Get your patient on Sulfacetamide Sodium And Prednisolone Sodium Phosphate - Sulfacetamide Sodium And Prednisolone Sodium Phosphate solution/ Drops (Sulfacetamide Sodium And Prednisolone Sodium Phosphate)
Sulfacetamide Sodium And Prednisolone Sodium Phosphate - Sulfacetamide Sodium And Prednisolone Sodium Phosphate solution/ Drops prescribing information
INDICATIONS AND USAGE
Sulfacetamide sodium and prednisolone sodium phosphate ophthalmic solution is indicated for corticosteroid-responsive inflammatory ocular conditions for which a corticosteroid is indicated and where superficial bacterial ocular infection or a risk of bacterial ocular infection exists.
Ocular corticosteroids are indicated in inflammatory conditions of the palpebral and bulbar conjunctiva, cornea, and anterior segment of the globe where the inherent risk of corticosteroid use in certain infective conjunctivitides is accepted to obtain diminution in edema and inflammation. They are also indicated in chronic anterior uveitis and corneal injury from chemical, radiation, or thermal burns or penetration of foreign bodies.
The use of a combination drug with an anti-infective component is indicated where the risk of superficial ocular infection is high or where there is an expectation that potentially dangerous numbers of bacteria will be present in the eye.
The particular anti-infective drug in this product is active against the following common bacterial eye pathogens: Escherichia coli, Staphylococcus aureus, Streptococcus pneumoniae, Streptococcus (viridans group), Haemophilus influenzae, Klebsiella species, and Enterobacter species.
This product does not provide adequate coverage against: Neisseria species, Serratia marcescens .
A significant percentage of staphylococcal isolates are completely resistant to sulfa drugs.
DOSAGE AND ADMINISTRATION
Instill two drops of sulfacetamide sodium and prednisolone sodium phosphate ophthalmic solution topically in the eye(s) every four hours. Not more than 20 mL should be prescribed initially. If signs and symptoms fail to improve after two days, patients should be re-evaluated (see PRECAUTIONS ).
Care should be taken not to discontinue therapy prematurely. In chronic conditions, withdrawal of treatment should be carried out by gradually decreasing the frequency of application.
FOR OPHTHALMIC USE ONLY
CONTRAINDICATIONS
Sulfacetamide sodium and prednisolone sodium phosphate ophthalmic solution is contraindicated in most viral diseases of the cornea and conjunctiva including epithelial herpes simplex keratitis (dendritic keratitis), vaccinia, and varicella, and also in mycobacterial infection of the eye and fungal diseases of ocular structures. This product is also contraindicated in individuals with known or suspected hypersensitivity to any of the ingredients of this preparation, to other sulfonamides, or to other corticosteroids. (Hypersensitivity to the antimicrobial components occurs at a higher rate than for other components.)
ADVERSE REACTIONS
Adverse reactions have occurred with corticosteroid/anti-infective combination drugs which can be attributed to the corticosteroid component, the anti-infective component, or the combination. Exact incidence figures are not available since no denominator of treated patients is available.
Reactions occurring most often from the presence of the anti-infective ingredient are allergic sensitizations. Fatalities have occurred, although rarely, due to severe reactions to sulfonamides including Stevens-Johnson syndrome, toxic epidermal necrolysis, fulminant hepatic necrosis, agranulocytosis, aplastic anemia, and other blood dyscrasias (see WARNINGS ) .
Sulfacetamide sodium may cause local irritation.
The reactions due to the corticosteroid component in decreasing order of frequency are: elevation of intraocular pressure (IOP) with possible development of glaucoma, and infrequent optic nerve damage; posterior subcapsular cataract formation; and delayed wound healing.
Although systemic effects are extremely uncommon, there have been rare occurrences of systemic hypercorticoidism after use of topical corticosteroids.
Corticosteroid-containing preparations can also cause acute anterior uveitis or perforation of the globe. Mydriasis, loss of accommodation and ptosis have occasionally been reported following local use of corticosteroids.
Secondary Infection
The development of secondary infection has occurred after use of combinations containing corticosteroids and antimicrobials. Fungal and viral infections of the cornea are particularly prone to develop coincidentally with long-term applications of corticosteroid. The possibility of fungal invasion must be considered in any persistent corneal ulceration where corticosteroid treatment has been used.
Secondary bacterial ocular infection following suppression of host responses also occurs.
To report SUSPECTED ADVERSE REACTIONS, contact Bausch & Lomb Incorporated at 1-800-553-5340 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
Sulfacetamide sodium and prednisolone sodium phosphate ophthalmic solution is incompatible with silver preparations. Local anesthetics related to p-aminobenzoic acid may antagonize the action of the sulfonamides.
DESCRIPTION
Sulfacetamide sodium and prednisolone sodium phosphate ophthalmic solution is a sterile topical ophthalmic solution combining an anti-infective and an adrenocortical steroid.
Each mL contains: Actives: sulfacetamide sodium 100 mg, prednisolone sodium phosphate 2.5 mg (equivalent to prednisolone phosphate 2.3 mg); Inactives: Poloxamer 407, boric acid, edetate disodium dihydrate, purified water. Hydrochloric acid and/or sodium hydroxide may be added to adjust pH (6.5-7.5). Preservative: thimerosal 0.01%.
