Get your patient on Ofloxacin - Ofloxacin tablet, Film Coated (Ofloxacin)
Ofloxacin - Ofloxacin tablet, Film Coated prescribing information
WARNING
SERIOUS ADVERSE REACTIONS INCLUDING TENDINITIS, TENDON RUPTURE, PERIPHERAL NEUROPATHY, CENTRAL NERVOUS SYSTEM EFFECTS AND EXACERBATION OF MYASTHENIA GRAVIS
Fluoroquinolones, including ofloxacin, have been associated with disabling and potentially irreversible serious adverse reactions that have occurred together, including: • Tendinitis and tendon rupture • Peripheral neuropathy • Central nervous system effects (see WARNINGS ). Discontinue ofloxacin immediately and avoid the use of fluoroquinolones, including ofloxacin, in patients who experience any of these serious adverse reactions (see WARNINGS). • Fluoroquinolones, including ofloxacin, may exacerbate muscle weakness in persons with myasthenia gravis. Avoid ofloxacin in patients with a known history of myasthenia gravis (See WARNINGS). •Because fluoroquinolones, including ofloxacin, have been associated with serious adverse reactions (see WARNINGS), reserve ofloxacin for use in patients who have no alternative treatment options for the following indications: o Acute exacerbation of chronic bronchitis o Uncomplicated cystitis (see INDICATIONS and USAGE )
INDICATIONS AND USAGE
To reduce the development of drug-resistant bacteria and maintain the effectiveness of ofloxacin tablets, USP and other antibacterial drugs, ofloxacin tablets, USP should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy. Ofloxacin tablets, USP are indicated for the treatment of adults with mild to moderate infections (unless otherwise indicated) caused by susceptible strains of the designated microorganisms in the infections listed below. Please see DOSAGE AND ADMINISTRATION for specific recommendations. Acute Bacterial Exacerbations of Chronic Bronchitis (ABECB) due to Haemophilus influenzae or Streptococcus pneumonia .
Because fluoroquinolones, including ofloxacin, have been associated with serious adverse reactions (see Warnings ), - and for some patients ABECB is self-limiting, reserve ofloxacin for treatment of ABECB in patients who have no alternative treatment options . Community-Acquired Pneumonia due to Haemophilus influenzae or Streptococcus pneumonia .
Uncomplicated Skin and Skin Structure Infections due to methicillin-susceptible Staphylococcus aureus, Streptococcus pyogenes, or Proteus mirabilis. Acute, Uncomplicated Urethral and Cervical Gonorrhea due to Neisseria gonorrhoeae (see WARNINGS ). Nongonococcal Urethritis and Cervicitis due to Chlamydia trachomatis (see WARNINGS ). Mixed Infections of the Urethra and Cervix due to Chlamydia trachomatis and Neisseria gonorrhoeae (see WARNINGS ). Acute Pelvic Inflammatory Disease (including severe infection) due to Chlamydia trachomatis and/or Neisseria gonorrhoeae (see WARNINGS ). NOTE: If anaerobic microorganisms are suspected of contributing to the infection, appropriate therapy for anaerobic pathogens should be administered. Uncomplicated Cystitis due to Citrobacter diversus, Enterobacter aerogenes, Escherichia coli, Klebsiella pneumoniae, Proteus mirabilis, or Pseudomonas aeruginosa. Because fluoroquinolones, including ofloxacin, have been associated with serious adverse reactions (see WARNINGS ), and for some patients uncomplicated cystitis is self-limiting, reserve ofloxacin for treatment of uncomplicated cystitis in patients who have no alternative treatment options. Complicated Urinary Tract Infections due to Escherichia coli, Klebsiella pneumoniae, Proteus mirabilis, Citrobacter diversus,• or Pseudomonas aeruginosa.•
Prostatitis due to Escherichia coli. • = Although treatment of infections due to this organism in this organ system demonstrated a clinically significant outcome, efficacy was studied in fewer than 10 patients.
Appropriate culture and susceptibility tests should be performed before treatment in order to isolate and identify organisms causing the infection and to determine their susceptibility to ofloxacin, USP. Therapy with ofloxacin, USP may be initiated before results of these tests are known; once results become available, appropriate therapy should be continued. As with other drugs in this class, some strains of Pseudomonas aeruginosa may develop resistance fairly rapidly during treatment with ofloxacin, USP. Culture and susceptibility testing performed periodically during therapy will provide information not only on the therapeutic effect of the antimicrobial agent but also on the possible emergence of bacterial resistance.
