Get your patient on Tryptyr - Acoltremon solution (Acoltremon)
Tryptyr - Acoltremon solution prescribing information
INDICATIONS AND USAGE
TRYPTYR is indicated for the treatment of the signs and symptoms of dry eye disease.
DOSAGE AND ADMINISTRATION
Instill one drop in each eye twice daily (approximately 12 hours apart). (2 )
Recommended Dosage
Instill one drop in each eye twice daily (approximately 12 hours apart).
Administration Instructions
Wash hands before use. The single-dose vials are to be used immediately after opening and can be used to dose both eyes. Discard the single-dose vial, including any remaining contents, immediately after use. TRYPTYR can be used concomitantly with other topical ophthalmic eye drops. If more than one topical ophthalmic drug is being used, the drugs should be administered at least five (5) minutes apart . Contact lenses should be removed prior to the administration of TRYPTYR and may be reinserted 15 minutes following administration. If one dose is missed, treatment should continue with the next dose.
DOSAGE FORMS AND STRENGTHS
TRYPTYR is a clear to slightly opalescent, colorless ophthalmic solution containing 0.003% acoltremon in a single-dose vial.
USE IN SPECIFIC POPULATIONS
Pregnancy
Risk Summary There are no adequate and well-controlled studies on TRYPTYR in pregnant women. Systemic exposure to acoltremon from ocular administration is negligible [see Clinical Pharmacology (12.3 )] . Intravenous administration of acoltremon to pregnant rats and rabbits during organogenesis did not produce embryofetal toxicity at 806- and 2151-fold the maximum recommended human ocular dose (MRHOD) of acoltremon on a mg/m 2 basis ( see Data ) .
All pregnancies have a risk of birth defect, loss, or other adverse outcomes. In the US general population, the estimated background risk of major birth defects is 2 to 4%, and of miscarriage is 15 to 20%, of clinically recognized pregnancies.
Data
Animal Data
In embryofetal developmental studies in pregnant rats and rabbits dosed by intravenous injection daily during organogenesis from gestation days 6-17 and gestation days 7-19, respectively, no maternal or fetal toxicity was observed at 806- and 2151-fold the MRHOD of acoltremon on a mg/m 2 basis.
Lactation
Risk Summary There are no data on the presence of acoltremon in human milk, the effects on the breastfed infant, or the effects on milk production. However, systemic exposure to acoltremon following topical ocular administration is low. The lack of clinical data during lactation precludes a clear determination of the risk of TRYPTYR to an infant during lactation; however, the developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for TRYPTYR.
Pediatric Use
The safety and effectiveness of TRYPTYR have not been established in pediatric patients.
Geriatric Use
No clinically relevant differences in safety have been observed between elderly and younger patients.
CONTRAINDICATIONS
None.
WARNINGS AND PRECAUTIONS
To avoid the potential for eye injury and contamination, do not touch the vial tip to the eye or other surfaces. (5.1 )
Potential for Eye Injury and Contamination
To avoid the potential for eye injury and contamination, do not touch the vial tip to the eye or other surfaces.
Use with Contact Lenses
TRYPTYR should not be administered while wearing contact lenses. If contact lenses are worn, they should be removed prior to administration of the solution. Lenses may be reinserted 15 minutes following administration of TRYPTYR.
ADVERSE REACTIONS
The most common adverse reaction was instillation site pain (50%). (6.1 )
To report SUSPECTED ADVERSE REACTIONS, contact Alcon Laboratories, Inc. at 1-800-757-9780, or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
In patients with dry eye disease, 766 patients received at least one dose of TRYPTYR in four randomized controlled clinical trials across 71 sites in the United States. The most common ocular adverse reaction observed in controlled clinical studies with TRYPTYR was instillation site pain (50%). Less than 1% of patients discontinued therapy due to burning or stinging sensation in the eyes.
DESCRIPTION
TRYPTYR (acoltremon ophthalmic solution) 0.003% contains an agonist of TRPM8 ion channels. Acoltremon’s chemical name is (1 R ,2 S ,5 R )-2-isopropyl- N -(4-methoxyphenyl)-5-methylcyclohexane-1-carboxamide. The molecular formula of acoltremon is C 18 H 27 NO 2 and has a molecular weight of 289.42 g/mol.
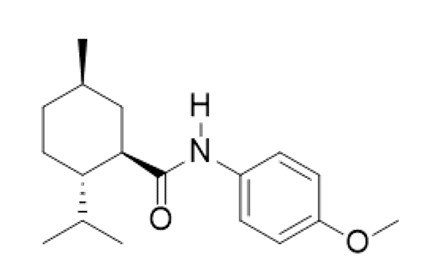
Acoltremon is a white to pale yellow crystalline solid, that is insoluble in water.
TRYPTYR is a sterile, clear to slightly opalescent, colorless, isotonic aqueous solution for topical ophthalmic use, with a pH of approximately 7 and an osmolality of 280 to 330 mOsm/kg. Each mL of TRYPTYR contains active: acoltremon 0.003%; and inactives: polyoxyl 35 castor oil, sodium dihydrogen phosphate dihydrate, sodium chloride, hypromellose and purified water. Additionally, sodium hydroxide is used to adjust pH. TRYPTYR does not contain an anti-microbial preservative.
