Get your patient on Tacrolimus - Tacrolimus ointment (Tacrolimus)
Tacrolimus - Tacrolimus ointment prescribing information
WARNING
Long-term Safety of Topical Calcineurin Inhibitors Has Not Been Established
Although a causal relationship has not been established, rare cases of malignancy (e.g., skin and lymphoma) have been reported in patients treated with topical calcineurin inhibitors, including tacrolimus ointment.
Therefore:
- Continuous long-term use of topical calcineurin inhibitors, including tacrolimus ointment, in any age group should be avoided, and application limited to areas of involvement with atopic dermatitis.
- Tacrolimus ointment is not indicated for use in children less than 2 years of age. Only 0.03% tacrolimus ointment is indicated for use in children 2-15 years of age.
INDICATIONS AND USAGE
Tacrolimus ointment, both 0.03% and 0.1% for adults, and only 0.03% for children aged 2 to 15 years, is indicated as second-line therapy for the short-term and non-continuous chronic treatment of moderate to severe atopic dermatitis in non-immunocompromised adults and children who have failed to respond adequately to other topical prescription treatments for atopic dermatitis, or when those treatments are not advisable.
Tacrolimus ointment is not indicated for children younger than 2 years of age (see boxed WARNING , WARNINGS and PRECAUTIONS: Pediatric Use ).
DOSAGE AND ADMINISTRATION
ADULT Tacrolimus ointment 0.03% and 0.1%
- Apply a thin layer of tacrolimus ointment to the affected skin twice daily. The minimum amount should be rubbed in gently and completely to control signs and symptoms of atopic dermatitis. Stop using when signs and symptoms of atopic dermatitis resolve.
- If signs and symptoms (e.g. itch, rash, and redness) do not improve within 6 weeks, patients should be re-examined by their healthcare provider to confirm the diagnosis of atopic dermatitis.
- Continuous long-term use of topical calcineurin inhibitors, including tacrolimus ointment should be avoided, and application should be limited to areas of involvement with atopic dermatitis.
The safety of tacrolimus ointment under occlusion, which may promote systemic exposure, has not been evaluated. Tacrolimus ointment should not be used with occlusive dressings.
PEDIATRIC – FOR CHILDREN 2-15 YEARS
Tacrolimus ointment 0.03%
- Apply a thin layer of tacrolimus ointment, 0.03% to the affected skin twice daily. The minimum amount should be rubbed in gently and completely to control signs and symptoms of atopic dermatitis. Stop using when signs and symptoms of atopic dermatitis resolve.
- If signs and symptoms (e.g. itch, rash, and redness) do not improve within 6 weeks, patients should be re-examined by their healthcare provider to confirm the diagnosis of atopic dermatitis.
- Continuous long-term use of topical calcineurin inhibitors, including tacrolimus ointment should be avoided, and application should be limited to areas of involvement with atopic dermatitis.
The safety of tacrolimus ointment under occlusion, which may promote systemic exposure, has not been evaluated. Tacrolimus ointment should not be used with occlusive dressings.
CONTRAINDICATIONS
Tacrolimus ointment is contraindicated in patients with a history of hypersensitivity to tacrolimus or any other component of the ointment.
ADVERSE REACTIONS
No phototoxicity and no photoallergenicity were detected in clinical studies with 12 and 216 normal volunteers, respectively. One out of 198 normal volunteers showed evidence of sensitization in a contact sensitization study. In three 12 week randomized vehicle-controlled studies and four safety studies, 655 and 9,163 patients respectively, were treated with tacrolimus ointment.
The duration of follow-up for adult and pediatric patients in the safety studies is tabulated below.
Time on Study | Adult | Pediatrics | Total |
< 1 year | 4682 | 4481 | 9163 |
≥ 1 year | 1185 | 1349 | 2534 |
≥ 2 years | 200 | 275 | 475 |
≥ 3 years | 118 | 182 | 300 |
The following table depicts the adjusted incidence of adverse events pooled across the 3 identically designed 12-week controlled studies for patients in vehicle, and tacrolimus ointment 0.03%, and tacrolimus ointment 0.1% treatment groups. The table also depicts the unadjusted incidence of adverse events in four safety studies, regardless of relationship to study drug.
