Get your patient on Pilocarpine Hydrochloride - Pilocarpine Hydrochloride Ophthalmic Solution solution/ Drops (Pilocarpine Hydrochloride Ophthalmic Solution)
Pilocarpine Hydrochloride - Pilocarpine Hydrochloride Ophthalmic Solution solution/ Drops prescribing information
INDICATIONS AND USAGE
Pilocarpine hydrochloride ophthalmic solution, is indicated for the:
Reduction of Elevated Intraocular Pressure (IOP) in Patients with Open-Angle Glaucoma or Ocular Hypertension
Management of Acute Angle-Closure Glaucoma
Prevention of Postoperative Elevated IOP Associated with Laser Surgery
Induction of Miosis
DOSAGE AND ADMINISTRATION
- Instill one drop in the eye(s) up to four times daily (2).
Reduction of Elevated Intraocular Pressure (IOP) in Patients with Open-Angle Glaucoma or Ocular Hypertension
One drop of pilocarpine hydrochloride ophthalmic solution 1%, 2% or 4% should be applied topically in the eye(s) up to four times daily. Pilocarpine-naïve patients should be started on the 1% concentration as higher concentrations are often not tolerated initially. The frequency of instillation and concentration of pilocarpine hydrochloride ophthalmic solution are determined by the severity of the elevated intraocular pressure and miotic response of the patient.
To limit systemic exposure to pilocarpine, patients may be instructed to perform punctal occlusion for 2 minutes after instillation of pilocarpine hydrochloride ophthalmic solution.
Management of Acute Angle-Closure Glaucoma
Prior to pilocarpine hydrochloride ophthalmic solution use, treatment with secretory suppressants and hyperosmotic agents may be needed to lower IOP below 50 mmHg and relieve iris ischemia. For initial management of acute angle-closure glaucoma, one drop of pilocarpine hydrochloride ophthalmic solution 1% or 2% may be applied topically in the eye(s) up to three times over a 30-minute period.
If laser iridoplasty or iridomy is used to break the attack, one drop of pilocarpine hydrochloride ophthalmic solution 4% should be administered prior to the procedure. Following laser iridoplasty, one drop of pilocarpine hydrochloride ophthalmic solution 1% should be administered four times daily until an iridotomy can be performed.
Prevention of Postoperative Elevated IOP Associated with Laser Surgery
One drop of pilocarpine hydrochloride ophthalmic solution 1%, 2% or 4% (or two drops administered five minutes apart) should be applied topically in the eye(s) 15 to 60 minutes prior to surgery.
Induction of Miosis
One drop of pilocarpine hydrochloride ophthalmic solution 1%, 2% or 4% (or two drops administered five minutes apart) should be applied topically in the eye(s).
Use with Other Topical Ophthalmic Medications
Pilocarpine hydrochloride ophthalmic solution may be used in combination with beta-blockers, carbonic anhydrase inhibitors, sympathomimetics or hyperosmotic agents. If more than one topical ophthalmic drug is being used, the drugs should be administered at least five (5) minutes apart.
Use in Pediatric Patients
In children under 2 years of age, one drop of pilocarpine hydrochloride ophthalmic solution 1% should be applied topically in the eye(s) three times daily. Children 2 years of age and over should be dosed as for adults. For the induction of miosis prior to goniotomy or trabeculotomy in children, one drop of pilocarpine hydrochloride ophthalmic solution 1% or 2% should be applied topically in the eye 15 to 60 minutes prior to surgery.
DOSAGE FORMS AND STRENGTHS
Bottle filled with 15 mL of 1% (10 mg/mL), 2% (20 mg/mL) or 4% (40 mg/mL) pilocarpine hydrochloride sterile ophthalmic solution.
USE IN SPECIFIC POPULATIONS
Pregnancy
Pregnancy. Category C. Animal reproduction studies have not been conducted with pilocarpine hydrochloride. It is also not known whether pilocarpine hydrochloride can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. Pilocarpine hydrochloride ophthalmic solution should be given to a pregnant woman only if clearly needed.
Nursing Mothers
It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when pilocarpine hydrochloride ophthalmic solution is administered to a nursing woman.
Pediatric Use
Safety and effectiveness of pilocarpine hydrochloride ophthalmic solution in pediatric patients have been established.
Geriatric Use
No overall differences in safety or effectiveness have been observed between elderly and younger patients.
CONTRAINDICATIONS
None.
WARNINGS AND PRECAUTIONS
- Poor illumination: Exercise caution in night driving and other hazardous occupations in poor illumination (5.1).
- Pre-existing retinal disease: Rare cases of retinal detachment have been reported; a thorough examination of the retina including funduscopy is advised in all patients prior to the initiation of therapy (5.2).
- Iritis: Caution is advised in patients with iritis. (5.3)
- Congenital glaucoma: Caution is advised in pediatric patients with primary congenital glaucoma for control of IOP as cases of a paradoxical increase in IOP have been reported. (5.4)
Poor Illumination
Patients should be advised to exercise caution in night driving and other hazardous occupations in poor illumination. In addition, miotics may cause accommodative spasm. Patients should be advised not to drive or use machinery if vision is not clear.
Pre-existing Retinal Disease
As with all miotics, rare cases of retinal detachment have been reported when used in certain susceptible individuals and those with pre-existing retinal disease; therefore, a thorough examination of the retina including funduscopy is advised in all patients prior to the initiation of therapy.
