Get your patient on Renacidin - Citric Acid, Gluconolactone And Magnesium Carbonate solution (Citric Acid, Gluconolactone And Magnesium Carbonate)
Renacidin - Citric Acid, Gluconolactone And Magnesium Carbonate solution prescribing information
INDICATIONS AND USAGE
Renacidin is indicated for dissolution of bladder calculi of the struvite or apatite variety by local intermittent irrigation through a urethral catheter or cystostomy tube as an alternative or adjunct to surgical procedures.
Renacidin is also indicated for use as an intermittent irrigating solution to prevent encrustations of indwelling urethral catheters and cystostomy tubes.
DOSAGE AND ADMINISTRATION
Renacidin for local irrigation within the lower urinary tract is available in single-use 30 mL containers.
Prepare and Administer the Dose:
Step 1: Inspect Renacidin visually for particulate matter and discoloration prior to administration. If particulate matter or discoloration are observed, do not administer.
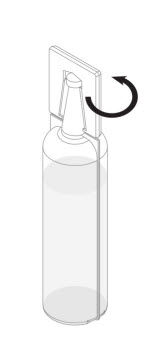
Step 2: Remove the plastic tab connected to the conical tip of the Renacidin container by twisting the plastic tab. See Figure 1.
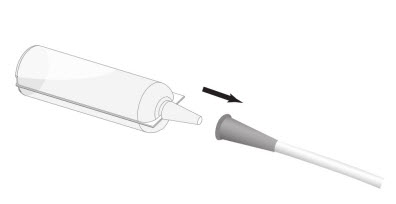
Step 3: Connect the conical tip of the Renacidin container to the end of the urethral catheter or cystostomy tube. See Figure 2.
Step 4: Squeeze the Renacidin container to expel the entire contents into the urethral catheter or cystostomy tube. See Figure 3.

For Dissolution of Bladder Calculi: Instill 30 mL (one container) of Renacidin into the bladder via a urethral catheter or cystostomy tube. Clamp the urethral catheter or cystostomy tube for 30 to 60 minutes. Release the clamp and drain the bladder. Repeat the instillation procedure 4 to 6 times a day. Monitor for dissolution of calculi.
For Prevention of Encrustations in Urethral Catheters and Cystostomy Tubes: Instill 30 mL (one container) of Renacidin into the urethral catheter or cystostomy tube. Clamp the urethral catheter or cystostomy tube for 10 minutes. Remove the clamp and drain the bladder. Repeat the instillation procedure 3 times a day.
CONTRAINDICATIONS
Renacidin is contraindicated in the presence of demonstrable urinary tract extravasation.
ADVERSE REACTIONS
The most common adverse reactions with use of Renacidin for dissolution of bladder calculi or prevention of encrustations of indwelling urethral catheters are “bladder irritability” and chemical cystitis, both reported to occur in approximately 3% of patients. A transient burning sensation in the bladder following Renacidin has been reported to occur in less than 1% of patients receiving Renacidin.
DESCRIPTION
Renacidin ® (Citric Acid, Glucono delta-lactone, and Magnesium Carbonate) is a sterile, non-pyrogenic irrigation solution for use within the lower urinary tract in the dissolution of bladder calculi of the struvite or apatite variety, and prevention of encrustations of urethral catheters and cystostomy tubes.
Each 30 mL of Renacidin contains:
Active ingredients: Citric Acid (anhydrous), USP 1980.6 mg C 6 H 8 O 7 Glucono delta-lactone, USP 59.4 mg C 6 H 10 O 6 Magnesium Carbonate, USP 980.4 mg (MgCO 3 ) 4 · Mg(OH) 2 · 5H 2 O
Citric Acid
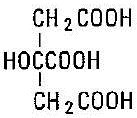
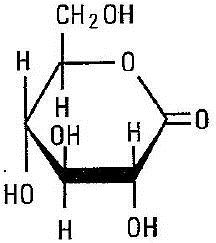
Glucono delta-lactone
Magnesium Carbonate
(MgCO 3 ) 4 · Mg(OH) 2 · 5H 2 O
Inert ingredients: Benzoic Acid, USP 6.9 mg Solution pH: 3.85 (3.5-4.2)
CLINICAL PHARMACOLOGY
Renacidin's action on susceptible apatite calculi results from an exchange of magnesium from the irrigating solution for calcium contained in the stone matrix. The magnesium salts thereby formed are soluble in the gluconocitrate irrigating solution resulting in the dissolution of the calculus. Struvite calculi are composed mainly of magnesium ammonium phosphates which are solubilized by Renacidin due to its acidic pH. Renacidin is not effective for dissolution of calcium oxalate, uric acid or cysteine stones.
HOW SUPPLIED
Renacidin is available as a sterile, non-pyrogenic irrigation solution in 30 mL single-use, low density, polyethylene containers, supplied in boxes of 30 containers each. Exposure of Renacidin to heat or cold should be minimized. Renacidin should be stored at room temperature, 59° to 86°F (15° to 30°C). Avoid excessive heat or cold (keep from freezing). Brief exposure to temperatures of up to 40°C or temperatures down to 5°C does not adversely affect the product.
NDC: 0327-0012-30
PRODUCT CODE: RN030
Revised: December, 2018

Manufactured for GUARDIAN LABORATORIES A DIVISION OF UNITED-GUARDIAN, INC. Hauppauge, N.Y. 11788