DOSAGE AND ADMINISTRATION
The usual dose of ofloxacin tablets is 200 mg to 400 mg orally every 12 h as described in the following dosing chart. These recommendations apply to patients with normal renal function (i.e., creatinine clearance > 50 mL/min). For patients with altered renal function (i.e., creatinine clearance < 50 mL/min), see the Patients With Impaired Renal Function subsection.
| Infection† | Unit Dose | Frequency | Duration | Daily Dose |
| Acute Bacterial Exacerbation of Chronic Bronchitis | 400 mg | q12h | 10 days | 800 mg |
| Comm. Acquired Pneumonia | 400 mg | q12h | 10 days | 800 mg |
| Uncomplicated Skin and Skin Structure Infections | 400 mg | q12h | 10 days | 800 mg |
| Acute, Uncomplicated Urethral and Cervical Gonorrhea | 400 mg | Single Dose | 1 day | 400 mg |
| Nongonococcal Cervicitis/Urethritis due to C. trachomatis | 300 mg | q12h | 7 days | 600 mg |
| Mixed Infection of the Urethra and Cervix due to C. trachomatis and N. gonorrhoeae | 300 mg | q12h | 7 days | 600 mg |
| Acute Pelvic Inflammatory Disease | 400 mg | q12h | 10 to 14 days | 800 mg |
| Uncomplicated Cystitis due to E. coli or K. pneumoniae | 200 mg | q12h | 3 days | 400 mg |
| Uncomplicated Cystitis due to Other Approved Pathogens | 200 mg | q12h | 7 days | 400 mg |
| Complicated UTI’s | 200 mg | q12h | 10 days | 400 mg |
| Prostatitis due to E. coli | 300 mg | q12h | 6 weeks | 600 mg |
| † DUE TO THE DESIGNATED PATHOGENS (see INDICATIONS AND USAGE ). | ||||
Antacids containing calcium, magnesium, or aluminum; sucralfate; divalent or trivalent cations such as iron; or multivitamins containing zinc; or didanosine, chewable/buffered tablets or the pediatric powder for oral solution should not be taken within the two-hour period before or within the two-hour period after taking ofloxacin (see PRECAUTIONS ). Patients With Impaired Renal Function Dosage should be adjusted for patients with a creatinine clearance < 50 mL/min. After a normal initial dose, dosage should be adjusted as follows:
| Creatinine Clearance | Maintenance Dose | Frequency |
| 20 to 50 mL/min | the usual recommended unit dose | q24h |
| < 20 mL/min | ½ the usual recommended unit dose | q24h |
When only the serum creatinine is known, the following formula may be used to estimate creatinine clearance. 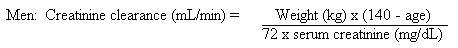 Women: 0.85 x the value calculated for men. The serum creatinine should represent a steady-state of renal function.
Women: 0.85 x the value calculated for men. The serum creatinine should represent a steady-state of renal function.
Patients With Cirrhosis:
The excretion of ofloxacin may be reduced in patients with severe liver function disorders (e.g., cirrhosis with or without ascites). A maximum dose of 400 mg of ofloxacin per day should therefore not be exceeded.
CONTRAINDICATIONS
Ofloxacin tablets are contraindicated in persons with a history of hypersensitivity associated with the use of ofloxacin or any member of the quinolone group of antimicrobial agents.
ADVERSE REACTIONS
The following is a compilation of the data for ofloxacin based on clinical experience with both the oral and intravenous formulations. The incidence of drug-related adverse reactions in patients during Phase 2 and 3 clinical trials was 11%. Among patients receiving multiple-dose therapy, 4% discontinued ofloxacin due to adverse experiences. In clinical trials, the following events were considered likely to be drug-related in patients receiving multiple doses of ofloxacin: nausea 3%, insomnia 3%, headache 1%, dizziness 1%, diarrhea 1%, vomiting 1%, rash 1%, pruritus 1%, external genital pruritus in women 1%, vaginitis 1%, dysgeusia 1%. In clinical trials, the most frequently reported adverse events, regardless of relationship to drug, were: nausea 10%, headache 9%, insomnia 7%, external genital pruritus in women 6%, dizziness 5%, vaginitis 5%, diarrhea 4%, vomiting 4%.
