Get your patient on Prometrium - Progesterone capsule (Progesterone)
Prometrium - Progesterone capsule prescribing information
INDICATIONS AND USAGE
PROMETRIUM ® (progesterone, USP) Capsules are indicated for use in the prevention of endometrial hyperplasia in nonhysterectomized postmenopausal women who are receiving conjugated estrogens tablets. They are also indicated for use in secondary amenorrhea.
DOSAGE AND ADMINISTRATION
Prevention of Endometrial Hyperplasia
PROMETRIUM Capsules should be given as a single daily dose at bedtime, 200 mg orally for 12 days sequentially per 28-day cycle, to a postmenopausal woman with a uterus who is receiving daily conjugated estrogens tablets.
Treatment of Secondary Amenorrhea
PROMETRIUM Capsules may be given as a single daily dose of 400 mg at bedtime for 10 days.
Some women may experience difficulty swallowing PROMETRIUM Capsules. For these women, PROMETRIUM Capsules should be taken with a glass of water while in the standing position.
CONTRAINDICATIONS
PROMETRIUM is contraindicated in women with any of the following conditions:
- Hypersensitivity to its ingredients. PROMETRIUM Capsules contain peanut oil and is contraindicated in patients allergic to peanuts.
- Abnormal genital bleeding of unknown etiology.
- Known, suspected, or history of breast cancer.
- Active deep vein thrombosis, pulmonary embolism or history of these conditions.
- Active arterial thromboembolic disease (for example, stroke and myocardial infarction), or a history of these conditions.
- Known liver dysfunction or disease.
ADVERSE REACTIONS
See WARNINGS and PRECAUTIONS .
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
In a multicenter, randomized, double-blind, placebo-controlled clinical trial, the effects of PROMETRIUM Capsules on the endometrium was studied in a total of 875 postmenopausal women. Table 7 lists adverse reactions greater than or equal to 2 percent of women who received cyclic PROMETRIUM Capsules 200 mg daily (12 days per calendar month cycle) with 0.625 mg conjugated estrogens or placebo.
| PROMETRIUM Capsules 200 mg with Conjugated Estrogens 0.625 mg | Placebo | |
|---|---|---|
| (n=178) | (n=174) | |
Headache | 31 | 27 |
Breast Tenderness | 27 | 6 |
Joint Pain | 20 | 29 |
Depression | 19 | 12 |
Dizziness | 15 | 9 |
Abdominal Bloating | 12 | 5 |
Hot Flashes | 11 | 35 |
Urinary Problems | 11 | 9 |
Abdominal Pain | 10 | 10 |
Vaginal Discharge | 10 | 3 |
Nausea / Vomiting | 8 | 7 |
Worry | 8 | 4 |
Chest Pain | 7 | 5 |
Diarrhea | 7 | 4 |
Night Sweats | 7 | 17 |
Breast Pain | 6 | 2 |
Swelling of Hands and Feet | 6 | 9 |
Vaginal Dryness | 6 | 10 |
Constipation | 3 | 2 |
Breast Carcinoma | 2 | <1 |
Breast Excisional Biopsy | 2 | <1 |
Cholecystectomy | 2 | <1 |
Effects on Secondary Amenorrhea
In a multicenter, randomized, double-blind, placebo-controlled clinical trial, the effects of PROMETRIUM ® (progesterone, USP) Capsules on secondary amenorrhea was studied in 49 estrogen-primed postmenopausal women. Table 8 lists adverse reactions greater than or equal to 5 percent of women who received PROMETRIUM Capsules or placebo.
Adverse Experience | PROMETRIUM Capsules 400 mg | Placebo |
n=25 | n=24 | |
Percentage (%) of Patients | ||
Fatigue | 8 | 4 |
Headache | 16 | 8 |
Dizziness | 24 | 4 |
Abdominal Distention (Bloating) | 8 | 8 |
Abdominal Pain (Cramping) | 20 | 13 |
Diarrhea | 8 | 4 |
Nausea | 8 | 0 |
Back Pain | 8 | 8 |
Musculoskeletal Pain | 12 | 4 |
Irritability | 8 | 4 |
Breast Pain | 16 | 8 |
Infection Viral | 12 | 0 |
Coughing | 8 | 0 |
In a multicenter, parallel-group, open label postmarketing dosing study consisting of three consecutive 28-day treatment cycles, 220 premenopausal women with secondary amenorrhea were randomized to receive daily conjugated estrogens therapy (0.625 mg conjugated estrogens) and PROMETRIUM Capsules, 300 mg per day (n=113) or PROMETRIUM Capsules, 400 mg per /day (n=107) for 10 days of each treatment cycle. Overall, the most frequently reported treatment-emergent adverse reactions, reported in greater than or equal to 5 percent of subjects, were nausea, fatigue, vaginal mycosis, nasopharyngitis, upper respiratory tract infection, headache, dizziness, breast tenderness, abdominal distension, acne, dysmenorrhea, mood swing, and urinary tract infection.
