Get your patient on Thiamine Hydrochloride - Thiamine Hydrochloride injection, Solution (Thiamine Hydrochloride)
Thiamine Hydrochloride - Thiamine Hydrochloride injection, Solution prescribing information
INDICATIONS AND USAGE:
Thiamine hydrochloride injection is effective for the treatment of thiamine deficiency or beriberi whether of the dry (major symptoms related to the nervous system) or wet (major symptoms related to the cardiovascular system) variety. Thiamine hydrochloride injection should be used where rapid restoration of thiamine is necessary, as in Wernicke’s encephalopathy, infantile beriberi with acute collapse, cardiovascular disease due to thiamine deficiency, or neuritis of pregnancy if vomiting is severe. It is also indicated when giving IV dextrose to individuals with marginal thiamine status to avoid precipitation of heart failure.
Thiamine hydrochloride injection is also indicated in patients with established thiamine deficiency who cannot take thiamine orally due to coexisting severe anorexia, nausea, vomiting, or malabsorption. Thiamine hydrochloride injection is not usually indicated for conditions of decreased oral intake or decreased gastrointestinal absorption, because multiple vitamins should usually be given.
DOSAGE AND ADMINISTRATION:
“Wet” beriberi with myocardial failure must be treated as an emergency cardiac condition, and thiamine must be administered slowly by the IV route in this situation (see WARNINGS ).
In the treatment of beriberi, 10 mg to 20 mg of thiamine hydrochloride are given IM three times daily for as long as two weeks. (See WARNINGS regarding repeated injections of thiamine.) An oral therapeutic multivitamin preparation containing 5 mg to 10 mg thiamine, administered daily for one month, is recommended to achieve body tissue saturation.
Infantile beriberi that is mild may respond to oral therapy, but if collapse occurs, doses of 25 mg may cautiously be given IV.
Poor dietary habits should be corrected and an abundant and well-balanced dietary intake should be prescribed.
Patients with neuritis of pregnancy in whom vomiting is severe enough to preclude adequate oral therapy should receive 5 mg to 10 mg of thiamine hydrochloride IM daily.
In the treatment of Wernicke-Korsakoff syndrome, thiamine hydrochloride has been administered IV in an initial dose of 100 mg, followed by IM doses of 50 mg to 100 mg daily until the patient is consuming a regular, balanced diet. (See WARNINGS regarding repeated injections of thiamine.)
Patients with marginal thiamine status to whom dextrose is being administered should receive 100 mg thiamine hydrochloride in each of the first few liters of IV fluid to avoid precipitating heart failure.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
CONTRAINDICATIONS:
A history of sensitivity to thiamine or to any of the ingredients in this drug is a contraindication. (See WARNINGS for further information.)
ADVERSE REACTIONS:
To report SUSPECTED ADVERSE REACTIONS, contact Aspiro Pharma Limited at 1- 866-495-1995, or the FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
An occasional individual may develop a hypersensitivity or life-threatening anaphylactic reaction to thiamine, especially after repeated injections. Collapse and death have been reported. A feeling of warmth, pruritus, urticaria, weakness, sweating, nausea, restlessness, tightness of the throat, angioneurotic edema, cyanosis, pulmonary edema, and hemorrhage into the gastrointestinal tract have also been reported. Some tenderness and induration may follow IM use (see WARNINGS ).
DESCRIPTION:
Thiamine Hydrochloride Injection, USP is a sterile, clear colorless solution of thiamine hydrochloride in Water for Injection for intramuscular (IM) or slow intravenous (IV) administration.
Each mL contains: Thiamine hydrochloride, USP 100 mg; chlorobutanol anhydrous (chloral derivative) 0.5%; monothioglycerol 0.5%; water for injection q.s. Sodium hydroxide may have been added for pH adjustment (2.5 to 4.5).
Thiamine hydrochloride, USP or vitamin B 1 , occurs as colorless or white crystalline powder that usually has a slight characteristic odor. Freely soluble in water; soluble in glycerin, slightly soluble in alcohol and insoluble in ether and benzene. Thiamine is rapidly destroyed in neutral or alkaline solutions but is stable in the dry state. It is reasonably stable to heat in acid solution.
The chemical name of thiamine hydrochloride is thiazolium, 3-[(4-amino-2-methyl-5-pyrimidinyl)methyl]-5-(2-hydroxyethyl)-4-methylchloride, monohydrochloride; and it has the following structural formula:
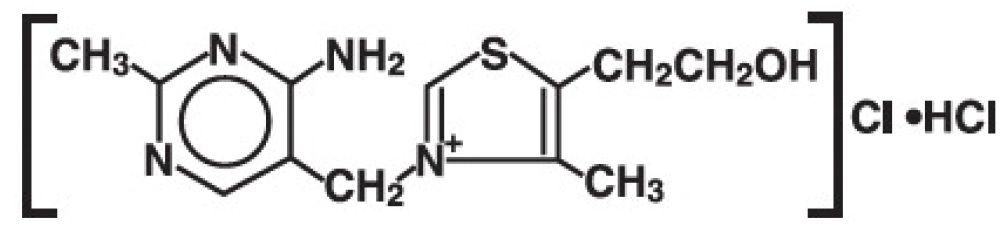
Molecular Formula: C 12 H 17 CIN 4 OS • HCl Molecular Weight: 337.3
CLINICAL PHARMACOLOGY:
The water soluble vitamins are widely distributed in both plants and animals. They are absorbed in man by both diffusion and active transport mechanisms. These vitamins are structurally diverse (derivatives of sugar, pyridine, purines, pyrimidine, organic acid complexes and nucleotide complex) and act as coenzymes, as oxidation-reduction agents, possibly as mitochondrial agents. Metabolism is rapid, and the excess is excreted in the urine.
Thiamine is distributed in all tissues. The highest concentrations occur in liver, brain, kidney and heart. When thiamine intake is greatly in excess of need, tissue stores increase two to three times. If intake is insufficient, tissues become depleted of their vitamin content. Absorption of thiamine following IM administration is rapid and complete.
Thiamine combines with adenosine triphosphate (ATP) to form thiamine pyrophosphate, also known as cocarboxylase, a coenzyme. Its role in carbohydrate metabolism is the decarboxylation of pyruvic acid in the blood and α-ketoacids to acetaldehyde and carbon dioxide. Increased levels of pyruvic acid in the blood indicate vitamin B 1 deficiency.
The requirement for thiamine is greater when the carbohydrate content of the diet is raised. Body depletion of vitamin B 1 can occur after approximately three weeks of total absence of thiamine in the diet.
HOW SUPPLIED:
Thiamine Hydrochloride Injection, USP is a sterile, clear, colorless solution and is supplied as follows:
200 mg per 2 mL (100 mg per mL):
2 mL Multiple-Dose Vials in a Carton of 25 NDC 31722-363-32
Store at 20° to 25°C (68° to 77° F) [see USP Controlled Room Temperature].
PROTECT FROM LIGHT.
Use only if solution is clear and seal intact.
The vial stoppers are not made with natural rubber latex.
Manufactured for: Camber Pharmaceuticals, Inc. Piscataway, NJ 08854
Manufactured by:
Aspiro Pharma Limited
Survey No. 321, Biotech Park, Phase – III,
Karkapatla Village, Markook (Mandal),
Siddipet, Telangana-502281, India.
Issued: 11/2024