Get your patient on Pentoxifylline - Pentoxifylline tablet, Extended Release (Pentoxifylline)
Pentoxifylline - Pentoxifylline tablet, Extended Release prescribing information
INDICATIONS AND USAGE
Pentoxifylline Extended-Release Tablets are indicated for the treatment of patients with intermittent claudication on the basis of chronic occlusive arterial disease of the limbs. Pentoxifylline Extended-Release Tablets can improve function and symptoms but is not intended to replace more definitive therapy, such as surgical bypass, or removal of arterial obstructions when treating peripheral vascular disease.
DOSAGE AND ADMINISTRATION
The usual dosage of pentoxifylline in extended-release tablet form is one tablet (400 mg) three times a day with meals.
While the effect of pentoxifylline may be seen within 2 to 4 weeks, it is recommended that treatment be continued for at least 8 weeks. Efficacy has been demonstrated in double-blind clinical studies of 6 months duration.
Digestive and central nervous system side effects are dose related. If patients develop these effects it is recommended that the dosage be lowered to one tablet twice a day (800 mg/day). If side effects persist at this lower dosage, the administration of pentoxifylline should be discontinued.
In patients with severe renal impairment (creatinine clearance below 30 mL/min) reduce dose to 400 mg once a day.
Dosing information cannot be provided for patients with hepatic impairment.
CONTRAINDICATIONS
Pentoxifylline Extended-Release Tablets should not be used in patients with recent cerebral and/or retinal hemorrhage or in patients who have previously exhibited intolerance to this product or methylxanthines such as caffeine, theophylline, and theobromine.
ADVERSE REACTIONS
Clinical trials were conducted using either extended-release pentoxifylline tablets for up to 60 weeks or immediate-release pentoxifylline capsules for up to 24 weeks. Dosage ranges in the tablet studies were 400 mg bid to tid and in the capsule studies, 200 to 400 mg tid. The table summarizes the incidence (in percent) of adverse reactions considered drug related, as well as the numbers of patients who received extended-release pentoxifylline tablets, immediate-release pentoxifylline capsules, or the corresponding placebos. The incidence of adverse reactions was higher in the capsule studies (where dose related increases were seen in digestive and nervous system side effects) than in the tablet studies. Studies with the capsule include domestic experience, whereas studies with the extended-release tablets were conducted outside the U.S.
The table indicates that in the tablet studies few patients discontinued because of adverse effects.
INCIDENCE (%) OF SIDE EFFECTS | ||||
Extended-Release Tablets | Immediate-Release Capsules | |||
Commercially Available | Used only for Controlled Clinical Trials | |||
Pentoxifylline | Placebo | Pentoxifylline | Placebo | |
(Numbers of Patients at Risk) | (321) | (128) | (177) | (138) |
Discontinued for Side Effect | 3.1 | 0 | 9.6 | 7.2 |
CARDIOVASCULAR SYSTEM | ||||
Angina/Chest Pain | 0.3 | 1.1 | 2.2 | |
Arrhythmia/Palpitation | 1.7 | 0.7 | ||
Flushing | 2.3 | 0.7 | ||
DIGESTIVE SYSTEM | ||||
Abdominal Discomfort | 4.0 | 1.4 | ||
Belching/Flatus/Bloating | 0.6 | 9.0 | 3.6 | |
Diarrhea | 3.4 | 2.9 | ||
Dyspepsia | 2.8 | 4.7 | 9.6 | 2.9 |
Nausea | 2.2 | 0.8 | 28.8 | 8.7 |
Vomiting | 1.2 | 4.5 | 0.7 | |
NERVOUS SYSTEM | ||||
Agitation/Nervousness | 1.7 | 0.7 | ||
Dizziness | 1.9 | 3.1 | 11.9 | 4.3 |
Drowsiness | 1.1 | 5.8 | ||
Headache | 1.2 | 1.6 | 6.2 | 5.8 |
Insomnia | 2.3 | 2.2 | ||
Tremor | 0.3 | 0.8 | ‑ | |
Blurred Vision | 2.3 | 1.4 | ||
Pentoxifylline has been marketed in Europe and elsewhere since 1972. In addition to the above symptoms, the following have been reported spontaneously since marketing or occurred in other clinical trials with an incidence of less than 1%; the causal relationship was uncertain:
- Cardiovascular - dyspnea, edema, hypotension. Digestive - anorexia, cholecystitis, constipation, dry mouth/thirst. Nervous - anxiety, confusion, depression, seizures, aseptic meningitis. Respiratory - epistaxis, flu-like symptoms, laryngitis, nasal congestion. Skin and Appendages - brittle fingernails, pruritus, rash, urticaria, angioedema. Special Senses - blurred vision, conjunctivitis, earache, scotoma. Miscellaneous - bad taste, excessive salivation, leukopenia, malaise, sore throat/swollen neck glands, weight change.