Postmarketing Experience:
The following additional adverse reactions have been reported with PROMETRIUM Capsules. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate the frequency or establish a causal relationship to drug exposure.
Genitourinary System: endometrial carcinoma, hypospadia, intra-uterine death, menorrhagia, menstrual disorder, metrorrhagia, ovarian cyst, spontaneous abortion.
Cardiovascular: circulatory collapse, congenital heart disease (including ventricular septal defect and patent ductus arteriosus), hypertension, hypotension, tachycardia.
Gastrointestinal: acute pancreatitis, cholestasis, cholestatic hepatitis, dysphagia, hepatic failure, hepatic necrosis, hepatitis, increased liver function tests (including alanine aminotransferase increased, aspartate aminotransferase increased, gamma-glutamyl transferase increased), jaundice, swollen tongue.
Skin: alopecia, pruritus, urticaria.
Eyes: blurred vision, diplopia, visual disturbance.
Central Nervous System: aggression, convulsion, depersonalization, depressed consciousness, disorientation, dysarthria, loss of consciousness, paresthesia, sedation, stupor, syncope (with and without hypotension), transient ischemic attack, suicidal ideation.
During initial therapy, a few women have experienced a constellation of many or all of the following symptoms: extreme dizziness and/or drowsiness, blurred vision, slurred speech, difficulty walking, loss of consciousness, vertigo, confusion, disorientation, feeling drunk, and shortness of breath.
Miscellaneous: abnormal gait, anaphylactic reaction, arthralgia, blood glucose increased, choking, cleft lip, cleft palate, difficulty walking, dyspnea, face edema, feeling abnormal, feeling drunk, hypersensitivity, asthma, muscle cramp, throat tightness, tinnitus, vertigo, weight decreased, weight increased.
DESCRIPTION
PROMETRIUM ® (progesterone, USP) Capsules contain micronized progesterone for oral administration. Progesterone has a molecular weight of 314.47 and a molecular formula of C 21 H 30 O 2 . Progesterone (pregn-4-ene-3, 20-dione) is a white or creamy white, odorless, crystalline powder practically insoluble in water, soluble in alcohol, acetone and dioxane and sparingly soluble in vegetable oils, stable in air, melting between 126° and 131°C. The structural formula is:
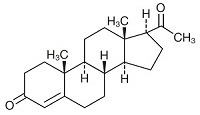
Progesterone is synthesized from a starting material from a plant source and is chemically identical to progesterone of human ovarian origin. PROMETRIUM Capsules are available in multiple strengths to afford dosage flexibility for optimum management. PROMETRIUM Capsules contain 100 mg or 200 mg micronized progesterone.
The inactive ingredients for PROMETRIUM Capsules 100 mg include: peanut oil NF, gelatin NF, glycerin USP, lecithin NF, titanium dioxide USP, FD&C Red No. 40, and D&C Yellow No. 10.
The inactive ingredients for PROMETRIUM Capsules 200 mg include: peanut oil NF, gelatin NF, glycerin USP, lecithin NF, titanium dioxide USP, D&C Yellow No. 10, and FD&C Yellow No. 6.
CLINICAL PHARMACOLOGY
PROMETRIUM Capsules are an oral dosage form of micronized progesterone which is chemically identical to progesterone of ovarian origin. The oral bioavailability of progesterone is increased through micronization.