In clinical trials, the following events, regardless of relationship to drug, occurred in 1 to 3% of patients: abdominal pain and cramps, chest pain, decreased appetite, dry mouth, dysgeusia, fatigue, flatulence, gastrointestinal distress, nervousness, pharyngitis, pruritus, fever, rash, sleep disorders, somnolence, trunk pain, vaginal discharge, visual disturbances, and constipation. Additional events, occurring in clinical trials at a rate of less than 1%, regardless of relationship to drug, were:
| Body as a Whole: | asthenia, chills, malaise, extremity pain, pain, epistaxis |
| Cardiovascular System: | cardiac arrest, edema, hypertension, hypotension, palpitations, vasodilation |
| Gastrointestinal System: | dyspepsia |
| Genital/Reproductive System: | burning, irritation, pain and rash of the female genitalia; dysmenorrhea; menorrhagia; metrorrhagia |
| Musculoskeletal System: | arthralgia, myalgia |
| Nervous System: | seizures, anxiety, cognitive change, depression, dream abnormality, euphoria, hallucinations, paresthesia, syncope, vertigo, tremor, confusion |
| Nutritional/Metabolic: | thirst, weight loss |
| Respiratory System: | respiratory arrest, cough, rhinorrhea |
| Skin/Hypersensitivity: | angioedema, diaphoresis, urticaria, vasculitis |
| Special Senses: | decreased hearing acuity, tinnitus, photophobia |
| Urinary System: | dysuria, urinary frequency, urinary retention |
The following laboratory abnormalities appeared in ≥ 1% of patients receiving multiple doses of ofloxacin. It is not known whether these abnormalities were caused by the drug or the underlying conditions being treated.
| Hematopoietic: | anemia, leukopenia, leukocytosis, neutropenia, neutrophilia, increased band forms, lymphocytopenia, eosinophilia, lymphocytosis, thrombocytopenia, thrombocytosis, elevated ESR |
| Hepatic: | elevated: alkaline phosphatase, AST (SGOT), ALT (SGPT) |
| Serum Chemistry: | hyperglycemia, hypoglycemia, elevated creatinine, elevated BUN |
| Urinary: | glucosuria, proteinuria, alkalinuria, hyposthenuria, hematuria, pyuria |
Postmarketing Adverse Events Additional adverse events, regardless of relationship to drug, reported from worldwide marketing experience with quinolones, including ofloxacin: Clinical
| Cardiovascular System: | cerebral thrombosis, pulmonary edema, tachycardia, hypotension/shock, syncope, torsade de pointes. |
| Endocrine/Metabolic: | hyper- or hypoglycemia, especially in diabetic patients on insulin or oral hypoglycemic agents (see PRECAUTIONS , General and Drug Interactions ). |
| Gastrointestinal System: | hepatic dysfunction including: hepatic necrosis, jaundice (cholestatic or hepatocellular), hepatitis; intestinal perforation; hepatic failure (including fatal cases); pseudomembranous colitis (the onset of pseudomembranous colitis symptoms may occur during or after antimicrobial treatment), GI hemorrhage; hiccough, painful oral mucosa, pyrosis (see WARNINGS ). |
| Genital/Reproductive System: | vaginal candidiasis |
| Hematopoietic: | anemia, including hemolytic and aplastic; hemorrhage, pancytopenia, agranulocytosis, leukopenia, reversible bone marrow depression, thrombocytopenia, thrombotic thrombocytopenic purpura, petechiae, ecchymosis/bruising (see WARNINGS ). |
| Musculoskeletal: | tendinitis/rupture; weakness; rhabdomyolysis (see WARNINGS ). |
| Nervous System: | nightmares; suicidal thoughts or acts, disorientation, psychotic reactions, paranoia; phobia, agitation, restlessness, aggressiveness/hostility, manic reaction, emotional lability; peripheral neuropathy that may be irreversible, ataxia, incoordination; exacerbation of: myasthenia gravis and extrapyramidal disorders; dysphasia, lightheadedness (see WARNINGS and PRECAUTIONS ). |
| Respiratory System: | dyspnea, bronchospasm, allergic pneumonitis, stridor (see WARNINGS ). |
| Skin/Hypersensitivity: | anaphylactic (-toid) reactions/shock; purpura, serum sickness, erythema multiforme/Stevens-Johnson syndrome, erythema nodosum, exfoliative dermatitis, hyperpigmentation, toxic epidermal necrolysis, conjunctivitis, photosensitivity/phototoxicity reaction, vesiculobullous eruption (see WARNINGS and PRECAUTIONS ). |
| Special Senses: | diplopia, nystagmus, blurred vision, disturbances of: taste, smell, hearing and equilibrium, usually reversible following discontinuation |
| Urinary System: | anuria, polyuria, renal calculi, renal failure, interstitial nephritis, hematuria (see WARNINGS and PRECAUTIONS ). |
Laboratory
| Hematopoietic: | prolongation of prothrombin time |
| Serum Chemistry: | acidosis, elevation of: serum triglycerides, serum cholesterol, serum potassium, liver function tests including: GGTP, LDH, bilirubin |
| Urinary: | albuminuria, candiduria |
In clinical trials using multiple-dose therapy, ophthalmologic abnormalities, including cataracts and multiple punctate lenticular opacities, have been noted in patients undergoing treatment with other quinolones. The relationship of the drugs to these events is not presently established. CRYSTALLURIA and CYLINDRURIA HAVE BEEN REPORTED with other quinolones.
