Get your patient on Triamcinolone Acetonide - Triamcinolone Acetonide cream (Triamcinolone Acetonide)
Triamcinolone Acetonide - Triamcinolone Acetonide cream prescribing information
INDICATIONS & USAGE
Topical corticosteroids are indicated for the relief of the inflammatory and pruritic manifestations of corticosteroid-responsive dermatoses.
DOSAGE & ADMINISTRATION
Topical corticosteroids are generally applied to the affected area as a thin film two to four times daily for the 0.025% strength and two or three times daily for the 0.1% and 0.5% strength depending on the severity of the condition.
Occlusive dressings may be used for the management of psoriasis or recalcitrant conditions.
If an infection develops, the use of occlusive dressings should be discontinued and appropriate antimicrobial therapy instituted.
CONTRAINDICATIONS
Topical corticosteroids are contraindicated in those patients with a history of hypersensitivity to any of the components of the preparation.
ADVERSE REACTIONS
The following local adverse reactions are reported infrequently with topical corticosteroids, but may occur more frequently with the use of occlusive dressings. These reactions are listed in an approximate decreasing order of occurrence: burning, itching, irritation, dryness, folliculitis, hypertrichosis, acneiform eruptions, hypopigmentation, perioral dermatitis, allergic contact dermatitis, maceration of the skin, secondary infection, skin atrophy, striae, and miliaria.
DESCRIPTION
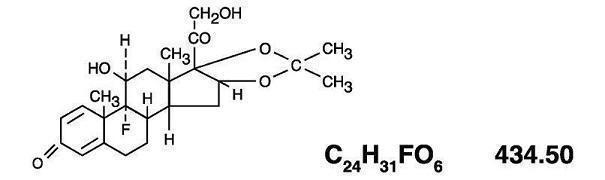
The topical corticosteroids constitute a class of primarily synthetic steroids used as anti-inflammatory and antipruritic agents. Triamcinolone acetonide is a member of this class. Chemically triamcinolone acetonide is pregna-1, 4-diene-3, 20-dione, 9-fluoro-11, 21-dihydroxy-16, 17-[(1-methylethylidene) bis(oxy)]-(11β16α) Its structural formula is:
Each gram of Triamcinolone Acetonide Cream USP, 0.025% contains 0.25 mg triamcinolone acetonide USP in a cream base consisting of purified water, emulsifying wax, mineral oil, propylene glycol, sorbitol solution, cetyl palmitate, sorbic acid, and potassium sorbate.
Each gram of Triamcinolone Acetonide Cream USP, 0.1% contains 1 mg triamcinolone acetonide USP in a cream base consisting of purified water, emulsifying wax, mineral oil, propylene glycol, sorbitol solution, cetyl palmitate, sorbic acid, and potassium sorbate.
Each gram of Triamcinolone Acetonide Cream USP, 0.5% contains 5 mg triamcinolone acetonide USP in a cream base consisting of purified water, emulsifying wax, mineral oil, propylene glycol, sorbitol solution, cetyl palmitate, sorbic acid, and potassium sorbate.
CLINICAL PHARMACOLOGY
Topical corticosteroids share anti-inflammatory, antipruritic and vasoconstrictive actions.
The mechanism of anti-inflammatory activity of the topical corticosteroids is unclear. Various laboratory methods, including vasoconstrictor assays, are used to compare and predict potencies and/or clinical efficacies of the topical corticosteroids. There is some evidence to suggest that a recognizable correlation exists between vasoconstrictor potency and therapeutic efficacy in man.
Pharmacokinetics
The extent of percutaneous absorption of topical corticosteroids is determined by many factors including the vehicle, the integrity of the epidermal barrier, and the use of occlusive dressings.
Topical corticosteroids can be absorbed from normal intact skin. Inflammation and/or other disease processes in the skin increase percutaneous absorption. Occlusive dressings substantially increase the percutaneous absorption of topical corticosteroids. Thus, occlusive dressings may be a valuable therapeutic adjunct for treatment of resistant dermatoses . (See DOSAGE AND ADMINISTRATION).
Once absorbed through the skin, topical corticosteroids are handled through pharmacokinetic pathways similar to systemically administered corticosteroids. Corticosteroids are bound to plasma proteins in varying degrees. Corticosteroids are metabolized primarily in the liver and are then excreted by the kidneys. Some of the topical corticosteroids and their metabolites are also excreted into the bile.
HOW SUPPLIED
Triamcinolone Acetonide Cream USP, 0.025% is supplied in:
15 grams tube NDC 67877-317-15
80 grams tube NDC 67877-317-80
454 grams jar NDC 67877-317-45
Triamcinolone Acetonide Cream USP, 0.1% is supplied in:
15 grams tube NDC 67877-251-15
30 grams tube NDC 67877-251-30
80 grams tube NDC 67877-251-80
454 grams jar NDC 67877-251-45
Triamcinolone Acetonide Cream USP, 0.5% is supplied in:
15 grams tube NDC 67877-318-15
Store at 20°–25°C (68°–77°F) [see USP Controlled Room Temperature].
Avoid excessive heat. Protect from freezing.
PRINTED IN USA
Manufactured for: Ascend Laboratories, LLC, Parsippany, NJ 07054
Manufactured by: Crown Laboratories, Inc., Johnson City, TN 37604
P1810.02
Revised: DEC 2016