Pharmacokinetics
A. Absorption
After oral administration of progesterone as a micronized soft-gelatin capsule formulation, maximum serum concentrations were attained within 3 hours. The absolute bioavailability of micronized progesterone is not known. Table 1 summarizes the mean pharmacokinetic parameters in postmenopausal women after five oral daily doses of PROMETRIUM Capsules 100 mg as a micronized soft-gelatin capsule formulation.
| Parameter | PROMETRIUM Capsules Daily Dose | ||
|---|---|---|---|
| 100 mg | 200 mg | 300 mg | |
C max (ng/mL) | 17.3 ± 21.9 Mean ± S.D. | 38.1 ± 37.8 | 60.6 ± 72.5 |
T max (hr) | 1.5 ± 0.8 | 2.3 ± 1.4 | 1.7 ± 0.6 |
AUC (0-10) (ng × hr/mL) | 43.3 ± 30.8 | 101.2 ± 66.0 | 175.7 ± 170.3 |
Serum progesterone concentrations appeared linear and dose proportional following multiple dose administration of PROMETRIUM ® (progesterone, USP) Capsules 100 mg over the dose range 100 mg per day to 300 mg per day in postmenopausal women. Although doses greater than 300 mg per day were not studied in females, serum concentrations from a study in male volunteers appeared linear and dose proportional between 100 mg per day and 400 mg per day. The pharmacokinetic parameters in male volunteers were generally consistent with those seen in postmenopausal women.
B. Distribution
Progesterone is approximately 96 percent to 99 percent bound to serum proteins, primarily to serum albumin (50 to 54 percent) and transcortin (43 to 48 percent).
C. Metabolism
Progesterone is metabolized primarily by the liver largely to pregnanediols and pregnanolones. Pregnanediols and pregnanolones are conjugated in the liver to glucuronide and sulfate metabolites. Progesterone metabolites which are excreted in the bile may be deconjugated and may be further metabolized in the intestine via reduction, dehydroxylation, and epimerization.
D. Excretion
The glucuronide and sulfate conjugates of pregnanediol and pregnanolone are excreted in the bile and urine. Progesterone metabolites are eliminated mainly by the kidneys. Progesterone metabolites which are excreted in the bile may undergo enterohepatic recycling or may be excreted in the feces.
E. Special Populations
The pharmacokinetics of PROMETRIUM Capsules have not been assessed in low body weight or obese patients.
Hepatic Insufficiency: The effect of hepatic impairment on the pharmacokinetics of PROMETRIUM Capsules has not been studied.
Renal Insufficiency: The effect of renal impairment on the pharmacokinetics of PROMETRIUM Capsules has not been studied.
F. Food–Drug Interaction
Concomitant food ingestion increased the bioavailability of PROMETRIUM Capsules relative to a fasting state when administered to postmenopausal women at a dose of 200 mg.
G. Drug Interactions
The metabolism of progesterone by human liver microsomes was inhibited by ketoconazole (IC 50 < 0.1 μM). Ketoconazole is a known inhibitor of cytochrome P450 3A4, hence these data suggest that ketoconazole or other known inhibitors of this enzyme may increase the bioavailability of progesterone. The clinical relevance of the in vitro findings is unknown.
Coadministration of conjugated estrogens and PROMETRIUM Capsules to 29 postmenopausal women over a 12-day period resulted in an increase in total estrone concentrations (C max 3.68 ng/mL to 4.93 ng/mL) and total equilin concentrations (C max 2.27 ng/mL to 3.22 ng/mL) and a decrease in circulating 17β estradiol concentrations (C max 0.037 ng/mL to 0.030 ng/mL). The half-life of the conjugated estrogens was similar with coadministration of PROMETRIUM Capsules. Table 2 summarizes the pharmacokinetic parameters.