Drug Interactions
Antacids, Sucralfate, Metal Cations, Multivitamins Quinolones form chelates with alkaline earth and transition metal cations. Administration of quinolones with antacids containing calcium, magnesium, or aluminum, with sucralfate, with divalent or trivalent cations such as iron, or with multivitamins containing zinc or with didanosine, chewable/buffered tablets or the pediatric powder for oral solution may substantially interfere with the absorption of quinolones resulting in systemic levels considerably lower than desired. These agents should not be taken within the two-hour period before or within the two-hour period after ofloxacin administration (see DOSAGE AND ADMINISTRATION ).
Caffeine Interactions between ofloxacin and caffeine have not been detected.
Cimetidine Cimetidine has demonstrated interference with the elimination of some quinolones. This interference has resulted in significant increases in half-life and AUC of some quinolones. The potential for interaction between ofloxacin and cimetidine has not been studied.
Cyclosporine Elevated serum levels of cyclosporine have been reported with concomitant use of cyclosporine with some other quinolones. The potential for interaction between ofloxacin and cyclosporine has not been studied.
Drugs Metabolized by Cytochrome P450 Enzymes Most quinolone antimicrobial drugs inhibit cytochrome P450 enzyme activity. This may result in a prolonged half-life for some drugs that are also metabolized by this system (e.g., cyclosporine, theophylline/methylxanthines, warfarin) when coadministered with quinolones. The extent of this inhibition varies among different quinolones (see other Drug Interactions ).
Non Steroidal Anti-Inflammatory Drugs The concomitant administration of a non-steroidal anti-inflammatory drug with a quinolone, including ofloxacin, may increase the risk of CNS stimulation and convulsive seizures (see WARNINGS and PRECAUTIONS , General ).
Probenecid The concomitant use of probenecid with certain other quinolones has been reported to affect renal tubular secretion. The effect of probenecid on the elimination of ofloxacin has not been studied.
Theophylline Steady-state theophylline levels may increase when ofloxacin and theophylline are administered concurrently. As with other quinolones, concomitant administration of ofloxacin may prolong the half-life of theophylline, elevate serum theophylline levels, and increase the risk of theophylline-related adverse reactions. Theophylline levels should be closely monitored and theophylline dosage adjustments made, if appropriate, when ofloxacin is coadministered. Adverse reactions (including seizures) may occur with or without an elevation in the serum theophylline level (see WARNINGS and PRECAUTIONS , General ).
Warfarin Some quinolones have been reported to enhance the effects of the oral anticoagulant warfarin or its derivatives. Therefore, if a quinolone antimicrobial is administered concomitantly with warfarin or its derivatives, the prothrombin time or other suitable coagulation test should be closely monitored.
Antidiabetic Agents (e.g., Insulin, Glyburide/Glibenclamide) Since disturbances of blood glucose, including hyperglycemia and hypoglycemia, have been reported in patients treated concurrently with quinolones and an antidiabetic agent, careful monitoring of blood glucose is recommended when these agents are used concomitantly (see PRECAUTIONS , General and Information for Patients ).
DESCRIPTION
Ofloxacin tablets are a synthetic broad-spectrum antimicrobial agent for oral administration. Chemically, ofloxacin, USP, a fluorinated carboxyquinolone, is the racemate, (±)-9-fluoro-2,3-dihydro-3-methyl-10-(4-methyl-1-piperazinyl)-7-oxo-7 H -pyrido[1,2,3- de ]-1,4-benzoxazine-6-carboxylic acid. The chemical structure is:
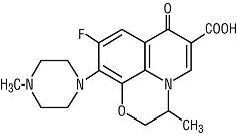
C 18 H 20 FN 3 O 4 M.W. 361.4
Ofloxacin, USP is an off-white to pale yellow crystalline powder. The molecule exists as a zwitterion at the pH conditions in the small intestine. The relative solubility characteristics of ofloxacin USP at room temperature, as defined by USP nomenclature, indicate that ofloxacin, USP is considered to be soluble in aqueous solutions with pH between 2 and 5. It is sparingly to slightly soluble in aqueous solutions with pH 7 (solubility falls to 4 mg/mL) and freely soluble in aqueous solutions with pH above 9. Ofloxacin USP has the potential to form stable coordination compounds with many metal ions. This in vitro chelation potential has the following formation order: Fe +3 > Al +3 > Cu +2 > Ni +2 > Pb +2 > Zn +2 > Mg +2 > Ca +2 > Ba +2
Ofloxacin tablets, USP contain the following inactive ingredients: lactose monohydrate, pregelatinized maize starch, hydroxy propyl methyl cellulose, talc, magnesium stearate, polyethylene glycol, sodium starch glycolate, and titanium dioxide. Additionally, the 200 mg and 400 mg tablets contain iron oxide yellow.
