Get your patient on Sucraid - Sacrosidase solution (Sacrosidase)
Sucraid - Sacrosidase solution prescribing information
INDICATIONS AND USAGE
Sucraid ® (sacrosidase) Oral Solution is indicated for the treatment of sucrase deficiency, which is part of congenital sucrase-isomaltase deficiency (CSID), in adult and pediatric patients 5 months of age and older.
DOSAGE AND ADMINISTRATION
Important Administration Information
- Administer Sucraid with each meal or snack.
- Mix Sucraid with cold or room temperature water, milk or infant formula prior to administration. Administration of Sucraid in liquids other than water, milk, or infant formula has not been studied and is not recommended. Do not mix or consume Sucraid with fruit juice.
- Do not warm or heat the water, milk, or infant formula before or after addition of Sucraid.
- Administer half of the dose at the beginning of the meal or snack and the other half of the dose during the meal or snack.
Recommended Dosage The recommended dosage is:
- Patients weighing 15 kg and less: 8,500 International Units (1 mL) administered orally with each meal or snack.
- Patients weighing more than 15 kg: 17,000 International Units (2 mL) administered orally with each meal or snack.
Preparation and Administration Instructions for Patients Weighing 15 kg or Less Multiple-Dose Bottle:
- Using the measuring scoop provided, add 1 scoop of Sucraid (1 mL) to 60 mL of cold or room temperature water, milk, or infant formula.
- Stir to mix well.
- Administer half of the mixed Sucraid solution (30 mL) at the beginning of the meal or snack and the other half of the mixed solution (30 mL) during the meal or snack.
- Do not save any of the mixed Sucraid solution for later use.
- Rinse the measuring scoop with water.
Single-Use Container:
- Empty the entire contents of the single-use container (2 mL) in 120 mL of cold or room temperature water, milk, or infant formula.
- Stir to mix well.
- Divide the mixed Sucraid solution into two separate 60 mL portions. The first portion (60 mL) is for immediate use. • Administer half of the first portion (30 mL) of the mixed Sucraid solution at the beginning of the meal or snack and the other half of the first portion (30 mL) of the mixed Sucraid solution during the meal or snack.
- Store the second portion of the mixed Sucraid solution (60 mL) at 2°C to 8°C (36°F to 46°F) for up to 24 hours for administration with the next meal or snack. • Discard the mixed Sucraid solution if not used within 24 hours.
Preparation and Administration Instructions for Patients Weighing More than 15 kg Multiple-Dose Bottle:
- Using the measuring scoop provided, add 2 scoops of Sucraid (2 mL) to 120 mL of cold or room temperature water, milk, or infant formula.
- Stir to mix well.
- Administer half of the mixed Sucraid solution (60 mL) at the beginning of the meal or snack and the other half of the mixed Sucraid solution (60 mL) during the meal or snack.
- Do not save any of the mixed Sucraid solution for later use.
- Rinse the measuring scoop with water.
Single-Use Container:
- Empty the entire contents of the single-use container (2 mL) in 120 mL of cold or room temperature water, milk, or infant formula.
- Stir to mix well.
- Administer half of the mixed Sucraid solution (60 mL) at the beginning of the meal or snack and the other half of the mixed solution during the meal or snack (60 mL).
- Do not save any of the mixed Sucraid solution for later use.
CONTRAINDICATIONS
Sucraid is contraindicated in patients known to be hypersensitive to yeast, yeast products, glycerin (glycerol), or papain (see WARNINGS).
ADVERSE REACTIONS
The following adverse reactions associated with the use of sacrosidase were identified in clinical studies or postmarketing reports. Because some of these reactions were reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
In clinical studies of up to 54 months duration, a total of 52 patients were treated with Sucraid. The reported adverse reactions (number of patients) were as follows: abdominal pain (4), vomiting (3), nausea (2), diarrhea (2), constipation (2), insomnia (1), headache (1), nervousness (1), and dehydration (1).