| Conjugated Estrogens | Conjugated Estrogens plus PROMETRIUM Capsules | |||||
|---|---|---|---|---|---|---|
| Drug | C max (ng/mL) | T max (hr) | AUC (0-24h) (ng × h/mL) | C max (ng/mL) | T max (hr) | AUC (0-24h) (ng × h/mL) |
Estradiol | 0.037 ± 0.048 | 12.7 ± 9.1 | 0.676 ± 0.737 | 0.030 ± 0.032 | 17.32 ± 1.21 | 0.561 ± 0.572 |
Estrone Total Total estrogens is the sum of conjugated and unconjugated estrogen. | 3.68 ± 1.55 | 10.6 ± 6.8 | 61.3 ± 26.36 | 4.93 ± 2.07 | 7.5 ± 3.8 | 85.9 ± 41.2 |
Equilin Total | 2.27 ± 0.95 | 6.0 ± 4.0 | 28.8 ± 13.0 | 3.22 ± 1.13 | 5.3 ± 2.6 | 38.1 ± 20.2 |
CLINICAL STUDIES
Effects on the endometrium
In a randomized, double-blind clinical trial, 358 postmenopausal women, each with an intact uterus, received treatment for up to 36 months. The treatment groups were: PROMETRIUM ® (progesterone, USP) Capsules at the dose of 200 mg per day for 12 days per 28-day cycle in combination with conjugated estrogens 0.625 mg per day (n=120); conjugated estrogens 0.625 mg per day only (n=119); or placebo (n=119). The subjects in all three treatment groups were primarily Caucasian women (87 percent or more of each group). The results for the incidence of endometrial hyperplasia in women receiving up to 3 years of treatment are shown in Table 3. A comparison of the PROMETRIUM Capsules plus conjugated estrogens treatment group to the conjugated estrogens only group showed a significantly lower rate of hyperplasia (6 percent combination product versus 64 percent estrogen alone) in the PROMETRIUM Capsules plus conjugated estrogens treatment group throughout 36 months of treatment.
| Endometrial Diagnosis | Treatment Group | |||||
|---|---|---|---|---|---|---|
| Conjugated Estrogens 0.625 mg + PROMETRIUM Capsules 200 mg (cyclical) | Conjugated Estrogens 0.625 mg (alone) | Placebo | ||||
| Number of patients | % of patients | Number of patients | % of patients | Number of patients | % of patients | |
| n=117 | n=115 | n=116 | ||||
HYPERPLASIA Most advanced result to least advanced result: Adenocarcinoma > atypical hyperplasia > complex hyperplasia > simple hyperplasia | 7 | 6 | 74 | 64 | 3 | 3 |
Adenocarcinoma | 0 | 0 | 0 | 0 | 1 | 1 |
Atypical hyperplasia | 1 | 1 | 14 | 12 | 0 | 0 |
Complex hyperplasia | 0 | 0 | 27 | 23 | 1 | 1 |
Simple hyperplasia | 6 | 5 | 33 | 29 | 1 | 1 |
The times to diagnosis of endometrial hyperplasia over 36 months of treatment are shown in Figure 1. This figure illustrates graphically that the proportion of patients with hyperplasia was significantly greater for the conjugated estrogens group (64 percent) compared to the conjugated estrogens plus PROMETRIUM Capsules group (6 percent).
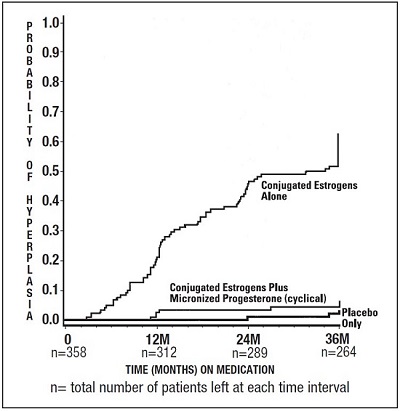
Figure 1. Time to Hyperplasia in Women Receiving up to 36 Months of Treatment
The discontinuation rates due to hyperplasia over the 36 months of treatment are as shown in Table 4. For any degree of hyperplasia, the discontinuation rate for patients who received conjugated estrogens plus PROMETRIUM Capsules was similar to that of the placebo only group, while the discontinuation rate for patients who received conjugated estrogens alone was significantly higher. Women who permanently discontinued treatment due to hyperplasia were similar in demographics to the overall study population.
| Most Advanced Biopsy Result Through 36 Months of Treatment | Treatment Group | |||||
|---|---|---|---|---|---|---|
| Conjugated Estrogens + PROMETRIUM Capsules (cyclical) | Conjugated Estrogens (alone) | Placebo | ||||
| n=120 | n=119 | n=119 | ||||
| Number of patients | % of patients | Number of patients | % of patients | Number of patients | % of patients | |
Adenocarcinoma | 0 | 0 | 0 | 0 | 1 | 1 |
Atypical hyperplasia | 1 | 1 | 10 | 8 | 0 | 0 |
Complex hyperplasia | 0 | 0 | 21 | 18 | 1 | 1 |
Simple hyperplasia | 1 | 1 | 13 | 11 | 0 | 0 |
Effects on secondary amenorrhea
In a single-center, randomized, double-blind clinical study that included premenopausal women with secondary amenorrhea for at least 90 days, administration of 10 days of PROMETRIUM ® (progesterone, USP) Capsules therapy resulted in 80 percent of women experiencing withdrawal bleeding within 7 days of the last dose of PROMETRIUM Capsules, 300 mg per day (n=20), compared to 10 percent of women experiencing withdrawal bleeding in the placebo group (n=21).