The imprinting ink for 200 mg, 300 mg and 400 mg strength contains FD&C blue #1, isopropyl alcohol, Nbutyl alcohol, propylene glycol, shellac and titanium dioxide.
CLINICAL PHARMACOLOGY
Following oral administration, the bioavailability of ofloxacin in the tablet formulation is approximately 98%. Maximum serum concentrations are achieved one to two hours after an oral dose. Absorption of ofloxacin after single or multiple doses of 200 to 400 mg is predictable, and the amount of drug absorbed increases proportionately with the dose. Ofloxacin has biphasic elimination. Following multiple oral doses at steady-state administration, the half-lives are approximately 4 to 5 hours and 20 to 25 hours. However, the longer half-life represents less than 5% of the total AUC. Accumulation at steady-state can be estimated using a half-life of 9 hours. The total clearance and volume of distribution are approximately similar after single or multiple doses. Elimination is mainly by renal excretion. The following are mean peak serum concentrations in healthy 70 to 80 kg male volunteers after single oral doses of 200, 300, or 400 mg of ofloxacin or after multiple oral doses of 400 mg.
| Oral Dose | Serum Concentration 2 Hours After Admin. (mcg/mL) | Area Under the Curve (AUC ( 0 to ∞) )(mcg•h/mL) |
| 200 mg single dose | 1.5 | 14.1 |
| 300 mg single dose | 2.4 | 21.2 |
| 400 mg single dose | 2.9 | 31.4 |
| 400 mg steady-state | 4.6 | 61.0 |
Steady-state concentrations were attained after four oral doses, and the area under the curve (AUC) was approximately 40% higher than the AUC after single doses. Therefore, after multiple-dose administration of 200 mg and 300 mg doses, peak serum levels of 2.2 mcg/mL and 3.6 mcg/mL, respectively, are predicted at steady-state.
In vitro , approximately 32% of the drug in plasma is protein bound.
The single dose and steady-state plasma profiles of ofloxacin injection were comparable in extent of exposure (AUC) to those of ofloxacin tablets when the injectable and tablet formulations of ofloxacin were administered in equal doses (mg/mg) to the same group of subjects. The mean steady-state AUC (0 to 12) attained after the intravenous administration of 400 mg over 60 min was 43.5 mcg•h/mL; the mean steady-state AUC (0 to 12) attained after the oral administration of 400 mg was 41.2 mcg•h/mL (two one-sided t-test, 90% confidence interval was 103 to 109). (see following chart).
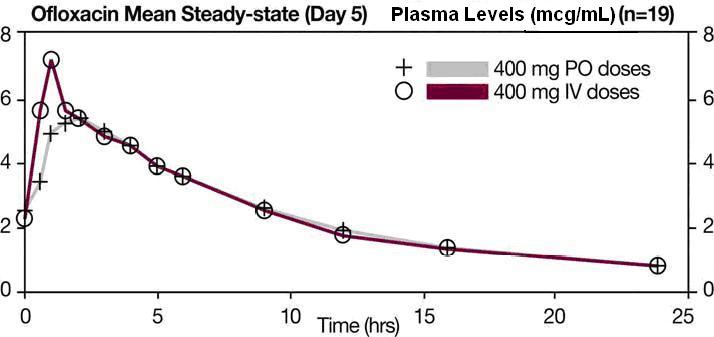
Between 0 and 6 h following the administration of a single 200 mg oral dose of ofloxacin to 12 healthy volunteers, the average urine ofloxacin concentration was approximately 220 mcg/mL. Between 12 and 24 hours after administration, the average urine ofloxacin level was approximately 34 mcg/mL. Following oral administration of recommended therapeutic doses, ofloxacin has been detected in blister fluid, cervix, lung tissue, ovary, prostatic fluid, prostatic tissue, skin, and sputum. The mean concentration of ofloxacin in each of these various body fluids and tissues after one or more doses was 0.8 to 1.5 times the concurrent plasma level. Inadequate data are presently available on the distribution or levels of ofloxacin in the cerebrospinal fluid or brain tissue. Ofloxacin has a pyridobenzoxazine ring that appears to decrease the extent of parent compound metabolism. Between 65% and 80% of an administered oral dose of ofloxacin is excreted unchanged via the kidneys within 48 hours of dosing. Studies indicate that less than 5% of an administered dose is recovered in the urine as the desmethyl or N-oxide metabolites. Four to eight percent of an ofloxacin dose is excreted in the feces. This indicates a small degree of biliary excretion of ofloxacin. The administration of ofloxacin tablets with food does not affect the C max and AUC ∞ of the drug, but T max is prolonged. Clearance of ofloxacin is reduced in patients with impaired renal function (creatinine clearance rate < 50 mL/min), and dosage adjustment is necessary. (see PRECAUTIONS, General and DOSAGE AND ADMINISTRATION ). Following oral administration to healthy elderly subjects (65 to 81 years of age), maximum plasma concentrations are usually achieved one to two hours after single and multiple twice-daily doses, indicating that the rate of oral absorption is unaffected by age or gender. Mean peak plasma concentrations in elderly subjects were 9 to 21% higher than those observed in younger subjects. Gender differences in the pharmacokinetic properties