Get your patient on Terconazole - Terconazole cream (Terconazole)
Terconazole - Terconazole cream prescribing information
INDICATIONS AND USAGE
Terconazole vaginal cream is indicated for the local treatment of vulvovaginal candidiasis (moniliasis). As terconazole vaginal cream is effective only for vulvovaginitis caused by the genus Candida , the diagnosis should be confirmed by KOH smears and/or cultures.
DOSAGE AND ADMINISTRATION
One full applicator (5 grams) of terconazole vaginal cream (20 mg terconazole) should be administered intravaginally once daily at bedtime for seven consecutive days.
Before prescribing another course of therapy, the diagnosis should be reconfirmed by smears and/or cultures and other pathogens commonly associated with vulvovaginitis ruled out. The therapeutic effect of terconazole vaginal cream is not affected by menstruation.
CONTRAINDICATIONS
Patients known to be hypersensitive to terconazole or to any of the components of the cream.
ADVERSE REACTIONS
Adverse Reactions from Clinical Trials
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
During controlled clinical studies conducted in the United States, 521 patients with vulvovaginal candidiasis were treated with terconazole 0.4% vaginal cream. Based on comparative analyses with placebo, the adverse experiences considered most likely related to terconazole 0.4% vaginal cream were headache (26% vs. 17% with placebo) and body pain (2.1% vs. 0% with placebo). Fever (1.7% vs. 0.5% with placebo) and chills (0.4% vs, 0% with placebo), vulvovaginal burning, itching and irritation have also been reported. The adverse drug experience on terconazole most frequently causing discontinuation was vulvovaginal itching.
Post-marketing Experience
The following adverse drug reactions have been first identified during post-marketing experience with terconazole:. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
General: Asthenia, Influenza-Like Illness consisting of multiple listed reactions including fever and chills, nausea, vomiting, myalgia, arthralgia, malaise
Immune: Hypersensitivity, Anaphylaxis, Face Edema
Nervous: Dizziness
Respiratory: Bronchospasm
Skin: Rash, Toxic Epidermal Necrolysis, Urticaria
Drug Interactions
The therapeutic effect of terconazole is not affected by oral contraceptive usage.
DESCRIPTION
Terconazole Vaginal Cream 0.4% is a white to off-white, water washable cream for intravaginal administration containing 0.4% of the antifungal agent terconazole, cis -1-[ p -[[2-(2,4-Dichlorophenyl)-2-(1 H -1,2,4-triazol-1-ylmethyl)-1,3-dioxolan-4-yl]methoxy]phenyl]-4-isopropylpiperazine, compounded in a cream base consisting of butylated hydroxyanisole, cetyl alcohol, isopropyl myristate, polysorbate 60, polysorbate 80, propylene glycol, purified water, and stearyl alcohol.
The structural formula of terconazole is as follows:
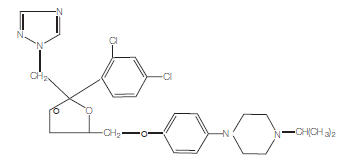 | TERCONAZOLE C 26 H 31 CI 2 N 5 O 3 |
Terconazole, a triazole derivative, is a white to almost white powder with a molecular weight of 532.47. It is insoluble in water; sparingly soluble in ethanol; and soluble in butanol.
CLINICAL PHARMACOLOGY
Absorption
Following a single intravaginal application of a suppository containing 240 mg 14 C-terconazole to healthy women, approximately 70% (range: 64 to 76%) of terconazole remains in the vaginal area during the suppository retention period (16 hours); approximately 10% (range: 5 to 16%) of the administered radioactivity was absorbed systemically over 7 days. Maximum plasma concentrations of terconazole occur 5 to 10 hours after intravaginal application of the cream or suppository. Systemic exposure to terconazole is approximately proportional to the applied dose, whether as the cream or suppository. The rate and extent of absorption of terconazole are similar in patients with vulvovaginal candidiasis (pregnant or non-pregnant) and healthy subjects.
Distribution
Terconazole is highly protein bound (94.9%) in human plasma and the degree of binding is independent of drug concentration over the range of 0.01 to 5 mcg/mL.
Metabolism
Systemically absorbed terconazole is extensively metabolized (>95%).
Elimination
Across various studies in healthy women, after single or multiple intravaginal administration of terconazole as the cream or suppository/ovule, the mean elimination half-life of unchanged terconazole ranged from 6.4 to 8.5 hours. Following a single intravaginal administration of a suppository containing 240 mg 14 C-terconazole to hysterectomized or tubal ligated women, approximately 3 to 10% (mean ± SD: 5.7 ± 3%) of the administered radioactivity was eliminated in the urine and 2 to 6% (mean ± SD: 4.2 ± 1.6%) was eliminated in the feces during the 7-day collection period.
Multiple Dosing
There is no significant increase in maximum plasma concentration or overall exposure (AUC) after multiple daily applications of the cream for 7 days or suppositories for 3 days.
Photosensitivity reactions were observed in some normal volunteers following repeated dermal application of terconazole 2% and 0.8% creams under conditions of filtered artificial ultraviolet light.
Photosensitivity reactions have not been observed in U.S. and foreign clinical trials in patients who were treated with terconazole suppositories or vaginal cream (0.4% and 0.8%).
Microbiology
Mechanism of action
Terconazole, an azole antifungal agent, inhibits fungal cytochrome P-450-mediated 14 alpha-lanosterol demethylase enzyme. This enzyme functions to convert lanosterol to ergosterol. The accumulation of 14 alpha-methyl sterols correlates with the subsequent loss of ergosterol in the fungal cell wall and may be responsible for the antifungal activity of terconazole. Mammalian cell demethylation is less sensitive to terconazole inhibition.
Activity in vitro
Terconazole exhibits antifungal activity in vitro against Candida albicans and other Candida species. The MIC values of terconazole against most Lactobacillus spp. typically found in the human vagina were ≥ 128 mcg/mL; therefore these beneficial bacteria are not affected by drug treatment.
HOW SUPPLIED
Terconazole Vaginal Cream 0.4% is available in 45 gram (NDC 51672-1304-6) tubes with a measured-dose applicator.
Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature].
Mechanism of action
Terconazole, an azole antifungal agent, inhibits fungal cytochrome P-450-mediated 14 alpha-lanosterol demethylase enzyme. This enzyme functions to convert lanosterol to ergosterol. The accumulation of 14 alpha-methyl sterols correlates with the subsequent loss of ergosterol in the fungal cell wall and may be responsible for the antifungal activity of terconazole. Mammalian cell demethylation is less sensitive to terconazole inhibition.