In a multicenter, parallel-group, open label, postmarketing dosing study that included premenopausal women with secondary amenorrhea for at least 90 days, administration of 10 days of PROMETRIUM Capsules during two 28-day treatment cycles, 300 mg per day (n=107) or 400 mg per day (n=99), resulted in 73.8 percent and 76.8 percent of women, respectively, experiencing withdrawal bleeding.
The rate of secretory transformation was evaluated in a multicenter, randomized, double-blind clinical study in estrogen-primed postmenopausal women. PROMETRIUM Capsules administered orally for 10 days at 400 mg per day (n=22) induced complete secretory changes in the endometrium in 45 percent of women compared to 0 percent in the placebo group (n=23).
A second multicenter, parallel-group, open label postmarketing dosing study in premenopausal women with secondary amenorrhea for at least 90 days also evaluated the rate of secretory transformation. All subjects received daily oral conjugated estrogens over 3 consecutive 28-day treatment cycles and PROMETRIUM Capsules, 300 mg per day (n=107) or 400 mg per day (n=99) for 10 days of each treatment cycle. The rate of complete secretory transformation was 21.5 percent and 28.3 percent, respectively.
Women's Health Initiative Estrogen Plus Progestin Trial
The WHI estrogen plus progestin trial enrolled predominantly healthy postmenopausal women to assess the risks and benefits of daily oral CE (0.625 mg) in combination with MPA (2.5 mg) compared to placebo in the prevention of certain chronic diseases. The primary endpoint was the incidence of CHD (defined as nonfatal MI, silent MI and CHD death), with invasive breast cancer as the primary adverse outcome. A "global index" included the earliest occurrence of CHD, invasive breast cancer, stroke, PE, endometrial cancer colorectal cancer, hip fracture, or death due to other causes. This trial did not evaluate the effects of CE plus MPA on menopausal symptoms.
The WHI estrogen plus progestin trial was stopped early. After an average follow-up of 5.6 years of treatment, the increased risk of invasive breast cancer crossed the predefined stopping threshold. The global index was supportive of a finding of overall harm. The absolute excess risk of events included in the global index was 19 per 10,000 women-years. Results of the trial, which enrolled 16,608 women (average 63 years of age, range 50 to 79; 83.9%White, 6.8% Black, 5.4% Hispanic, 3.9% Other) are presented in Table 5. These results reflect centrally adjudicated data after an average follow-up of 5.6 years.
| Event | Relative Ratio (95% CI) Nominal confidence intervals unadjusted for multiple looks and multiple comparisons. | Risk Difference (CE/MPA vs placebo/10,000 WYs) |
|---|---|---|
| CHD events Non-fatal MI CHD death | 1.18 (0.95-1.45) 1.24 (0.98-1.56) 1.05 (0.76-1.45) | 6 (41 vs 35) 6 (35 vs 29) 1 (16 vs 15) |
| All Strokes | 1.37 (1.07-1.76) | 9 (33 vs 24) |
| Deep vein thrombosis Not included in Global Index. | 1.87 (1.37-2.54) | 12 (25 vs 14) |
| Pulmonary embolism | 1.98 (1.36-2.87) | 9 (18 vs 9) |
| Invasive breast cancer Includes metastatic and non-metastatic breast cancer with the exception of in situ breast cancer. | 1.24 (1.01-1.53) | 9 (43 vs 35) |
| Colorectal cancer | 0.62 (0.43-0.89) | -6 (10 vs 17) |
| Endometrial cancer | 0.83 (0.49-1.40) | -1 (6 vs 7) |
| Hip fracture | 0.67 (0.47-0.95) | -6 (11 vs 17) |
| Vertebral fractures | 0.68 (0.48-0.96) | -6 (12 vs 17) |
| Total fractures | 0.76 (0.69-0.83) | -51 (161 vs 212) |
| Overall mortality , All deaths, except from breast or colorectal cancer, definite or probable CHD, PE or cerebrovascular disease. | 0.97 (0.81-1.16) | -1 (52 vs 53) |
| Global Index A subset of the events was combined in a "global index" defined as the earliest occurrence of CHD events, invasive breast cancer, stroke, pulmonary embolism, endometrial cancer, colorectal cancer, hip fracture, or death due to other causes. | 1.12 (1.02-1.24) | 20 (189 vs 168) |
Timing of the initiation of estrogen plus progestin therapy relative to the start of menopause may affect the overall risk benefit profile. The results of the WHI estrogen plus progestin trial in women 50 to 59 years of age (N=2,837 for CE/MPA; N=2,683 for placebo) are shown in Table 6.