of elderly subjects have been observed. Peak plasma concentrations were 114% and 54% higher in elderly females compared to elderly males following single and multiple twice-daily doses. [This interpretation was based on study results collected from two separate studies.] Plasma concentrations increase dose-dependently with the increase in doses after single oral dose and at steady state. No differences were observed in the volume of distribution values between elderly and younger subjects. As in younger subjects, elimination is mainly by renal excretion as unchanged drug in elderly subjects, although less drug is recovered from renal excretion in elderly subjects. Consistent with younger subjects, less than 5% of an administered dose was recovered in the urine as the desmethyl and N-oxide metabolites in the elderly. A longer plasma half-life of approximately 6.4 to 7.4 hours was observed in elderly subjects, compared with 4 to 5 hours for young subjects. Slower elimination of ofloxacin is observed in elderly subjects as compared with younger subjects which may be attributable to the reduced renal function and renal clearance observed in the elderly subjects. Because ofloxacin is known to be substantially excreted by the kidney, and elderly patients are more likely to have decreased renal function, dosage adjustment is necessary for elderly patients with impaired renal function as recommended for all patients. (see PRECAUTIONS, General and DOSAGE AND ADMINISTRATION ).
MICROBIOLOGY
Ofloxacin is a quinolone antimicrobial agent. The mechanism of action of ofloxacin and other fluoroquinolone antimicrobials involves inhibition of bacterial topoisomerase IV and DNA gyrase (both of which are type II topoisomerases), enzymes required for DNA replication, transcription, repair and recombination.
Ofloxacin has in vitro activity against a wide range of gram-negative and gram-positive microorganisms. Ofloxacin is often bactericidal at concentrations equal to or slightly greater than inhibitory concentrations.
Fluoroquinolones, including ofloxacin, differ in chemical structure and mode of action from aminoglycosides, macrolides and β-lactam antibiotics, including penicillins. Fluoroquinolones may, therefore, be active against bacteria resistant to these antimicrobials.
Resistance to ofloxacin due to spontaneous mutation in vitro is a rare occurrence (range: 10 -9 to 10 -11 ). Although cross-resistance has been observed between ofloxacin and some other fluoroquinolones, some microorganisms resistant to other fluoroquinolones may be susceptible to ofloxacin.
Ofloxacin has been shown to be active against most strains of the following microorganisms both in vitro and in clinical infections as described in the INDICATIONS AND USAGE section:
Aerobic Gram-Positive Microorganisms
Staphylococcus aureus (methicillin-susceptible strains)
Streptococcus pneumoniae (penicillin-susceptible strains)
Streptococcus pyogenes
Aerobic Gram-Negative Microorganisms
Citrobacter (diversus) koseri
Enterobacter aerogenes
Escherichia coli
Haemophilus influenzae
Klebsiella pneumoniae
Neisseria gonorrhoeae
Proteus mirabilis
Pseudomonas aeruginosa
As with other drugs in this class, some strains of Pseudomonas aeruginosa may develop resistance fairly rapidly during treatment with ofloxacin.
Other Microorganisms
Chlamydia trachomatis
The following in vitro data are available, but their clinical significance is unknown.
Ofloxacin exhibits in vitro minimum inhibitory concentrations (MIC values) of 2 mcg/mL or less against most (≥ 90%) strains of the following microorganisms; however, the safety and effectiveness of ofloxacin in treating clinical infections due to these microorganisms have not been established in adequate and well-controlled trials.
Aerobic Gram-Positive Microorganisms
Staphylococcus epidermidis (methicillin-susceptible strains)
Staphylococcus saprophyticus
Streptococcus pneumoniae (penicillin-resistant strains)
Aerobic Gram-Negative Microorganisms
Acinetobacter calcoaceticus
Bordetella pertussis
Citrobacter freundii
Enterobacter cloacae
Haemophilus ducreyi
Klebsiella oxytoca
Moraxella catarrhalis
Morganella morganii
Proteus vulgaris
Providencia rettgeri
Providencia stuartii
Serratia marcescens
Anaerobic Microorganisms
Clostridium perfringes
Other Microorganisms
Chlamydia pneumoniae
Gardnerella vaginalis
Legionella pneumophila
Mycoplasma hominis
Mycoplasma pneumoniae
Ureaplasma urealyticum
Ofloxacin is not active against Treponema pallidum (see WARNINGS ).