A few rare events have been reported spontaneously worldwide since marketing in 1972. Although they occurred under circumstances in which a causal relationship with pentoxifylline could not be established, they are listed to serve as information for physicians. Cardiovascular — angina, arrhythmia, tachycardia. Digestive — hepatitis, jaundice, cholestasis, increased liver enzymes; and Hemic and Lymphatic — decreased serum fibrinogen, pancytopenia, aplastic anemia, leukemia, purpura, thrombocytopenia. Immune system disorders — anaphylactic reaction, anaphylactoid reaction, anaphylactic shock.
To report SUSPECTED ADVERSE REACTIONS, contact ANI Pharmaceuticals, Inc. at 1-800-308-6755 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
Bleeding has been reported in patients treated with pentoxifylline with or without concomitant NSAIDs, anticoagulants, or platelet aggregation inhibitors.
Increased prothrombin time has been reported in patients concomitantly treated with pentoxifylline and vitamin K antagonists. Monitoring of anticoagulant activity in these patients is recommended when pentoxifylline is introduced or the dose is changed.
Concomitant administration of pentoxifylline and theophylline-containing drugs leads to increased theophylline levels and theophylline toxicity in some individuals. Monitor theophylline levels when starting pentoxifylline or changing dose.
Concomitant administration of strong CYP1A2 inhibitors (including e.g. ciprofloxacin or fluvoxamine) may increase the exposure to pentoxifylline (see ADVERSE REACTIONS ).
Pentoxifylline has been used concurrently with antihypertensive drugs, beta blockers, digitalis, diuretics, and antiarrhythmics, without observed problems. Small decreases in blood pressure have been observed in some patients treated with pentoxifylline; periodic systemic blood pressure monitoring is recommended for patients receiving concomitant antihypertensive therapy. If indicated, dosage of the antihypertensive agents should be reduced.
Postmarketing cases of increased anticoagulant activity have been reported in patients concomitantly treated with pentoxifylline and vitamin K antagonists. Monitoring of anticoagulant activity in these patients is recommended when pentoxifylline is introduced or the dose is changed.
Concomitant administration with cimetidine is reported to increase the average steady state plasma concentration of pentoxifylline (~25%) and the Metabolite I (~30%).
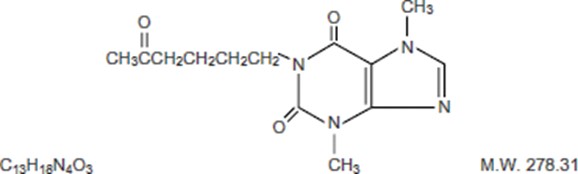
DESCRIPTION
Pentoxifylline Extended-Release Tablets USP for oral administration contain 400 mg of the active drug and the following inactive ingredients: hypromellose, povidone, talc, magnesium stearate, maltodextrin, polyethylene glycol, titanium dioxide, triacetin, D&C yellow #10 aluminum lake and FD&C yellow #6 in an extended-release formulation. Pentoxifylline is a tri-substituted xanthine derivative designated chemically as 3,7-Dihydro-3,7-dimethyl-1-(5-oxohexyl)-1 H ‑purine-2,6-dione that, unlike theophylline, is a hemorrheologic agent, i.e. an agent that affects blood viscosity. Pentoxifylline is soluble in water and ethanol, and sparingly soluble in toluene.
The chemical structure is:
Pentoxifylline Extended-Release Tablets USP comply with USP dissolution test 7.
CLINICAL PHARMACOLOGY
Mode of Action
Pentoxifylline and its metabolites improve the flow properties of blood by decreasing its viscosity. In patients with chronic peripheral arterial disease, this increases blood flow to the affected microcirculation and enhances tissue oxygenation. The precise mode of action of pentoxifylline and the sequence of events leading to clinical improvement are still to be defined. Pentoxifylline administration has been shown to produce dose-related hemorrheologic effects, lowering blood viscosity, and improving erythrocyte flexibility. Leukocyte properties of hemorrheologic importance have been modified in animal and in vitro human studies. Pentoxifylline has been shown to increase leukocyte deformability and to inhibit neutrophil adhesion and activation. Tissue oxygen levels have been shown to be significantly increased by therapeutic doses of pentoxifylline in patients with peripheral arterial disease.