| Event | Relative Ratio (95% CI) In the WHI studies, hazard ratios were estimated using Cox proportional hazards models comparing treatment to placebo; however, they are described here as relative risks. Nominal confidence intervals unadjusted for multiple looks and multiple comparisons. | Risk Difference (CE/MPA vs placebo/10,000 WYs) |
|---|---|---|
| CHD events Non-fatal MI CHD death | 1.34 (0.82-2.19) 1.32 (0.77-2.25) 0.77 (0.33-1.79) | 5 (23 vs 17) 4 (19 vs 15) -2 (6 vs 8) |
| All Strokes | 1.51 (0.81-2.82) | 5 (15 vs 10) |
| Deep vein thrombosis Not included in “global index.” | 3.01 (1.36-6.66) | 10 (15 vs 5) |
| Pulmonary embolism | 2.05 (0.89-4.71) | 6 (11 vs 5) |
| Invasive breast cancer Includes metastatic and non-metastatic breast cancer with the exception of in situ cancer. | 1.21 (0.81-1.80) | 6 (33 vs 27) |
| Colorectal cancer | 0.79 (0.29-2.18) | -1 (4 vs 5) |
| Endometrial cancer | 1.07 (0.33-3.53) | 0 (4 vs 3) |
| Hip fracture | 0.17 (0.22-1.45) | -3 (1 vs 3) |
| Vertebral fractures | 0.38 (0.15-0.97) | -6 (4 vs 10) |
| Total fractures | 0.82 (0.68-1.00) | -25 (120 vs 145) |
| Overall mortality , All deaths, except from breast or colorectal cancer, definite or probable CHD, PE or cerebrovascular disease. | 0.67 (0.43-1.04) | -10 (21 vs 31) |
| Global Index A subset of the events was combined in a “global index,” defined as the earliest occurrence of CHD events, invasive breast cancer, stroke, PE, colorectal cancer, hip fracture, or death due to other causes. | 1.12 (0.89-1.40) | 12 (103 vs 91) |
Women's Health Initiative Memory Study
The estrogen plus progestin Women's Health Initiative Memory Study (WHIMS), an ancillary study of WHI, enrolled 4,532 predominantly healthy postmenopausal women 65 years of age and older (47 percent were 65 to 69 years of age; 35 percent were 70 to 74 years of age; and 18 percent were 75 years of age and older) to evaluate the effects of daily CE (0.625 mg) plus MPA (2.5 mg) on the incidence of probable dementia (primary outcome) compared to placebo.
After an average follow-up of 4 years, the relative risk of probable dementia for CE plus MPA versus placebo was 2.05 (95 percent CI, 1.21 – 3.48). The absolute risk of probable dementia for CE plus MPA versus placebo was 45 versus 22 per 10,000 women-years. Probable dementia as defined in this study included Alzheimer's disease (AD), vascular dementia (VaD) and mixed type (having features of both AD and VaD). The most common classification of probable dementia in the treatment group and the placebo group was AD. Since the ancillary study was conducted in women 65 to 79 years of age, it is unknown whether these findings apply to younger postmenopausal women. (See PRECAUTIONS, Geriatric Use .)
HOW SUPPLIED
PROMETRIUM ® (progesterone, USP) Capsules 100 mg are round, peach-colored capsules branded with black imprint "SV."
NDC 72989-372-30 (Bottle of 30)
PROMETRIUM ® (progesterone, USP) Capsules 200 mg are oval, pale yellow-colored capsules branded with black imprint "SV2."
NDC 72989-373-30 (Bottle of 30)
Store at 25°C (77°F); excursions permitted to 15° to 30°C (59° to 86°F) [See USP Controlled Room Temperature]. Protect from excessive moisture. Dispense in tight, light-resistant container as defined in USP/NF, accompanied by a Patient Insert. Keep out of reach of children.