Susceptibility Tests
Dilution Techniques
Quantitative methods are used to determine antimicrobial minimum inhibitory concentrations (MIC values). These MIC values provide estimates of the susceptibility of bacteria to antimicrobial compounds. The MIC values should be determined using a standardized procedure. Standardized procedures are based on a dilution method 1,3 (broth or agar) or equivalent with standardized inoculum concentrations and standardized concentrations of ofloxacin powder. The MIC values should be interpreted according to the following criteria:
For testing Enterobacteriaceae , methicillin-susceptible Staphylococcus aureus , and Pseudomonas aeruginosa :
| MIC (mcg/mL) | Interpretation |
| ≤ 2 | Susceptible (S) |
| 4 | Intermediate (I) |
| ≥ 8 | Resistant (R) |
For testing Haemophilus influenzae: a
| MIC (mcg/mL) | Interpretation |
| ≤ 2 | Susceptible (S) |
a This interpretive standard is applicable only to broth microdilution susceptibility tests with Haemophilus influenzae using Haemophilus Test Medium. 1,3
The current absence of data on resistant strains precludes defining any results other than “Susceptible.” Strains yielding MIC results suggestive of a “nonsusceptible” category should be submitted to a reference laboratory for further testing. For testing Neisseria gonorrhoeae : b
| MIC (mcg/mL) | Interpretation |
| < 0.25 | Susceptible (S) |
| 0.5 to 1 | Intermediate (I) |
| > 2 | Resistant (R) |
b These interpretive standards are applicable only to agar dilution tests using GC agar base and 1% defined growth supplement incubated in 5% CO 2 . For testing Streptococcus pneumoniae and Streptococcus pyogenes : c
| MIC (mcg/mL) | Interpretation |
| ≤ 2 | Susceptible (S) |
| 4 | Intermediate (I) |
| ≥ 8 | Resistant (R) |
c These interpretive standards are applicable only to broth microdilution susceptibility tests using cation-adjusted Mueller-Hinton broth with 2 to 5% lysed horse blood.
A report of “Susceptible” indicates that the pathogen is likely to be inhibited if the antimicrobial compound in the blood reaches the concentration usually achievable. A report of “Intermediate” indicates that the result should be considered equivocal, and, if the microorganism is not fully susceptible to alternative, clinically feasible drugs, the test should be repeated. This category implies possible clinical applicability in body sites where the drug is physiologically concentrated or in situations where a high dosage of drug can be used. This category also provides a buffer zone which prevents small uncontrolled technical factors from causing major discrepancies in interpretation. A report of “Resistant” indicates that the pathogen is not likely to be inhibited if the antimicrobial compound in the blood reaches the concentration usually achievable; other therapy should be selected.
Standardized susceptibility test procedures require the use of laboratory control microorganisms to control the technical aspects of the laboratory procedures. Standard ofloxacin powder should provide the following MIC values:
| Microorganism | MIC Range (mcg/mL) | |
| Escherichia coli | ATCC 25922 | 0.015 to 0.12 |
| Haemophilus influenzae | ATCC 49247 d | 0.016 to 0.06 |
| Neisseria gonorrhoeae | ATCC 49226 e | 0.004 to 0.016 |
| Pseudomonas aeruginosa | ATCC 27853 | 1 to 8 |
| Staphylococcus aureus | ATCC 29213 | 0.12 to 1 |
| Streptococcus pneumoniae | ATCC 49619 f | 1 to 4 |
d This quality control range is applicable only to H. influenzae ATCC 49247 tested by a microdilution procedure using Haemophilus Test Medium (HTM). 1,3
e This quality control range is applicable only to N. gonorrhoeae ATCC 49226 tested by an agar dilution procedure using GC agar base with 1% defined growth supplement incubated in 5% CO 2 .
f This quality control range is applicable only to S. pneumoniae ATCC 49619 tested by a microdilution procedure using cation-adjusted Mueller-Hinton broth with 2 to 5% lysed horse blood.
DIffusion Techniques
Quantitative methods that require measurement of zone diameters also provide reproducible estimates of the susceptibility of bacteria to antimicrobial compounds. One such standardized procedure 2 requires the use of standardized inoculum concentrations. This procedure uses paper disks impregnated with 5 mcg ofloxacin to test the susceptibility of microorganisms to ofloxacin.
