Get your patient on Pyridostigmine Bromide - Pyridostigmine Bromide tablet (Pyridostigmine Bromide)
Pyridostigmine Bromide - Pyridostigmine Bromide tablet prescribing information
INDICATIONS AND USAGE
Pyridostigmine bromide tablets are useful in the treatment of myasthenia gravis.
DOSAGE AND ADMINISTRATION
Pyridostigmine bromide tablets, USP are available in following dosage form:
Conventional Tablets
each containing 30 mg pyridostigmine bromide.
Dosage
The size and frequency of the dosage must be adjusted to the needs of the individual patient.
Conventional Tablets
The average dose is ten 60 mg tablets, spaced to provide maximum relief when maximum strength is needed. In severe cases as many as 25 tablets a day may be required, while in mild cases two to twelve tablets a day may suffice.
NOTE: For information on a diagnostic test for myasthenia gravis, and for the evaluation and stabilization of therapy, please see product literature on Tensilon (edrophonium chloride).
CONTRAINDICATIONS
Pyridostigmine bromide tablets are contraindicated in mechanical intestinal or urinary obstruction, and particular caution should be used in its administration to patients with bronchial asthma. Care should be observed in the use of atropine for counteracting side effects, as discussed below.
ADVERSE REACTIONS
The side effects of pyridostigmine bromide tablets are most commonly related to overdosage and generally are of two varieties, muscarinic and nicotinic. Among those in the former group are nausea, vomiting, diarrhea, abdominal cramps, increased peristalsis, increased salivation, increased bronchial secretions, miosis and diaphoresis. Nicotinic side effects are comprised chiefly of muscle cramps, fasciculation and weakness. Muscarinic side effects can usually be counteracted by atropine, but for reasons shown in the preceding section the expedient is not without danger. As with any compound containing the bromide radical, a skin rash may be seen in an occasional patient. Such reactions usually subside promptly upon discontinuance of the medication.
To report SUSPECTED ADVERSE REACTIONS, contact FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DESCRIPTION
Pyridostigmine bromide tablets, USP (pyridostigmine bromide) are an orally active cholinesterase inhibitor. Chemically, pyridostigmine bromide is 3-hydroxy-1-methylpyridinium bromide dimethylcarbamate. Its structural formula is:
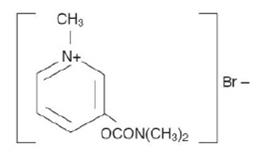
Pyridostigmine bromide tablets,USP are available in the following form:
Tablets containing 60 mg pyridostigmine bromide; each tablet also contains anhydrous lactose, colloidal silicon dioxide and stearic acid.
CLINICAL PHARMACOLOGY
Pyridostigmine bromide tablets inhibits the destruction of acetylcholine by cholinesterase and thereby permits freer transmission of nerve impulses across the neuromuscular junction. Pyridostigmine is an analog of neostigmine (Prostigmin TM ), but differs from it in certain clinically significant respects; for example, pyridostigmine is characterized by a longer duration of action and fewer gastrointestinal side effects.
HOW SUPPLIED
Tablets, are available as white, flat-faced tablets containing 60 mg pyridostigmine bromide, USP in bottles of 30 (NDC 58657-811-30), 90 (NDC 58657-811-90) and 100 (NDC 58657-811-01). Each tablet is white to off-white, round, flat-faced tablet, debossed with "C 60" on one side and a quadrisect scored on the other side.
Store 25°C (77°F); excursions permitted to 15° to 30°C (59° to 86°F) [See USP Controlled Room Temperature]. Preserve in a tight, light-resistant container.
Keep pyridostigmine bromide tablets, USP in a dry place with the silica gel enclosed