Iritis
Pilocarpine hydrochloride ophthalmic solution is not recommended to be used when iritis is present.
Primary Congenital Glaucoma
Caution is advised when using pilocarpine hydrochloride ophthalmic solution in pediatric patients with primary congenital glaucoma for control of intraocular pressure (IOP) as cases of a paradoxical increase in IOP have been reported. In addition, the use of pilocarpine hydrochloride ophthalmic solution is not recommended in pediatric patients diagnosed with glaucoma secondary to anterior segment dysgenesis or uveitis (especially if uveitis is active).
Contact Lens Wear
Contact lens wearers should be advised to remove their lenses prior to the instillation of pilocarpine hydrochloride ophthalmic solution and to wait 10 minutes after dosing before reinserting their contact lenses.
ADVERSE REACTIONS
Clinical Studies Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety data described below reflect exposure in four controlled clinical trials of 90 days to 2 years duration in 317 patients diagnosed with open-angle glaucoma or ocular hypertension. In the four clinical trials, patients were treated with pilocarpine hydrochloride ophthalmic solution 2%, two to four times daily or with pilocarpine 1%, 1.75% or 2% in fixed combination with betaxolol 0.25%, two or three times daily. The most frequently reported adverse reactions occurring in ≥ 5% of patients in the pilocarpine 2% populations were: headache/browache, accommodative change, blurred vision, eye irritation, visual impairment (dim, dark, or "jumping" vision), and eye pain.
The adverse reaction profile reported for the use of pilocarpine hydrochloride ophthalmic solution in pediatric patients is comparable to that seen in adult patients.
DESCRIPTION
Pilocarpine hydrochloride ophthalmic solution is a cholinergic agonist prepared as a sterile topical ophthalmic solution. The active ingredient is represented by the chemical structure:
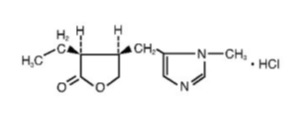
Established name: pilocarpine hydrochloride
Chemical name: 2(3 H )-furanone, 3-ethyldihydro-4-[(1-methyl-1 H -imidazol-5-yl)-methyl]- monohydrochloride, (3S- cis )-. Molecular Formula: C 11 H 16 N 2 O 2 • HCl Molecular Weight: 244.72.
Each mL of pilocarpine hydrochloride ophthalmic solution contains: Active: pilocarpine hydrochloride 1% (10 mg/mL), 2% (20 mg/mL), or 4% (40 mg/mL).
Preservative: benzalkonium chloride 0.01%.
Inactives: hypromellose 2910, boric acid, sodium citrate, sodium chloride (present in 1% only); hydrochloric acid and/or sodium hydroxide (to adjust pH); water for injection. Pilocarpine hydrochloride ophthalmic solution has a pH of 3.5 to 5.5 and an osmolality of 290 to 350 mOsm/kg (1% and 2% products) and 500 to 600 mOsm/kg (4% product).
CLINICAL PHARMACOLOGY
Mechanism of Action
Pilocarpine hydrochloride is a direct acting cholinergic parasympathomimetic agent which acts through direct stimulation of muscarinic receptors and smooth muscle such as the iris and secretory glands. Pilocarpine contracts the ciliary muscle, causing increased tension on the scleral spur and opening of the trabecular meshwork spaces to facilitate outflow of aqueous humor. Outflow resistance is reduced, lowering intraocular pressure (IOP). Pilocarpine also produces miosis through contraction of the iris sphincter muscle. Miosis relieves appositional angle narrowing and closure, which lowers IOP in certain types of angle-closure glaucoma.
Pharmacokinetics
Systemic exposure to pilocarpine was evaluated in 14 healthy subjects administered 2 drops of pilocarpine hydrochloride ophthalmic solution 4% to both eyes four times daily for eight days. A comparison of C max values on Days 5 and 8 indicated that pilocarpine concentrations in plasma reached steady-state following topical administration of pilocarpine hydrochloride ophthalmic solution 4%. The mean (SD) C max and AUC 0-last values on Day 8 were 3.7 (3.2) ng/mL and 7.7 (8.4) ng×hour/mL, respectively. The T max values on Day 8 ranged from 0.5 to 1 hour.
NONCLINICAL TOXICOLOGY
Carcinogenesis, Mutagenesis, Impairment of Fertility
There have been no long-term studies done using pilocarpine hydrochloride in animals to evaluate carcinogenic potential.
CLINICAL STUDIES
In clinical trials reported in the medical literature, pilocarpine ophthalmic solution reduced intraocular pressure (IOP) by 3-7 mmHg in patients with open-angle glaucoma. Pilocarpine ophthalmic solution has also been shown to be effective in the induction of miosis, in the prevention of postoperative elevated IOP, and in the management of acute angle-closure glaucoma.
HOW SUPPLIED/STORAGE AND HANDLING
Pilocarpine hydrochloride ophthalmic solution USP, 1%, 2% and 4% is clear colorless solution supplied sterile in natural LDPE bottles plugged with natural LDPE nozzle and green coloured HDPE cap.
15 mL in 15 mL bottles
1%: NDC 70069- 181 -01
2%: NDC 70069- 191 -01
4%: NDC 70069- 201 -01
STORAGE: Store between 15º to 25ºC (59º to 77ºF) and protect from freezing; [see USP Controlled Room Temperature].