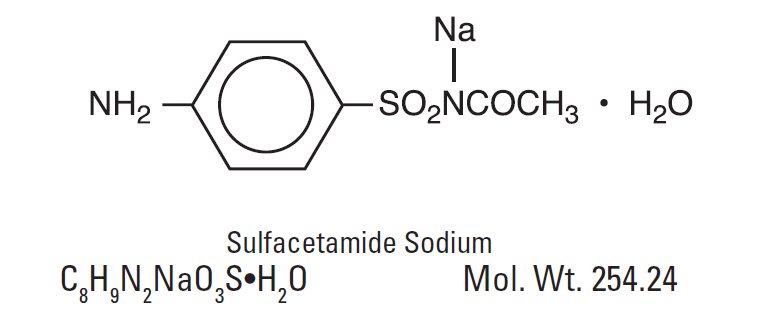
The chemical name for sulfacetamide sodium is N -sulfanilylacetamide monosodium salt monohydrate.
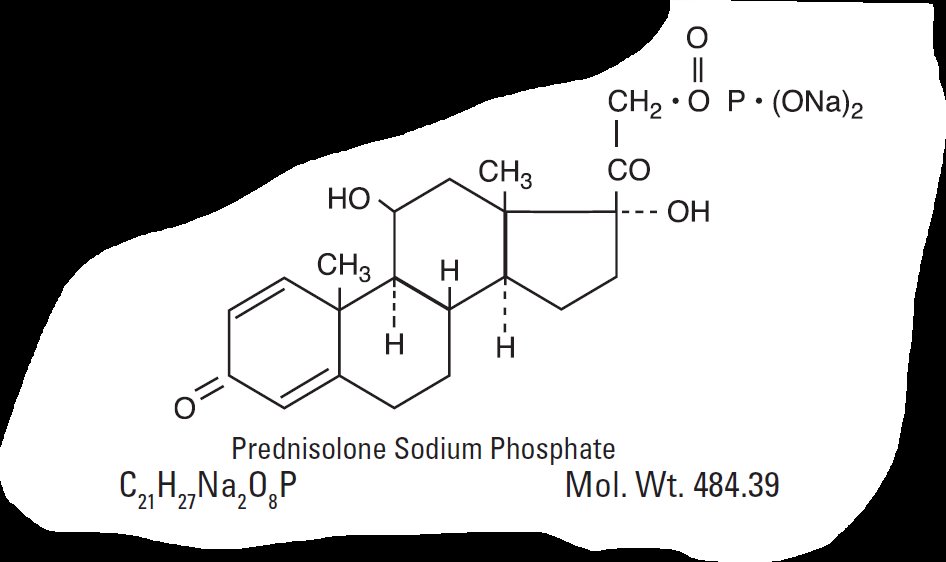
The chemical name for prednisolone sodium phosphate is 11β,17,21-trihydroxypregna-1, 4-diene-3,20-dione, 21-(disodium phosphate).
They have the following structural formulas:
CLINICAL PHARMACOLOGY
Corticosteroids suppress the inflammatory response to a variety of agents and they probably delay or slow healing. Since corticosteroids may inhibit the body’s defense mechanism against infection, a concomitant antimicrobial drug may be used when this inhibition is considered to be clinically significant in a particular case.
When a decision to administer both a corticosteroid and an antimicrobial is made, the administration of such drugs in combination has the advantage of greater patient compliance and convenience, with the added assurance that the appropriate dosage of both drugs is administered, plus assured compatibility of ingredients when both types of drugs are in the same formulation and, particularly, that the correct amount of drug is delivered and retained.
The relative potency of a corticosteroid depends on the molecular structure, concentration, and release from the vehicle.
Microbiology
Sulfacetamide sodium exerts a bacteriostatic effect against susceptible bacteria by restricting the synthesis of folic acid required for growth through competition with p-aminobenzoic acid.
Some strains of bacteria may be resistant to sulfacetamide or resistant strains may emerge in vivo.
The anti-infective component in sulfacetamide sodium and prednisolone sodium phosphate ophthalmic solution is included to provide action against specific organisms susceptible to it. Sulfacetamide sodium is active in vitro against susceptible strains of the following microorganisms: Escherichia coli, Staphylococcus aureus, Streptococcus (viridans group), Haemophilus influenzae, Klebsiella species and Enterobacter species. The product does not provide adequate coverage against: Neisseria species, Pseudomonas species, Serratia marcescens (see INDICATIONS AND USAGE ).
HOW SUPPLIED
Sulfacetamide sodium and prednisolone sodium phosphate ophthalmic solution, 10%/0.23% (prednisolone phosphate) is supplied in a plastic bottle with a white cap and controlled drop tip in the following sizes:
- NDC 24208-317-05 5 mL fill in a 10 mL bottle
- NDC 24208-317-10 10 mL fill in a 10 mL bottle
DO NOT USE IF IMPRINTED NECKBAND IS NOT INTACT. |
Storage:
Store between 15°C to 25°C (59°F to 77°F). KEEP FROM FREEZING. PROTECT FROM LIGHT. KEEP TIGHTLY CLOSED. Sulfonamide solutions darken on prolonged standing and exposure to heat and light. Do not use if solution has darkened. Yellowing does not affect activity.
Keep out of reach of children.
Distributed by:
Bausch & Lomb Americas Inc. Bridgewater, NJ 08807 USA
Manufactured by:
Bausch & Lomb Incorporated
Tampa, FL 33637 USA
© 2023 Bausch & Lomb Incorporated or its affiliates
Revised: August 2023
9118006 (Folded) 9118106 (Flat)