Get your patient on Tolmetin Sodium
Tolmetin Sodium prescribing information
Cardiovascular Thrombotic Events
- Nonsteroidal anti-inflammatory drugs (NSAIDs) cause an increased risk of serious cardiovascular thrombotic events, including myocardial infarction and stroke, which can be fatal. This risk may occur early in treatment and may increase with duration of use (see WARNINGS and PRECAUTIONS ).
- TOLECTIN capsules are contraindicated in the setting of coronary artery bypass graft (CABG) surgery (see CONTRAINDICATIONS and WARNINGS ).
Gastrointestinal Risk
- NSAIDs cause an increased risk of serious gastrointestinal adverse events including bleeding, ulceration, and perforation of the stomach or intestines, which can be fatal. These events can occur at any time during use and without warning symptoms. Elderly patients are at greater risk for serious gastrointestinal events (see WARNINGS ).
INDICATIONS AND USAGE: Carefully consider the potential benefits and risks of TOLECTIN capsules, USP and other treatment options before deciding to use TOLECTIN capsules. Use the lowest effective dose for the shortest duration consistent with individual patient treatment goals (see WARNINGS ).
TOLECTIN capsules are indicated for the relief of signs and symptoms of rheumatoid arthritis and osteoarthritis. TOLECTIN capsules are indicated in the treatment of acute flares and the long-term management of the chronic disease.
TOLECTIN capsules are also indicated for treatment of juvenile rheumatoid arthritis. The safety and effectiveness of TOLECTIN capsules have not been established in pediatric patients under 2 years of age (see PRECAUTIONS: Pediatric Use and DOSAGE AND ADMINISTRATION ).
DOSAGE AND ADMINISTRATION: Carefully consider the potential benefits and risks of TOLECTIN capsules and other treatment options before deciding to use TOLECTIN capsules. Use the lowest effective dose for the shortest duration consistent with individual patient treatment goals (see WARNINGS ).
After observing the response to initial therapy with TOLECTIN capsules, the dose and frequency should be adjusted to suit an individual patient’s needs.
For the relief of rheumatoid arthritis or osteoarthritis, the recommended starting dose for adults is 400 mg three times daily (1200 mg daily), preferably including a dose on arising and a dose at bedtime. To achieve optimal therapeutic effect the dose should be adjusted according to the patient’s response after 1 or 2 weeks. Control is usually achieved at doses of 600 mg to 1800 mg daily in divided doses (generally t.i.d.). Doses larger than 1800 mg/day have not been studied and are not recommended.
For the relief of juvenile rheumatoid arthritis, the recommended starting dose for pediatric patients (2 years and older) is 20 mg/kg/day in divided doses (t.i.d. or q.i.d.). When control has been achieved, the usual dose ranges from 15 to 30 mg/kg/day. Doses higher than 30 mg/kg/day have not been studied, and, therefore, are not recommended.
A therapeutic response to TOLECTIN can be expected in a few days to a week. Progressive improvement can be anticipated during succeeding weeks of therapy. If gastrointestinal symptoms occur, TOLECTIN capsules can be administered with antacids other than sodium bicarbonate. TOLECTIN bioavailability and pharmacokinetics are not significantly affected by acute or chronic administration of magnesium and aluminum hydroxides; however, bioavailability is affected by food or milk (see PRECAUTIONS: Drug-Food Interaction ).
CONTRAINDICATIONS: TOLECTIN capsules are contraindicated in patients with known hypersensitivity to tolmetin sodium.
TOLECTIN should not be given to patients who have experienced asthma, urticaria or allergic-type reactions after taking aspirin or other NSAIDs. Severe, rarely fatal, anaphylactic-like reactions to NSAIDs have been reported in such patients (see WARNINGS: Anaphylactoid Reactions and PRECAUTIONS: General: Preexisting Asthma ).
TOLECTIN is contraindicated in the setting of coronary artery bypass graft (CABG) surgery (see WARNINGS ).
ADVERSE REACTIONS: The adverse reactions which have been observed in clinical trials encompass observations in about 4,370 patients treated with TOLECTIN, over 800 of whom have undergone at least one year of therapy. These adverse reactions, reported below by body system, are among those typical of nonsteroidal anti-inflammatory drugs and, as expected, gastrointestinal complaints were most frequent. In clinical trials with TOLECTIN, about 10% of patients dropped out because of adverse reactions, mostly gastrointestinal in nature.