12-Week, Randomized, Double-Blind, Phase 3 Studies 12-Week Adjusted Incidence Rate (%) | Open-Label Studies (up to 3 years) 0.1% and 0.03% Tacrolimus Ointment Incidence Rate (%) | |||||||
Adult | Pediatric | Adult | Pediatric | Total | ||||
Vehicle (n=212) % | 0.03% Tacrolimus Ointment (n=210) % | 0.1% Tacrolimus Ointment (n=209) % | Vehicle (n=116) % | 0.03% Tacrolimus Ointment (n=118) % | (n=4682) % | (n=4481) % | (n=9163) % | |
Skin Burning• | 26 | 46 | 58 | 29 | 43 | 28 | 20 | 24 |
Pruritus• | 37 | 46 | 46 | 27 | 41 | 25 | 19 | 22 |
Flu-like symptoms• | 19 | 23 | 31 | 25 | 28 | 22 | 34 | 28 |
Allergic Reaction | 8 | 12 | 6 | 8 | 4 | 9 | 13 | 11 |
Skin Erythema | 20 | 25 | 28 | 13 | 12 | 12 | 7 | 9 |
Headache• | 11 | 20 | 19 | 8 | 5 | 13 | 9 | 11 |
Skin Infection | 11 | 12 | 5 | 14 | 10 | 9 | 16 | 12 |
Fever | 4 | 4 | 1 | 13 | 21 | 2 | 14 | 8 |
Infection | 1 | 1 | 2 | 9 | 7 | 6 | 10 | 8 |
Cough Increased | 2 | 1 | 1 | 14 | 18 | 3 | 10 | 6 |
Asthma | 4 | 6 | 4 | 6 | 6 | 4 | 13 | 8 |
Herpes Simplex | 4 | 4 | 4 | 2 | 0 | 4 | 3 | 3 |
Eczema Herpeticum | 0 | 1 | 1 | 0 | 2 | 0 | 0 | 0 |
Pharyngitis | 3 | 3 | 4 | 11 | 6 | 4 | 12 | 8 |
Accidental Injury | 4 | 3 | 6 | 3 | 6 | 6 | 8 | 7 |
Pustular Rash | 2 | 3 | 4 | 3 | 2 | 2 | 7 | 5 |
Folliculitis• | 1 | 6 | 4 | 0 | 2 | 4 | 2 | 3 |
Rhinitis | 4 | 3 | 2 | 2 | 6 | 2 | 4 | 3 |
Otitis Media | 4 | 0 | 1 | 6 | 12 | 2 | 11 | 6 |
Sinusitis• | 1 | 4 | 2 | 8 | 3 | 6 | 7 | 6 |
Diarrhea | 3 | 3 | 4 | 2 | 5 | 2 | 4 | 3 |
Urticaria | 3 | 3 | 6 | 1 | 1 | 3 | 4 | 4 |
Lack of Drug Effect | 1 | 1 | 0 | 1 | 1 | 6 | 6 | 6 |
Bronchitis | 0 | 2 | 2 | 3 | 3 | 4 | 4 | 4 |
Vomiting | 0 | 1 | 1 | 7 | 6 | 1 | 4 | 3 |
Maculopapular Rash | 2 | 2 | 2 | 3 | 0 | 2 | 1 | 1 |
Rash• | 1 | 5 | 2 | 4 | 2 | 2 | 3 | 3 |
Abdominal Pain | 3 | 1 | 1 | 2 | 3 | 1 | 3 | 2 |
Fungal Dermatitis | 0 | 2 | 1 | 3 | 0 | 2 | 4 | 3 |
Gastroenteritis | 1 | 2 | 2 | 3 | 0 | 2 | 4 | 3 |
Alcohol Intolerance• | 0 | 3 | 7 | 0 | 0 | 4 | 0 | 2 |
Acne• | 2 | 4 | 7 | 1 | 0 | 3 | 2 | 3 |
Sunburn | 1 | 2 | 1 | 0 | 0 | 2 | 1 | 1 |
Skin Disorder | 2 | 2 | 1 | 1 | 4 | 2 | 2 | 2 |
Conjunctivitis | 0 | 2 | 2 | 2 | 1 | 3 | 3 | 3 |
Pain | 1 | 2 | 1 | 0 | 1 | 2 | 1 | 2 |
Vesiculobullous Rash• | 3 | 3 | 2 | 0 | 4 | 2 | 1 | 1 |
Lymphadenopathy | 2 | 2 | 1 | 0 | 3 | 1 | 2 | 1 |
Nausea | 4 | 3 | 2 | 0 | 1 | 2 | 1 | 2 |
Skin Tingling• | 2 | 3 | 8 | 1 | 2 | 2 | 1 | 1 |
Face Edema | 2 | 2 | 1 | 2 | 1 | 1 | 1 | 1 |
Dyspepsia• | 1 | 1 | 4 | 0 | 0 | 2 | 2 | 2 |
Dry Skin | 7 | 3 | 3 | 0 | 1 | 1 | 1 | 1 |
Hyperesthesia• | 1 | 3 | 7 | 0 | 0 | 2 | 0 | 1 |
Skin Neoplasm Benign† | 1 | 1 | 1 | 0 | 0 | 1 | 2 | 2 |