Reports from the laboratory providing results of the standard single-disk susceptibility test with a 5 mcg ofloxacin disk should be interpreted according to the following criteria:
For testing Enterobacteriaceae , methicillin-susceptible Staphylococcus aureus , and Pseudomonas aeruginosa :
| Zone Diameter (mm) | Interpretation |
| ≥ 16 | Susceptible (S) |
| 13 to 15 | Intermediate (I) |
| ≤ 12 | Resistant (R) |
For testing Haemophilus influenzae: g
| Zone Diameter (mm) | Interpretation |
| ≥ 16 | Susceptible (S) |
g This zone diameter standard is applicable only to disk diffusion tests with Haemophilus influenzae using Haemophilus Test Medium (HTM) 2 incubated in 5% CO 2 .
The current absence of data on resistant strains precludes defining any results other than “Susceptible.” Strains yielding zone diameter results suggestive of a “nonsusceptible” category should be submitted to a reference laboratory for further testing.
For testing Neisseria gonorrhoeae : h
| Zone Diameter (mm) | Interpretation |
| ≥ 31 | Susceptible (S) |
| 25 to 30 | Intermediate (I) |
| ≤ 24 | Resistant (R) |
h These zone diameter standards are applicable only to disk diffusion tests using GC agar base and 1% defined growth supplement incubated in 5% CO 2 .
For testing Streptococcus pneumoniae and Streptococcus pyogenes : i
| Zone Diameter (mm) | Interpretation |
| ≥ 16 | Susceptible (S) |
| 13 to 15 | Intermediate (I) |
| ≤ 12 | Resistant (R) |
i These zone diameter standards are applicable only to disk diffusion tests performed using Mueller-Hinton agar supplemented with 5% defibrinated sheep blood and incubated in 5% CO 2 .
Interpretation should be as stated above for results using dilution techniques. Interpretation involves correlation of the diameter obtained in the disk test with the MIC for ofloxacin.
As with standardized dilution techniques, diffusion methods require the use of laboratory control microorganisms that are used to control the technical aspects of the laboratory procedures. For the diffusion technique, the 5 mcg ofloxacin disk should provide the following zone diameters in these laboratory quality control strains:
| Microorganism | Zone Diameter (mm) | |
| Escherichia coli | ATCC 25922 | 29 to 33 |
| Haemophilus influenzae | ATCC 49247 j | 31 to 40 |
| Neisseria gonorrhoeae | ATCC 49226 k | 43 to 51 |
| Pseudomonas aeruginosa | ATCC 27853 | 17 to 21 |
| Staphylococcus aureus | ATCC 25923 | 24 to 28 |
| Streptococcus pneumoniae | ATCC 49619 l | 16 to 21 |
j This quality control range is applicable only to H. influenzae ATCC 49247 tested by a disk diffusion procedure using Haemophilus Test Medium (HTM) 2 incubated in 5% CO 2 .
k This quality control range is applicable only to N. gonorrhoeae ATCC 49226 tested by a disk diffusion procedure using GC agar base with 1% defined growth supplement incubated in 5% CO 2 .
l This quality control range is applicable only to S. pneumoniae ATCC 49619 tested by a disk diffusion procedure using Mueller-Hinton agar supplemented with 5% defibrinated sheep blood and incubated in 5% CO 2 .
HOW SUPPLIED
Ofloxacin tablets USP, 200 mg are available as light yellow to yellow, oval, biconvex, film coated tablets, imprinted with “C213” in blue ink on one side and plain on the other side. They are available in bottles of 50, 100 and 500 tablets..
Bottles of 50 (NDC 71209-097-02)
Bottles of 100 (NDC 71209-097-05)
Bottles of 500 (NDC 71209-097-10)
Ofloxacin tablets USP, 300 mg are available as white to off white, oval, biconvex, film coated tablets, imprinted with “C212” in blue ink on one side and plain on the other side.. They are available in bottles of 50, 100 and 500 tablets.
Bottles of 50 (NDC 71209-098-02)
Bottles of 100 (NDC 71209-098-05)
Bottles of 500 (NDC 71209-098-10)
Ofloxacin tablets USP, 400 mg are available as yellow to dark yellow, oval, biconvex, film coated tablets, imprinted with ‘C211’ in blue ink on one side and plain on other side. They are available in bottles of 50, 100 and 500 tablets.
Bottles of 50 (NDC 71209-099-02)
Bottles of 100 (NDC 71209-099-05)
Bottles of 500 (NDC 71209-099-10)
Store at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature]. Dispense in a tight, light-resistant container as defined in the USP, with a child-resistant closure (as required) KEEP THIS AND ALL MEDICATIONS OUT OF THE REACH OF CHILDREN.