Incidence Greater Than 1%: The following adverse reactions which occurred more frequently than 1 in 100 were reported in controlled clinical trials:
Gastrointestinal: nausea (11%), dyspepsia•, gastrointestinal distress•, abdominal pain•, diarrhea•, flatulence•, vomiting•, constipation, gastritis, and peptic ulcer. Forty percent of the ulcer patients had a prior history of peptic ulcer disease and/or were receiving concomitant anti-inflammatory drugs including corticosteroids, which are known to produce peptic ulceration.
Body as a Whole: headache•, asthenia•, chest pain
Cardiovascular: elevated blood pressure•, edema•
Central Nervous System: dizziness•, drowsiness, depression
Metabolic/Nutritional: weight gain•, weight loss•
Dermatologic: skin irritation
Special Senses: tinnitus, visual disturbance
Hematologic: Small and transient decreases in hemoglobin and hematocrit not associated with gastrointestinal bleeding have occurred. These are similar to changes reported with other nonsteroidal anti-inflammatory drugs.
Urogenital: elevated BUN, urinary tract infection
•Reactions occurring in 3% to 9% of patients treated with TOLECTIN. Reactions occurring in fewer than 3% of the patients are unmarked.
Incidence Less Than 1%: (Causal Relationship Probable) The following adverse reactions were reported less frequently than 1 in 100 in controlled clinical trials or were reported since marketing. The probability exists that there is a causal relationship between TOLECTIN and these adverse reactions.
Gastrointestinal: gastrointestinal bleeding with or without evidence of peptic ulcer, perforation, glossitis, stomatitis, hepatitis, liver function abnormalities
Body as a Whole: anaphylactoid reactions, fever, lymphadenopathy, serum sickness
Hematologic: hemolytic anemia, thrombocytopenia, granulocytopenia, agranulocytosis
Cardiovascular: congestive heart failure in patients with marginal cardiac function
Dermatologic: urticaria, purpura, erythema multiforme, exfoliative dermatitis, Stevens-Johnson Syndrome (SJS), toxic epidermal necrolysis, and fixed drug eruption (FDE)
Urogenital: hematuria, proteinuria, dysuria, renal failure
Incidence Less Than 1%: (Causal Relationship Unknown) Other adverse reactions were reported less frequently than 1 in 100 in controlled clinical trials or were reported since marketing, but a causal relationship between TOLECTIN and the reaction could not be determined. These rarely reported reactions are being listed as alerting information for the physician since the possibility of a causal relationship cannot be excluded.
Body as a Whole: epistaxis
Special Senses: optic neuropathy, retinal and macular changes
To report SUSPECTED ADVERSE REACTIONS, contact Galt Pharmaceuticals, LLC at 1-833-757-0904 or FDA at 1-800-FDA-1088 or WWW.FDA.GOV/MEDWATCH .
DESCRIPTION: Each capsule for oral administration contains 492 mg of tolmetin sodium, USP as the dihydrate in an amount equivalent to 400 mg of tolmetin. Each capsule contains 36 mg (1.568 mEq) of sodium and the following inactive ingredients: colloidal silicon dioxide, croscarmellose sodium, magnesium stearate, microcrystalline cellulose, and sodium lauryl sulfate. The empty gelatin capsule shells contain FD&C Blue No. 1, gelatin, and titanium dioxide. In addition, the imprinting ink contains black iron oxide, D&C Yellow No. 10 Aluminum Lake, FD&C Blue No. 1 Aluminum Lake, FD&C Blue No. 2 Aluminum Lake, FD&C Red No. 40 Aluminum Lake, propylene glycol, and shellac glaze.
