Get your patient on Vancomycin Hydrochloride
Vancomycin Hydrochloride prescribing information
DOSAGE AND ADMINISTRATION
Infusion-related events are related to both the concentration and the rate of administration of vancomycin. Concentrations of no more than 5 mg/mL and rates of no more than 10 mg/min, are recommended in adults (see also age-specific recommendations). In selected patients in need of fluid restriction, a concentration up to 10 mg/mL may be used; use of such higher concentrations may increase the risk of infusion-related events. An infusion rate of 10 mg/min or less is associated with fewer infusion-related events (see ADVERSE REACTIONS ). Infusion-related events may occur, however, at any rate or concentration.
INDICATIONS AND USAGE
Vancomycin Hydrochloride for injection, USP is indicated for the treatment of serious or severe infections caused by susceptible strains of methicillin-resistant (β-lactam-resistant) staphylococci. It is indicated for penicillin-allergic patients, for patients who cannot receive or who have failed to respond to other drugs, including the penicillins or cephalosporins, and for infections caused by vancomycin-susceptible organisms that are resistant to other antimicrobial drugs. Vancomycin Hydrochloride for injection, USP is indicated for initial therapy when methicillin-resistant staphylococci are suspected, but after susceptibility data are available, therapy should be adjusted accordingly.
Vancomycin Hydrochloride for injection, USP is effective in the treatment of staphylococcal endocarditis. Its effectiveness has been documented in other infections due to staphylococci, including septicemia, bone infections, lower respiratory tract infections, skin and skin structure infections. When staphylococcal infections are localized and purulent, antibiotics are used as adjuncts to appropriate surgical measures.
Vancomycin Hydrochloride for injection, USP has been reported to be effective alone or in combination with an aminoglycoside for endocarditis caused by S. viridans or S. bovis. For endocarditis caused by enterococci (e.g., E. faecalis), vancomycin has been reported to be effective only in combination with an aminoglycoside.
Vancomycin Hydrochloride for injection, USP has been reported to be effective for the treatment of diphtheroid endocarditis. Vancomycin Hydrochloride for injection, USP has been used successfully in combination with either rifampin, an aminoglycoside, or both in early-onset prosthetic valve endocarditis caused by S. epidermidis or diphtheroids.
Specimens for bacteriologic cultures should be obtained in order to isolate and identify causative organisms and to determine their susceptibilities to vancomycin.
To reduce the development of drug-resistant bacteria and maintain the effectiveness of Vancomycin Hydrochloride for injection, USP and other antibacterial drugs, Vancomycin Hydrochloride for injection, USP should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
The parenteral form of vancomycin hydrochloride may be administered orally for treatment of antibiotic-associated pseudomembranous colitis produced by C. difficile and for staphylococcal enterocolitis. Parenteral administration of vancomycin hydrochloride alone is of unproven benefit for these indications. Vancomycin is not effective by the oral route for other types of infections.
DOSAGE AND ADMINISTRATION
Infusion-related events are related to both the concentration and the rate of administration of vancomycin. Concentrations of no more than 5 mg/mL and rates of no more than 10 mg/min, are recommended in adults (see also age-specific recommendations). In selected patients in need of fluid restriction, a concentration up to 10 mg/mL may be used; use of such higher concentrations may increase the risk of infusion-related events. An infusion rate of 10 mg/min or less is associated with fewer infusion-related events (see ADVERSE REACTIONS). Infusion-related events may occur, however, at any rate or concentration.
Patients With Normal Renal Function
Adults
The usual daily intravenous dose is 2 g divided either as 500 mg every 6 hours or 1 g every 12 hours. Each dose should be administered at no more than 10 mg/min or over a period of at least 60 minutes, whichever is longer. Other patient factors, such as age or obesity, may call for modification of the usual intravenous daily dose.
Pediatric patients
The usual intravenous dosage of vancomycin is 10 mg/kg per dose given every 6 hours. Each dose should be administered over a period of at least 60 minutes. Close monitoring of serum concentrations of vancomycin may be warranted in these patients.
Neonates
In pediatric patients up to the age of 1 month, the total daily intravenous dosage may be lower. In neonates, an initial dose of 15 mg/kg is suggested, followed by 10 mg/kg every 12 hours for neonates in the 1st week of life and every 8 hours thereafter up to the age of 1 month. Each dose should be administered over 60 minutes. In premature infants, vancomycin clearance decreases as postconceptional age decreases. Therefore, longer dosing intervals may be necessary in premature infants. Close monitoring of serum concentrations of vancomycin is recommended in these patients.
Patients With Impaired Renal Function and Elderly Patients
Dosage adjustment must be made in patients with impaired renal function. In premature infants and the elderly, greater dosage reductions than expected may be necessary because of decreased renal function. Measurement of vancomycin serum concentrations can be helpful in optimizing therapy, especially in seriously ill patients with changing renal function. Vancomycin serum concentrations can be determined by use of microbiologic assay, radioimmunoassay, fluorescence polarization immunoassay, fluorescence immunoassay, or high-pressure liquid chromatography. If creatinine clearance can be measured or estimated accurately, the dosage for most patients with renal impairment can be calculated using the following table. The dosage of vancomycin hydrochloride for injection per day in mg is about 15 times the glomerular filtration rate in mL/min (see following table).