Back Pain• | 0 | 2 | 2 | 1 | 1 | 3 | 0 | 2 |
Peripheral Edema | 2 | 4 | 3 | 0 | 0 | 2 | 0 | 1 |
Varicella Zoster/ Herpes Zoster•‡ | 0 | 1 | 0 | 0 | 5 | 1 | 2 | 2 |
Contact Dermatitis | 1 | 3 | 3 | 3 | 4 | 2 | 2 | 2 |
Asthenia | 1 | 2 | 3 | 0 | 0 | 1 | 0 | 1 |
Pneumonia | 0 | 1 | 1 | 2 | 0 | 1 | 3 | 2 |
Eczema | 2 | 2 | 2 | 0 | 0 | 1 | 0 | 1 |
Insomnia | 3 | 4 | 3 | 1 | 1 | 2 | 0 | 1 |
Exfoliative Dermatitis | 3 | 3 | 1 | 0 | 0 | 0 | 1 | 0 |
Dysmenorrhea | 2 | 4 | 4 | 0 | 0 | 2 | 1 | 1 |
Periodontal Abscess | 1 | 0 | 1 | 0 | 0 | 1 | 1 | 1 |
Myalgia• | 0 | 3 | 2 | 0 | 0 | 2 | 1 | 1 |
Cyst• | 0 | 1 | 3 | 0 | 0 | 1 | 0 | 1 |
Cellulitis | 1 | 1 | 1 | 0 | 0 | 1 | 1 | 1 |
Exacerbation of Untreated Area | 1 | 0 | 1 | 1 | 0 | 1 | 1 | 1 |
Procedural Complication | 1 | 0 | 0 | 1 | 0 | 1 | 1 | 1 |
Hypertension | 0 | 0 | 1 | 0 | 0 | 2 | 0 | 1 |
Tooth Disorder | 0 | 1 | 1 | 1 | 0 | 2 | 1 | 1 |
Arthralgia | 1 | 1 | 3 | 2 | 0 | 2 | 1 | 2 |
Depression | 1 | 2 | 1 | 0 | 0 | 1 | 0 | 1 |
Paresthesia | 1 | 3 | 3 | 0 | 0 | 2 | 1 | 2 |
Alopecia | 0 | 1 | 1 | 0 | 0 | 1 | 1 | 1 |
Urinary Tract Infection | 0 | 0 | 1 | 0 | 0 | 2 | 1 | 2 |
Ear Pain | 1 | 0 | 1 | 0 | 1 | 0 | 1 | 1 |
• May be reasonably associated with the use of this drug product † Generally “warts”. ‡ All the herpes zoster cases in the pediatric 12-week study and the majority of cases in the open-label pediatric studies were reported as chicken pox.
Other adverse events which occurred at an incidence between 0.2% and less than 1% in clinical studies in the above table include: abnormal vision, abscess, anaphylactoid reaction, anemia, anorexia, anxiety, arthritis, arthrosis, bilirubinemia, blepharitis, bone disorder, breast neoplasm benign, bursitis, cataract NOS, chest pain, chills, colitis, conjunctival edema, constipation, cramps, cutaneous moniliasis, cystitis, dehydration, dizziness, dry eyes, dry mouth/nose, dyspnea, ear disorder, ecchymosis, edema, epistaxis, eye pain, furunculosis, gastritis, gastrointestinal disorder, hernia, hypercholesterolemia, hypertonia, hypothyroidism, joint disorder, laryngitis, leukoderma, lung disorder, malaise, migraine, moniliasis, mouth ulceration, nail disorder, neck pain, neoplasm benign, oral moniliasis, otitis externa, photosensitivity reaction, rectal disorder, seborrhea, skin carcinoma, skin discoloration, skin hypertrophy, skin ulcer, stomatitis, tendon disorder, thinking abnormal, tooth caries, sweating, syncope, tachycardia, taste perversion, unintended pregnancy, vaginal moniliasis, vaginitis, valvular heart disease, vasodilatation, and vertigo.
