Get your patient on Prednisolone Acetate - Prednisolone Acetate suspension/ Drops (Prednisolone Acetate)
Prednisolone Acetate - Prednisolone Acetate suspension/ Drops prescribing information
INDICATIONS AND USAGE
Prednisolone acetate ophthalmic suspension is indicated for the treatment of steroid-responsive inflammation of the palpebral and bulbar conjunctiva, cornea, and anterior segment of the globe.
DOSAGE AND ADMINISTRATION
Shake well before using. Instill one to two drops into the conjunctival sac two to four times daily. During the initial 24 to 48 hours, the dosing frequency may be increased if necessary. Care should be taken not to discontinue therapy prematurely.
If signs and symptoms fail to improve after 2 days, the patient should be re-evaluated ( see PRECAUTIONS ).
CONTRAINDICATIONS
Prednisolone acetate ophthalmic suspension is contraindicated in acute untreated purulent ocular infections, in most viral diseases of the cornea and conjunctiva including epithelial herpes simplex keratitis (dendritic keratitis), vaccinia, and varicella, and also in mycobacterial infection of the eye and fungal diseases of ocular structures.
Prednisolone acetate ophthalmic suspension is also contraindicated in individuals with known or suspected hypersensitivity to any of the ingredients of this preparation and to other corticosteroids.
ADVERSE REACTIONS
The following adverse reactions have been identified during use of Prednisolone acetate ophthalmic suspension. Because reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Adverse reactions include elevation of intraocular pressure (IOP) with possible development of glaucoma and infrequent optic nerve damage, posterior subcapsular cataract formation, and delayed wound healing.
The development of secondary ocular infection (bacterial, fungal, and viral) has occurred. Fungal and viral infections of the cornea are particularly prone to develop coincidentally with long-term applications of steroids. The possibility of fungal invasion should be considered in any persistent corneal ulceration where steroid treatment has been used ( see PRECAUTIONS ).
Other adverse reactions reported with the use of prednisolone acetate ophthalmic suspension include: allergic reactions; dysgeusia; eye pain; foreign body sensation; headache; pruritus; rash; transient burning and stinging upon instillation and other minor symptoms of ocular irritation; urticaria; and visual disturbance (blurry vision).
Keratitis, conjunctivitis, corneal ulcers, mydriasis, conjunctival hyperemia, loss of accommodation and ptosis have occasionally been reported following local use of corticosteroids. Corticosteroid-containing preparations have also been reported to cause acute anterior uveitis and perforation of the globe.
To report SUSPECTED ADVERSE REACTIONS, contact Lupin Pharmaceuticals, Inc at 1-800-399-2561 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DESCRIPTION
Prednisolone acetate ophthalmic suspension USP, 1% is a sterile, topical anti-inflammatory agent for ophthalmic use. Its chemical name is 11ß,17, 21-Trihydroxypregna-1,4-diene-3, 20-dione 21-acetate and it has the following structure:
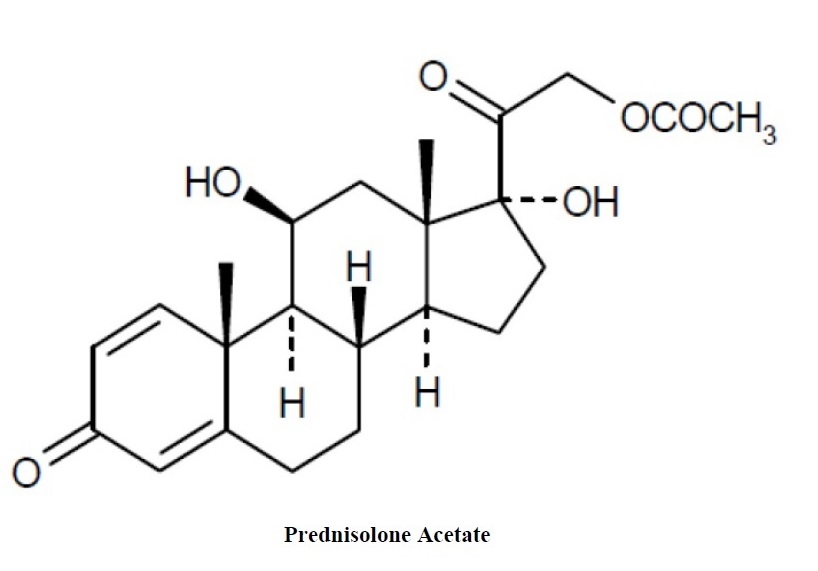
Each mL of prednisolone acetate ophthalmic suspension USP contains:
Active: prednisolone acetate (micronized) 1%
Inactive: benzalkonium chloride as preservative, boric acid, edetate disodium, hypromellose, polysorbate 80, sodium bisulfite, sodium chloride, sodium citrate and water for injection.
The pH during its shelf life ranges from 5.0 to 6.0.
CLINICAL PHARMACOLOGY
Prednisolone acetate is a glucocorticoid that, on the basis of weight, has 3 to 5 times the anti-inflammatory potency of hydrocortisone. Glucocorticoids inhibit the edema, fibrin deposition, capillary dilation, and phagocytic migration of the acute inflammatory response, as well as capillary proliferation, deposition of collagen, and scar formation.
HOW SUPPLIED
Prednisolone acetate ophthalmic suspension USP, 1% is supplied sterile in opaque white bottle with opaque white nozzle with pink cap as follows:
NDC 70748-332-02: 5 mL in 10 mL bottle
NDC 70748-332-03: 10 mL in 15 mL bottle
NDC 70748-332-04: 15 mL in 15 mL bottle
Storage: Store at up to 25°C (77°F). Protect from freezing. Store in an upright position.
LUPIN and the  are registered trademarks of Lupin Pharmaceuticals, Inc.
are registered trademarks of Lupin Pharmaceuticals, Inc.
Manufactured for:
Lupin Pharmaceuticals, Inc.
Naples, FL 34108
United States.
Manufactured by:
Lupin Limited
Pithampur (M. P.) - 454 775
India.
Revised: December 2024 ID#: 275716