Pharmacokinetics and Metabolism
After oral administration in aqueous solution pentoxifylline is almost completely absorbed. It undergoes a first-pass effect and the various metabolites appear in plasma very soon after dosing. Peak plasma levels of the parent compound and its metabolites are reached within 1 hour. The major metabolites are Metabolite I (1-[5-hydroxyhexyl]-3,7-dimethylxanthine) and Metabolite V (1-[3-carboxypropyl]-3,7-dimethylxanthine), and plasma levels of these metabolites are 5 and 8 times greater, respectively, than pentoxifylline.
Following oral administration of aqueous solutions containing 100 to 400 mg of pentoxifylline, the pharmacokinetics of the parent compound and Metabolite I are dose-related and not proportional (non-linear), with half-life and area under the blood-level time curve (AUC) increasing with dose. The elimination kinetics of Metabolite V are not dose-dependent. The apparent plasma half-life of pentoxifylline varies from 0.4 to 0.8 hours and the apparent plasma half-lives of its metabolites vary from 1 to 1.6 hours. There is no evidence of accumulation or enzyme induction (Cytochrome P450) following multiple oral doses.
Excretion is almost totally urinary; the main biotransformation product is Metabolite V. Essentially no parent drug is found in the urine. Despite large variations in plasma levels of parent compound and its metabolites, the urinary recovery of Metabolite V is consistent and shows dose proportionality. Less than 4% of the administered dose is recovered in feces. Food intake shortly before dosing delays absorption of an immediate-release dosage form but does not affect total absorption. The pentoxifylline AUC was increased and elimination rate decreased in an older population (60-68 years, n=6) compared to younger individuals (22-30 years, n=6) (see PRECAUTIONS, Geriatric Use ).
After administration of the 400 mg Pentoxifylline Extended-Release Tablet, plasma levels of the parent compound and its metabolites reach their maximum within 2 to 4 hours and remain constant over an extended period of time. Coadministration of Pentoxifylline Extended-Release Tablets with meals resulted in an increase in mean AUC and C max of about 1.1 and 1.3 fold for pentoxifylline, respectively. C max for Metabolite I also increased about1.2 fold. The extended release of pentoxifylline from the tablet eliminates peaks and troughs in plasma levels for improved gastrointestinal tolerance.
Patients with Hepatic Impairment
In patients with mild to moderate liver impairment AUC and C max of pentoxifylline increased 6.5 and 7.5 fold, respectively, after a single 400 mg dose of Pentoxifylline Extended-Release Tablets. AUC and C max of the active Metabolite I also increased 6.9 and 8.2 fold, respectively, in hepatic impaired subjects.
Pentoxifylline Extended-Release Tablets have not been studied in patients with severe hepatic failure.
Patients with Renal Impairment
In patients with mild, moderate, or severe renal impairment the exposure to pentoxifylline and its active Metabolite I are not increased. In contrast, AUC 0-tss and C max of the active Metabolite V in patients with mild to moderate renal impairment increased 2.4 and 2.1 fold, respectively, with a 400 mg TID regimen of Pentoxifylline Extended-Release Tablets. In severe renal impairment AUC 0-tss and C max of the active Metabolite V increased 12.9 and 10.6 fold, respectively, with a 400 mg Pentoxifylline Extended-Release Tablet TID regimen. The increase in exposure to Metabolite V is only slightly smaller in both renal impairment groups if Pentoxifylline Extended-Release Tablets are administered BID.
HOW SUPPLIED
Pentoxifylline Extended-Release Tablets USP, 400 mg are available for oral administration as yellow, oblong, film-coated tablets, embossed with “N668” on one side and plain on the other. They are supplied in bottles of 100 (NDC 70954-668-10) and bottles of 500 (NDC 70954-668-20).
Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature].
Dispense in a tight, light-resistant container as defined in the USP.
Rx only
Distributed by: ANI Pharmaceuticals, Inc. Baudette, MN 56623 
Issued: 04/2023 LB4581-00