Get your patient on Phenoxybenzamine Hydrochloride - Phenoxybenzamine Hydrochloride capsule (Phenoxybenzamine Hydrochloride)
Phenoxybenzamine Hydrochloride - Phenoxybenzamine Hydrochloride capsule prescribing information
INDICATIONS & USAGE
Phenoxybenzamine Hydrochloride Capsules are indicated in the treatment of pheochromocytoma, to control episodes of hypertension and sweating. If tachycardia is excessive, it may be necessary to use a beta -blocking agent concomitantly.
DOSAGE & ADMINISTRATION
The dosage should be adjusted to fit the needs of each patient. Small initial doses should be slowly increased until the desired effect is obtained or the side effects from blockade become troublesome. After each increase, the patient should be observed on that level before instituting another increase. The dosage should be carried to a point where symptomatic relief and/or objective improvement are obtained, but not so high that the side effects from blockade become troublesome.
Initially, 10 mg of phenoxybenzamine hydrochloride twice a day. Dosage should be increased every other day, usually to 20 to 40 mg 2 or 3 times a day, until an optimal dosage is obtained, as judged by blood pressure control.
Long-term use of phenoxybenzamine is not recommended (see PRECAUTIONS Carcinogenesis and Mutagenesis).
CONTRAINDICATIONS
Conditions where a fall in blood pressure may be undesirable; hypersensitivity to the drug or any of its components.
ADVERSE REACTIONS
The following adverse reactions have been observed, but there are insufficient data to support an estimate of their frequency.
Autonomic Nervous System•: Postural hypotension, tachycardia, inhibition of ejaculation, nasal congestion, miosis.
•These so-called "side effects" are actually evidence of adrenergic blockade and vary according to the degree of blockade.
Miscellaneous: Gastrointestinal irritation, drowsiness, fatigue.
To report SUSPECTED ADVERSE REACTIONS, contact Novitium Pharma, LLC at 1-855-204-1431 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
Phenoxybenzamine hydrochloride may interact with compounds that stimulate both alpha - and beta -adrenergic receptors (i.e., epinephrine) to produce an exaggerated hypotensive response and tachycardia. (See WARNINGS.)
Phenoxybenzamine hydrochloride blocks hyperthermia production by levarterenol, and blocks hypothermia production by reserpine.
DESCRIPTION
Each Phenoxybenzamine Hydrochloride Capsule, USP 10 mg is a size #3 capsule with red transparent cap and body, imprinted “365” on cap and “novitium 10 mg” on body with grey ink, filled with white to off white powder containing 10 mg of Phenoxybenzamine Hydrochloride USP and the following inactive ingredients: lactose and talc.
Each of the empty hard gelatin capsule contains gelatin, purified water, sodium lauryl sulfate, D&C Red 33, FD&C Yellow 6 and FD&C Red 3.
The imprinting ink contains the following: shellac, dehydrated alcohol, isopropyl alcohol, butyl alcohol, propylene glycol, strong ammonia solution, potassium hydroxide, titanium dioxide, black iron oxide and purified water.
Phenoxybenzamine Hydrochloride is N-(2-Chloroethyl)-N-(1-methyl-2-phenoxyethyl) benzylamine hydrochloride:
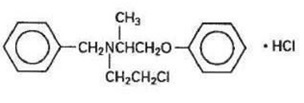
Phenoxybenzamine hydrochloride is a white to off-white powder with a molecular weight of 340.3, which melts between 136° and 141°C. It is soluble in water, alcohol and chloroform; insoluble in ether.
CLINICAL PHARMACOLOGY
Phenoxybenzamine hydrochloride is a long-acting, adrenergic, alpha- receptor-blocking agent, which can produce and maintain "chemical sympathectomy" by oral administration. It increases blood flow to the skin, mucosa and abdominal viscera, and lowers both supine and erect blood pressures. It has no effect on the parasympathetic system.
Twenty to 30 percent of orally administered phenoxybenzamine appears to be absorbed in the active form. 1
The half-life of orally administered phenoxybenzamine hydrochloride is not known; however, the half-life of intravenously administered drug is approximately 24 hours. Demonstrable effects with intravenous administration persist for at least 3 to 4 days, and the effects of daily administration are cumulative for nearly a week. 1
HOW SUPPLIED
Phenoxybenzamine Hydrochloride Capsules, USP 10 mg are supplied as size #3 capsule with red transparent cap and body, imprinted “365” on cap and “novitium 10 mg” on body with grey ink, filled with white to off white powder in bottles of 100 (NDC 70954-365-10).