Hypersensitivity reactions (wheezing, rash, and pruritis) have been reported (see WARNINGS).
Drug Interactions
Fruit Juice
The acidity in fruit juice may reduce the enzyme activity in Sucraid. Administration of Sucraid with liquids other than water, milk, or infant formula has not been studied and is not recommended (see DOSAGE AND ADMINISTRATION, Administration Instructions).
DESCRIPTION
Sacrosidase is an enzyme with the chemical name of β,D-fructofuranoside fructohydrolase. The enzyme is derived from baker’s yeast ( Saccharomyces cerevisiae ). It has been reported that the primary amino acid structure of this protein consists of 513 amino acids with an apparent molecular weight of 100,000 Da for the glycosylated monomer (range 66,000- 116,000 Da). Reports also suggest that the protein exists in solution as a monomer, dimer, tetramer, and octomer ranging from 100,000 Da to 800,000 Da. It has an isoelectric point (pI) of 4.5.
Sucraid ® (sacrosidase) Oral Solution is an oral enzyme replacement therapy.
Sucraid is a pale yellow to colorless, clear solution with a pleasant, sweet taste. Each milliliter (mL) of Sucraid contains 8,500 International Units (I.U.) of the enzyme sacrosidase, the active ingredient.
Sucraid may contain small amounts of papain (see WARNINGS). Papain is a protein-cleaving enzyme that is introduced in the manufacturing process to digest the cell wall of the yeast and may not be completely removed during subsequent process steps. Sucraid contains sacrosidase in a vehicle comprised of glycerin, water, citric acid, and sodium hydroxide to maintain the pH at 4.0 to 4.7. Glycerol (glycerin) in the amount consumed in the recommended doses of Sucraid has no expected toxicity.
This enzyme preparation is fully soluble with water, milk, and infant formula. DO NOT HEAT SOLUTIONS CONTAINING SUCRAID. Do not put Sucraid in warm or hot liquids (see DOSAGE AND ADMINISTRATION, Administration Instructions).
CLINICAL PHARMACOLOGY
Congenital sucrase-isomaltase deficiency (CSID) is a chronic, autosomal recessive, inherited, phenotypically heterogeneous disease with very variable enzyme activity. CSID is usually characterized by a complete or almost complete lack of endogenous sucrase activity, a very marked reduction in isomaltase activity, and a moderate decrease in maltase activity.
Sucrase is naturally produced in the brush border of the small intestine, primarily the distal duodenum and jejunum. Sucrase hydrolyzes the disaccharide sucrose into its component monosaccharides, glucose and fructose. Isomaltase breaks down disaccharides from starch into simple sugars. Sucraid does not contain isomaltase.
In the absence of endogenous human sucrase, as in CSID, sucrose is not metabolized. Unhydrolyzed sucrose and starch are not absorbed from the intestine and their presence in the intestinal lumen may lead to osmotic retention of water. This may result in loose stools.
Unabsorbed sucrose in the colon is fermented by bacterial flora to produce increased amounts of hydrogen, methane, and water. As a consequence, excessive gas, bloating, abdominal cramps, diarrhea, nausea, and vomiting may occur.
Chronic malabsorption of disaccharides may result in malnutrition. Undiagnosed/untreated CSID patients often fail to thrive and fall behind in their expected growth and development curves. Previously, the treatment of CSID has required the continual use of a strict sucrose-free diet.
CLINICAL STUDIES
A two-phase (dose response preceded by a breath hydrogen phase) double-blind, multi-site, crossover trial was conducted in 28 pediatric patients (approximately 5 months to 12 years of age) with confirmed CSID. During the dose response phase, the patients were challenged with an ordinary sucrose-containing diet while receiving each of four doses of sacrosidase: full strength (9000 I.U./mL) and three dilutions (1:10 [900 I.U./mL], 1:100 [90 I.U./mL], and 1:1000 [9 I.U./mL]) in random order for a period of 10 days. Patients who weighed no more than 15 kg received 1 mL per meal; those weighing more than 15 kg received 2 mL per meal. The dose did not vary with age or sucrose intake. A dose-response relationship was shown between the two higher and the two lower doses. The two higher doses of sacrosidase were associated with significantly fewer total stools and higher proportions of patients having lower total symptom scores, the primary efficacy end-points. In addition, higher doses of sacrosidase were associated with a significantly greater number of hard and formed stools as well as with fewer watery and soft stools, the secondary efficacy end-points.