Post-Marketing Events
The following adverse reactions have been identified during postapproval use of tacrolimus ointment. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
CNS
Seizures
Neoplasms
Lymphomas, basal cell carcinoma, squamous cell carcinoma, malignant melanoma
Infections
Bullous impetigo, osteomyelitis, septicemia
Renal
Acute renal failure in patients with or without Netherton's syndrome, renal impairment
Skin
Rosacea, application site edema
Drug Interactions
Formal topical drug interaction studies with tacrolimus ointment have not been conducted. Based on its extent of absorption, interactions of tacrolimus ointment with systemically administered drugs are unlikely to occur but cannot be ruled out (see CLINICAL PHARMACOLOGY ). The concomitant administration of known CYP3A4 inhibitors in patients with widespread and/or erythrodermic disease should be done with caution. Some examples of such drugs are erythromycin, itraconazole, ketoconazole, fluconazole, calcium channel blockers and cimetidine.
DESCRIPTION
Tacrolimus ointment contains tacrolimus, a macrolide immunosuppressant produced by Streptomyces tsukubaensis .
It is for topical dermatologic use only. Chemically, tacrolimus is designated as [3 S- [3 R • [ E (1 S • ,3 S • ,4 S • )],4 S • ,5 R • ,8 S • ,9 E ,12 R • ,14 R • ,15 S • ,16 R • ,18 S • ,19 S • ,26a R • ]]-5,6,8,11,12,13,14,15,16,17,18,19,24,25,26,26a-hexadecahydro-5,19-dihydroxy-3-[2-(4-hydroxy-3-methoxycyclohexyl)-1-methylethenyl]-14,16-dimethoxy-4,10,12,18-tetramethyl-8-(2-propenyl)-15,19-epoxy-3H-pyrido[2,1- c ][1,4]oxaazacyclotricosine-1,7,20,21(4H,23H)-tetrone, monohydrate.
It has the following structural formula:
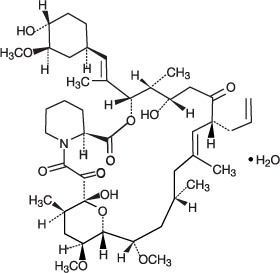
Tacrolimus has a molecular formula of C 44 H 69 NO 12 •H 2 O and a formula weight of 822.03.
Each gram of tacrolimus ointment contains (w/w) either 0.03% or 0.1% of tacrolimus in a base of mineral oil, paraffin, propylene carbonate, white petrolatum and white wax.
CLINICAL PHARMACOLOGY
Mechanism of Action
The mechanism of action of tacrolimus in atopic dermatitis is not known. While the following have been observed, the clinical significance of these observations in atopic dermatitis is not known. It has been demonstrated that tacrolimus inhibits T-lymphocyte activation by first binding to an intracellular protein, FKBP-12.
A complex of tacrolimus-FKBP-12, calcium, calmodulin, and calcineurin is then formed and the phosphatase activity of calcineurin is inhibited. This effect has been shown to prevent the dephosphorylation and translocation of nuclear factor of activated T-cells (NF-AT), a nuclear component thought to initiate gene transcription for the formation of lymphokines (such as interleukin-2, gamma interferon). Tacrolimus also inhibits the transcription for genes which encode IL-3, IL-4, IL-5, GM-CSF, and TNF-α, all of which are involved in the early stages of T-cell activation.
Additionally, tacrolimus has been shown to inhibit the release of pre-formed mediators from skin mast cells and basophils, and to down regulate the expression of FcεRI on Langerhans cells.