CLINICAL PHARMACOLOGY
Mechanism of Action
Studies in animals suggest that acoltremon, the active substance in TRYPTYR, is an agonist of transient receptor potential melastatin 8 (TRPM8) thermoreceptors. TRPM8 thermoreceptor stimulation has been shown to activate trigeminal nerve signaling leading to increased basal tear production. The exact mechanism of action for TRYPTYR in dry eye disease is unknown.
Pharmacokinetics
PK was assessed in 25 patients with dry eye disease receiving TRYPTYR administration (1 drop twice daily) on Days 1, 14, and 90. A total of three (3) (12.0%) had plasma concentrations above 20 pg/mL (the lower limit of quantification), with the highest plasma concentration of 213 pg/mL.
NONCLINICAL TOXICOLOGY
Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity Long term studies in animals have not been performed to evaluate the carcinogenic potential of acoltremon. Mutagenicity Acoltremon was not mutagenic or clastogenic in the standard battery of genotoxicity tests including a bacterial reverse mutation assay, an in vitro chromosomal aberration assay in human peripheral lymphocytes and micronucleus assay in rats. Impairment of Fertility Studies to evaluate the potential effects of acoltremon on male or female fertility in animals have not been performed.
CLINICAL STUDIES
The efficacy of TRYPTYR for the treatment of dry eye disease was supported by two randomized, multi-center, double-masked, vehicle-controlled studies (COMET-2 [NCT-05285644] and COMET-3 [NCT-05360966]) enrolling a total of 931 dry eye patients (462 of which received TRYPTYR).
Patients were randomized to TRYPTYR or vehicle (placebo) in a 1:1 ratio and dosed twice a day for 90 days. Use of artificial tears was not allowed during the studies. The mean age was 61 years (range, 30-93 years). The majority of patients were female (74.8%). Enrollment criteria included signs (i.e., corneal fluorescein staining score [2-15] and anesthetized Schirmer tear test [2-9 mm]) and symptoms (i.e., SANDE Score [≥ 50] and Ocular Discomfort Score [≥ 50]) of dry eye disease.
Efficacy Tear film production was measured by unanesthetized Schirmer tear test assessed using a Schirmer strip (0 - 35 mm). The average baseline unanesthetized Schirmer scores for TRYPTYR and Vehicle treated patients was 6.2 mm and 5.9 mm in the COMET-2 study, and 6.8 mm and 6.4 mm in the COMET-3 study, respectively. Of the patients treated at Day 14 (primary endpoint) with TRYPTYR, 42.6% achieved ≥ 10 mm increase in Schirmer score from baseline in the COMET-2 study and 53.2% achieved ≥ 10 mm increase in Schirmer score from baseline at Day 14 in the COMET-3 study, compared to 8.2% and 14.4% of vehicle-treated patients in the COMET-2 study and the COMET-3 study, respectively. A statistically significant improvement in tear production favoring TRYPTYR (p<0.01) was observed in both studies (Table 1 ).
| Tear Production | ||||
| COMET-2 | COMET-3 | |||
| TRYPTYR N = 230 | Vehicle N = 235 | TRYPTYR N = 232 | Vehicle N = 234 | |
| ≥ 10 mm increase in tear production at Day 14 | 42.6% | 8.2% | 53.2% | 14.4% |
| Difference (95% CI) | 34.4% (26.9, 42.0) | 38.8% (30.8, 46.8) | ||
| P-value versus vehicle | < 0.01 | < 0.01 | ||
Consistent results were observed at all timepoints through Day 90.
HOW SUPPLIED/STORAGE AND HANDLING
TRYPTYR (acoltremon ophthalmic solution) 0.003% is supplied as a sterile, clear to slightly opalescent, and colorless solution in a low-density polyethylene (LDPE), single-dose vial with a 0.4 mL fill. One strip of 5 single-dose vials is packaged in a foil pouch with twelve (12) pouches in a carton. NDC 0065-8595-02; Carton of 60 Single-Dose Vials.
Storage Store refrigerated at 2°C to 8°C (36°F to 46°F). After opening the carton, TRYPTYR may be stored refrigerated or at room temperature at 2°C to 25°C (36°F to 77°F). If stored at room temperature, TRYPTYR should be used within 30 days, not to exceed the expiration date printed on the carton and foil pouch.
After opening each foil pouch, the single-dose vials should be used within 7 days, not to exceed the expiration date printed on the vial. Store unopened single-dose vials in the original foil pouch until ready to use.
Mechanism of Action
Studies in animals suggest that acoltremon, the active substance in TRYPTYR, is an agonist of transient receptor potential melastatin 8 (TRPM8) thermoreceptors. TRPM8 thermoreceptor stimulation has been shown to activate trigeminal nerve signaling leading to increased basal tear production. The exact mechanism of action for TRYPTYR in dry eye disease is unknown.