Analysis of the overall symptomatic response as a function of age indicated that in CSID pediatric patients up to 3 years of age, 86% became asymptomatic. In pediatric patients over 3 years of age, 77% became asymptomatic. Thus, the therapeutic response did not differ significantly according to pediatric age.
A second study of similar design and execution as the first used 4 different dilutions of sacrosidase: 1:100 (90 I.U./mL), 1:1000 (9 I.U./mL), 1:10,000 (0.9 I.U./mL), and 1:100,000 (0.09 I.U./mL). There were inconsistent results with regards to the primary efficacy parameters.
In both trials, however, pediatric patients showed a marked decrease in breath hydrogen output when they received sacrosidase in comparison to placebo.
The effects of Sucraid have not been evaluated in patients with secondary (acquired) sucrase deficiency.
HOW SUPPLIED
118 mL Multiple-Dose Bottle
Sucraid (sacrosidase) Oral Solution is available in 118 mL (4 fluid ounces) multiple-dose translucent plastic bottles, packaged two bottles per carton. Each mL of solution contains 8,500 International Units of sacrosidase. A 1 mL measuring scoop is provided with each bottle. A full measuring scoop is 1 mL.
NDC# 67871-111-04 (2 x 118 mL multiple-dose bottles)
Store under refrigeration at 2°C to 8°C (36°F to 46°F). Discard four weeks after first opening due to the potential for bacterial growth. Protect from heat and light.
2 mL Single-Use Container
Sucraid (sacrosidase) Oral Solution is available in 2 mL, single-use containers that are packaged into a foil pouch. Each 2 mL single-use container contains 17,000 International Units of sacrosidase. Each foil pouch holds a card of 5 containers. Five pouches are then packaged in a box (25 containers). Six boxes are further packaged in a carton (150 containers).
NDC# 67871-111-07 (150 x 2 mL single-use containers)
Store under refrigeration, 2°C to 8°C (36°F to 46°F). Protect from light. Single-use container can be removed from refrigeration and stored at 15°C to 25°C (59°F to 77°F) for up to 3 days (72 hours).
Manufactured by: QOL Medical, LLC Vero Beach, FL 32963 U.S. License No. 2195
www.sucraid.com For questions call 1-866-469-3773
Rev <08/24>
Instructions for Use
Sucraid ® (Su-kreid) (sacrosidase) Oral Solution:
2-mL Single-Use Container
Read this Instructions for Use before you start taking or giving Sucraid to a child, and each time you get a refill. There may be new information. This information does not take the place of talking to your healthcare provider about your or your child’s medical condition or treatment.
Important information you need to know before taking or giving Sucraid:
- The 2-mL single-use container is for children and adults.
- Sucraid is supplied in 2-mL single-use containers in a foil pouch. Each foil pouch holds 5 single-use containers.
Each container is one 2 mL Sucraid dose.
- Your healthcare provider will decide the right dose of Sucraid for you or your child. Do not change the dose of Sucraid without talking to your healthcare provider.
- Sucraid can only be dissolved with cold or room temperature water, milk, or infant formula. Do not put Sucraid in warm or hot liquids. Do not dissolve Sucraid with fruit juice. Do not give or take Sucraid with fruit juice.
- Do not warm or heat the mixed solution before taking or giving Sucraid.
- Sucraid should be taken or given with each meal or snack. Half of the Sucraid dose should be taken at the beginning of each meal or snack. Take or give the remaining Sucraid dose during the meal or snack.