CLINICAL STUDIES
Three randomized, double-blind, vehicle-controlled, multi-center, phase 3 studies were conducted to evaluate tacrolimus ointment for the treatment of patients with moderate to severe atopic dermatitis. One (Pediatric) study included 351 patients 2-15 years of age, and the other two (Adult) studies included a total of 632 patients 15-79 years of age. Fifty-five percent (55%) of the patients were women and 27% were black. At baseline, 58% of the patients had severe disease and the mean body surface area (BSA) affected was 46%. Over 80% of patients had atopic dermatitis affecting the face and/or neck region. In these studies, patients applied either tacrolimus ointment 0.03%, tacrolimus ointment 0.1%, or vehicle ointment twice daily to 10% - 100% of their BSA for up to 12 weeks.
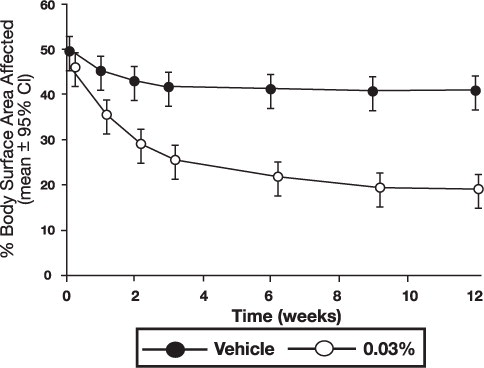
In the pediatric study, a significantly greater (p < 0.001) percentage of patients achieved at least 90% improvement based on the physician's global evaluation of clinical response (the pre-defined primary efficacy endpoint) in the tacrolimus ointment 0.03% treatment group compared to the vehicle treatment group, but there was insufficient evidence that tacrolimus ointment 0.1% provided more efficacy than tacrolimus ointment 0.03%.
In both adult studies, a significantly greater (p < 0.001) percentage of patients achieved at least 90% improvement based on the physician's global evaluation of clinical response in the tacrolimus ointment 0.03% and tacrolimus ointment 0.1% treatment groups compared to the vehicle treatment group. There was evidence that tacrolimus ointment 0.1% may provide more efficacy than tacrolimus ointment 0.03%. The difference in efficacy between tacrolimus ointment 0.1% and 0.03% was particularly evident in adult patients with severe disease at baseline, adults with extensive BSA involvement, and black adults. Response rates for each treatment group are shown below by age groups. Because the two adult studies were identically designed, the results from these studies were pooled in this table.
Physician's Global Evaluation of Clinical Response (% Improvement) | Pediatric Study (2-15 Years of Age) | Adult Studies | |||
Vehicle Ointment N=116 | Tacrolimus Ointment 0.03% N=117 | Vehicle Ointment N=212 | Tacrolimus Ointment 0.03% N=211 | Tacrolimus Ointment 0.1% N=209 | |
100% ≥ 90% ≥ 75% ≥ 50% | 4 (3%) 8 (7%) 18 (16%) 31 (27%) | 14 (12%) 42 (36%) 65 (56%) 85 (73%) | 2 (1%) 14 (7%) 30 (14%) 42 (20%) | 21 (10%) 58 (28%) 97 (46%) 130 (62%) | 20 (10%) 77 (37%) 117 (56%) 152 (73%) |
A statistically significant difference in the percentage of adult patients with ≥ 90% improvement was achieved by week 1 for those treated with tacrolimus ointment 0.1%, and by week 3 for those treated with tacrolimus ointment 0.03%.
A statistically significant difference in the percentage of pediatric patients with ≥ 90% improvement was achieved by week 2 for those treated with tacrolimus ointment 0.03%.
In adult patients who had achieved ≥ 90% improvement at the end of treatment, 35% of those treated with tacrolimus ointment 0.03% and 41% of those treated with tacrolimus ointment 0.1%, regressed from this state of improvement at 2 weeks after end-of-treatment. In pediatric patients who had achieved ≥ 90% improvement, 54% of those treated with tacrolimus ointment 0.03% regressed from this state of improvement at 2 weeks after end-of-treatment. Because patients were not followed for longer than 2 weeks after end-of-treatment, it is not known how many additional patients regressed at periods longer than 2 weeks after cessation of therapy.
