomas or squamous cell carcinomas was observed in males and females, respectively, at the highest dose level of 1,250 mcg/kg/day (10 x the human exposure based on BSA) compared to an incidence of 8% to 10% in the vehicle-control groups. The increased incidence of injection-site tumors was most probably caused by irritation and the high sensitivity of the rat to repeated subcutaneous injections at the same site. Rotating injection sites would prevent chronic irritation in humans. There have been no reports of injection-site tumors in patients treated with octreotide acetate injection for up to 5 years. There was also a 15% incidence of uterine adenocarcinomas in the 1,250 mcg/kg/day females compared to 7% in the saline-control females and 0% in the vehicle-control females. The presence of endometritis coupled with the absence of corpora lutea, the reduction in mammary fibroadenomas, and the presence of uterine dilatation suggest that the uterine tumors were associated with estrogen dominance in the aged female rats which does not occur in humans.
Octreotide did not impair fertility in rats at doses up to 1,000 mcg/kg/day, which represents 7-times the human exposure based on BSA.
16 HOW SUPPLIED/STORAGE AND HANDLING
How Supplied
Octreotide acetate injection is available in 1 mL single-dose vials as follows:
Single-dose vials
100 mcg/mL octreotide (as acetate)
Package of 10 single-dose vials ............................................................................................................................... NDC 52958-046-10
Storage and Handling
For prolonged storage, octreotide acetate injection single-dose vials should be stored at refrigerated temperatures 2°C to 8°C (36°F to 46°F) and store in outer carton in order to protect from light. At room temperature (20°C to 30°C or 70°F to 86°F), octreotide acetate injection is stable for 14 days if protected from light. The solution can be allowed to come to room temperature prior to administration. Do not warm artificially. Single-dose vials should be opened just prior to administration and the unused portion discarded. Dispose unused product or waste properly. Octreotide acetate injection is stable in sterile isotonic saline solutions or sterile solutions of dextrose 5% in water for 24 hours.
12.1 Mechanism of Action
Octreotide acetate injection exerts pharmacologic actions similar to the natural hormone, somatostatin. It is an even more potent inhibitor of GH, glucagon, and insulin than somatostatin. Like somatostatin, it also suppresses luteinizing hormone (LH) response to gonadotropin releasing hormone (GnRH), decreases splanchnic blood flow, and inhibits release of serotonin, gastrin, VIP, secretin, motilin, and pancreatic polypeptide.
By virtue of these pharmacological actions, octreotide has been used to treat the symptoms associated with metastatic carcinoid tumors (flushing and diarrhea), and VIP secreting adenomas (watery diarrhea).