- Do not use the Sucraid single-use container if the seal has been damaged. Contact your pharmacist or healthcare provider if you cannot use the Sucraid single-use container.
Supplies needed to take or give Sucraid:
- 1 Sucraid 2-mL container
- 4 ounces of cold or room temperature water, milk, or infant formula (not included)
- Meal or snack (not included)
- Spoon to mix (not included)
How to take or give Sucraid:
Step 1: Check the expiration date on the Sucraid foil pouch. Do not use Sucraid if it is past the expiration date. Remove 1 Sucraid 2-mL container from a foil pouch.
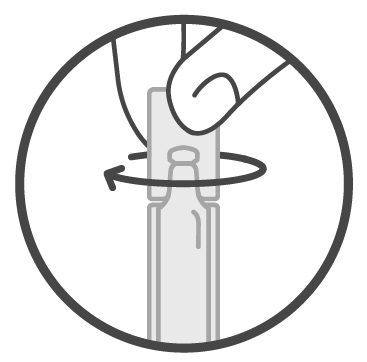
Step 2: Twist the cap to the left to remove it from the container. See Figure 1.
.
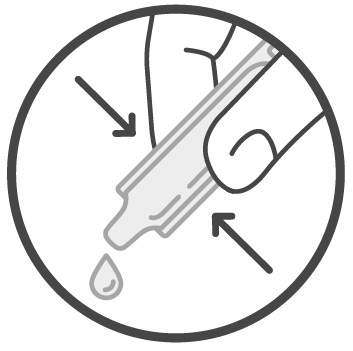
Step 3: Squeeze all the Sucraid solution in the container into 4 ounces of cold or room temperature water, milk, or infant formula. See Figure 2 .
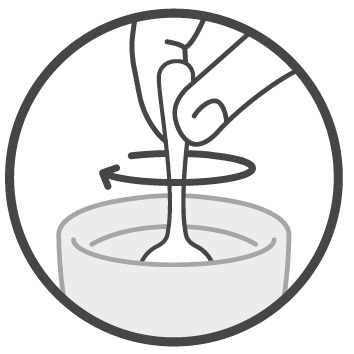
Step 4: Mix your or your child’s prescribed dose in 4 ounces of cold or room temperature water, milk, or infant formula. See Figure 3 .
Step 5: For patients weighing more than 33 pounds (15 kilograms):
- The entire 4 ounces of mixed solution will be taken or given during each meal or snack. Take or give half of the mixed solution (2 ounces) at the beginning of the meal or snack and take or give the other half of the mixed solution (2 ounces) during the meal or snack.
For patients weighing 33 pounds (15 kilograms) or less:
- Divide the 4-ounce mixed solution into two separate 2-ounce portions.
- Take or give half of the first portion (1 ounce) at the beginning of the meal or snack and take or give the other half of the first portion (1 ounce) during the meal or snack.
- Store the second portion (2 ounces) in the refrigerator at 36°F to 46°F (2°C to 8°C) for the next meal or snack. Take or give half of the second portion (1 ounce) at the beginning of the next meal or snack and take or give the other half of the second portion (1 ounce) during the meal or snack.
- Throw away the second portion (2 ounces) if you do not use it within 24 hours.
Throwing away (disposal of) Sucraid:
- Throw away expired or empty Sucraid containers in your household trash.
How should I store Sucraid?
• Store the Sucraid single-use container in the refrigerator between 36°F to 46°F (2°C to 8°C).
• The Sucraid single-use container may be stored between 59°F to 77°F (15°C to 25°C) for up to 3 days.
• Protect Sucraid from heat and light.
Keep Sucraid and all medicines out of the reach of children.
Manufactured by: QOL Medical, LLC Vero Beach, FL 32963 U.S. License No. 2195
For more information, go to www.Sucraid.com or call 1-866-469-3773.
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
Revised: July 2024