In both tacrolimus ointment treatment groups in adults and in the tacrolimus ointment 0.03% treatment group in pediatric patients, a significantly greater improvement compared to vehicle (p < 0.001) was observed in the secondary efficacy endpoints of percent body surface area involved, patient evaluation of pruritus, erythema, edema, excoriation, oozing, scaling, and lichenification.
The following two graphs depict the time course of improvement in the percent body surface area affected in adult and in pediatric patients as a result of treatment.
Figure 1 - Adult Patients Body Surface Area Over Time
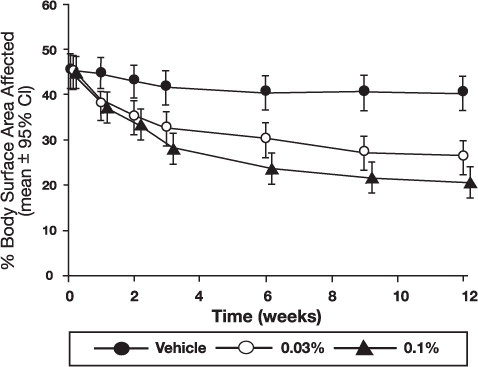
Figure 2 – Pediatric Patients Body Surface Area Over Time
The following two graphs depict the time course of improvement in erythema in adult and in pediatric patients as a result of treatment.
Figure 3 - Adult Patients Mean Erythema Over Time
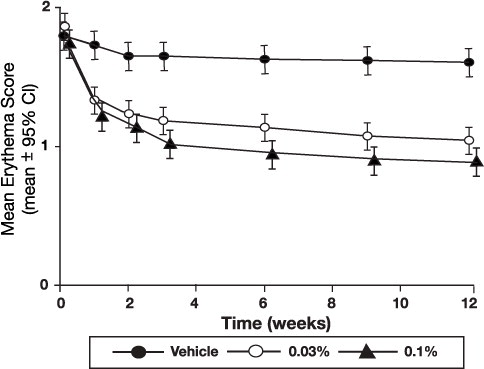
Figure 4 - Pediatric Patients Mean Erythema Over Time
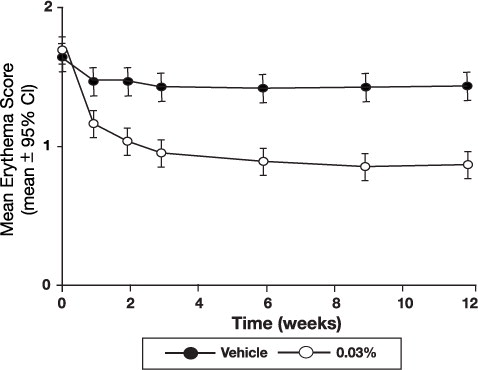
The time course of improvement in the remaining secondary efficacy variables was similar to that of erythema, with improvement in lichenification slightly slower.
HOW SUPPLIED
Tacrolimus ointment 0.03%
NDC 0168-0417-30 | NDC 0168-0417-60 | NDC 0168-0417-99 |
30 gram laminate tube | 60 gram laminate tube | 100 gram laminate tube |
Tacrolimus ointment 0.1%
NDC 0168-0416-30 | NDC 0168-0416-60 | NDC 0168-0416-99 |
30 gram laminate tube | 60 gram laminate tube | 100 gram laminate tube |
Store at room temperature 25°C (77°F); excursions permitted to 15°-30°C (59°-86°F).
Mechanism of Action
The mechanism of action of tacrolimus in atopic dermatitis is not known. While the following have been observed, the clinical significance of these observations in atopic dermatitis is not known. It has been demonstrated that tacrolimus inhibits T-lymphocyte activation by first binding to an intracellular protein, FKBP-12.
A complex of tacrolimus-FKBP-12, calcium, calmodulin, and calcineurin is then formed and the phosphatase activity of calcineurin is inhibited. This effect has been shown to prevent the dephosphorylation and translocation of nuclear factor of activated T-cells (NF-AT), a nuclear component thought to initiate gene transcription for the formation of lymphokines (such as interleukin-2, gamma interferon). Tacrolimus also inhibits the transcription for genes which encode IL-3, IL-4, IL-5, GM-CSF, and TNF-α, all of which are involved in the early stages of T-cell activation.
Additionally, tacrolimus has been shown to inhibit the release of pre-formed mediators from skin mast cells and basophils, and to down regulate the expression of FcεRI on Langerhans cells.