The pKa of tolmetin is 3.5 and TOLECTIN is freely soluble in water, soluble in methanol and slightly soluble in alcohol. TOLECTIN is a nonselective nonsteroidal anti-inflammatory agent. The structural formula is:
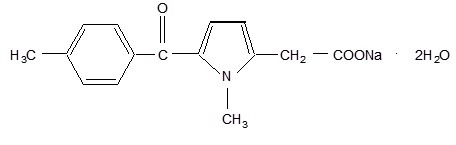
(C 15 H 14 NNaO 3 • 2H 2 O)
Sodium 1-methyl-5 p -toluoylpyrrole-2-acetate dihydrate.
CLINICAL PHARMACOLOGY: Studies in animals have shown TOLECTIN (tolmetin sodium) to possess anti-inflammatory, analgesic, and antipyretic activity. In the rat, TOLECTIN prevents the development of experimentally induced polyarthritis and also decreases established inflammation.
The mode of action of TOLECTIN is not known. However, studies in laboratory animals and man have demonstrated that the anti-inflammatory action of TOLECTIN is not due to pituitary-adrenal stimulation. TOLECTIN inhibits prostaglandin synthetase in vitro and lowers the plasma level of prostaglandin E in man. This reduction in prostaglandin synthesis may be responsible for the anti-inflammatory action. TOLECTIN does not appear to alter the course of the underlying disease in man.
In patients with rheumatoid arthritis and in normal volunteers, TOLECTIN is rapidly and almost completely absorbed with peak plasma levels being reached within 30 to 60 minutes after an oral therapeutic dose. In controlled studies, the time to reach peak tolmetin plasma concentration is approximately 20 minutes longer following administration of a 600 mg tablet, compared to an equivalent dose given as 200 mg tablets. The clinical meaningfulness of this finding, if any, is unknown. Tolmetin displays a biphasic elimination from the plasma consisting of a rapid phase with a half-life of 1 to 2 hours followed by a slower phase with a half-life of about 5 hours. Peak plasma levels of approximately 40 mcg/mL are obtained with a 400 mg oral dose. Essentially all of the administered dose is recovered in the urine in 24 hours either as an inactive oxidative metabolite or as conjugates of tolmetin. An 18-day multiple dose study demonstrated no accumulation of tolmetin when compared with a single dose.
In two fecal blood loss studies of 4 to 6 days duration involving 15 subjects each, TOLECTIN did not induce an increase in blood loss over that observed during a 4-day drug free control period. In the same studies, aspirin produced a greater blood loss than occurred during the drug free control period, and a greater blood loss than occurred during the TOLECTIN treatment period. In one of the two studies, indomethacin produced a greater fecal blood loss than occurred during the drug free control period; in the second study, indomethacin did not induce a significant increase in blood loss.
TOLECTIN is effective in treating both the acute flares and in the long-term management of the symptoms of rheumatoid arthritis, osteoarthritis and juvenile rheumatoid arthritis.
In patients with either rheumatoid arthritis or osteoarthritis, TOLECTIN is as effective as aspirin and indomethacin in controlling disease activity, but the frequency of the milder gastrointestinal adverse effects and tinnitus was less than in aspirin-treated patients, and the incidence of central nervous system adverse effects was less than in indomethacin-treated patients.
In patients with juvenile rheumatoid arthritis, TOLECTIN is as effective as aspirin in controlling disease activity, with a similar incidence of adverse reactions. Mean SGOT values, initially elevated in patients on previous aspirin therapy, remained elevated in the aspirin group and decreased in the TOLECTIN group.
TOLECTIN has produced additional therapeutic benefit when added to a regimen of gold salts and, to a lesser extent, with corticosteroids. TOLECTIN should not be used in conjunction with salicylates since greater benefit from the combination is not likely, but the potential for adverse reactions is increased.
HOW SUPPLIED: TOLECTIN Capsules, USP are available containing 492 mg of tolmetin sodium, USP as the dihydrate in an amount equivalent to 400 mg of tolmetin.
The 400 mg capsules are size “0”, hard-shell gelatin capsule with light blue opaque cap and light blue opaque body imprinted with “R26” on body with black ink containing off-white powder.
They are available as follows:
Bottles of 30 capsules NDC 61825-310-30
Store at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature.]
Protect from light.
Dispense in a tight, light-resistant container as defined in the USP using a child-resistant closure.
Manufactured for: Galt Pharmaceuticals, LLC Atlanta, GA 30339
Revised: 03/2026 GLT.310.99