DOSAGE TABLE FOR VANCOMYCIN IN PATIENTS WITH IMPAIRED RENAL FUNCTION (Adapted from Moellering et al. 1 ) | |
Creatinine Clearance mL/min | Vancomycin Dose mg/24 h |
| 100 | 1,545 |
| 90 | 1,390 |
| 80 | 1,235 |
| 70 | 1,080 |
| 60 | 925 |
| 50 | 770 |
| 40 | 620 |
| 30 | 465 |
| 20 | 310 |
| 10 | 155 |
The initial dose should be no less than 15 mg/kg, even in patients with mild to moderate renal insufficiency. The table is not valid for functionally anephric patients. For such patients, an initial dose of 15 mg/kg of body weight should be given to achieve prompt therapeutic serum concentrations. The dose required to maintain stable concentrations is 1.9 mg/kg/24 hr. In patients with marked renal impairment, it may be more convenient to give maintenance doses of 250 to 1,000 mg once every several days rather than administering the drug on a daily basis. In anuria, a dose of 1,000 mg every 7 to 10 days has been recommended. When only serum creatinine is known, the following formula (based on sex, weight and age of the patient) may be used to calculate creatinine clearance. Calculated creatinine clearances (mL/min) are only estimates. The creatinine clearance should be measured promptly.
| Men: | [Weight (kg) x (140 - age in years)] 72 x serum creatinine concentration (mg/dL) |
| Women: | 0.85 x above value |
The serum creatinine must represent a steady state of renal function. Otherwise, the estimated value for creatinine clearance is not valid. Such a calculated clearance is an overestimate of actual clearance in patients with conditions: (1) characterized by decreasing renal function, such as shock, severe heart failure, or oliguria; (2) in which a normal relationship between muscle mass and total body weight is not present, such as in obese patients or those with liver disease, edema, or ascites; and (3) accompanied by debilitation, malnutrition, or inactivity. The safety and efficacy of vancomycin administration by the intrathecal (intralumbar or intraventricular) routes have not been established. Intermittent infusion is the recommended method of administration.
Compatibility with Other Drugs and IV Fluids
The following diluents are physically and chemically compatible (with 4 g/L vancomycin hydrochloride):
5% Dextrose Injection, USP
5% Dextrose Injection and 0.9% Sodium Chloride Injection, USP
Lactated Ringer’s Injection, USP
5% Dextrose and Lactated Ringer’s Injection
Normosol ® -M and 5% Dextrose
0.9% Sodium Chloride Injection, USP
Isolyte ® E
Good professional practice suggests that compounded admixtures should be administered as soon after preparation as is feasible.
Vancomycin solution has a low pH and may cause physical instability of other compounds.
Preparation and Stability
For Administration by Intravenous Drip
Refer to Directions for Proper Use of a Pharmacy Bulk Package Bottle.
DIRECTIONS FOR PROPER USE OF PHARMACY BULK PACKAGE
a.The container closure may be penetrated only one time after reconstitution, utilizing a suitable sterile dispensing set which allows measured distribution of the contents. b.Use of this product is restricted to a suitable work area, such as a laminar flow hood. c.Once this container closure has been punctured, withdrawal of the contents should be completed without delay. If prompt fluid transfer cannot be accomplished, discard the contents no later than 4 HOURS after initial closure puncture. This time limit should begin with the introduction of solvent for diluent into the Pharmacy Bulk Package.
Parenteral drug products should be visually inspected for particulate matter and discoloration prior to administration, whenever solution and container permit.
Do not add supplementary medication to Vancomycin for Injection, USP.
Preparation and Stability
5 g Pharmacy Bulk Package bottle
At the time of use, reconstitute by adding 100 mL of Sterile Water for Injection to the 5 g Pharmacy Bulk Package bottle of dry, sterile vancomycin powder. The resultant solution will contain vancomycin equivalent to 500 mg/10 mL. FURTHER DILUTION IS REQUIRED.
Reconstituted solutions of vancomycin (500 mg/10 mL) must be further diluted in at least 100 mL of a suitable infusion solution. Doses of 1 gram/20 mL must be further diluted in at least 200 mL of a suitable infusion solution. The desired dose diluted in this manner should be administered by intermittent intravenous infusion over a period of at least 60 minutes.
10 g Pharmacy Bulk Package bottle
At the time of use, reconstitute by adding 95 mL of Sterile Water for Injection, USP to the 10 g bottle of dry, sterile vancomycin powder. The resultant solution will contain vancomycin equivalent to 500 mg/5 mL (1 g/10 mL). AFTER RECONSTITUTION, FURTHER DILUTION IS REQUIRED.
Reconstituted solutions of vancomycin (500 mg/5 mL) must be further diluted in at least 100 mL of a suitable infusion solution. For doses of 1 gram (10 mL), at least 200 mL of solution must be used. The desired dose diluted in this manner should be administered by intermittent intravenous infusion over a period of at least 60 minutes.
Parenteral drug products should be visually inspected for particulate matter and discoloration prior to administration, whenever solution and container permit.
For Oral Administration
Oral vancomycin is used in treating antibiotic-associated pseudomembranous colitis caused by C. difficile and for staphylococcal enterocolitis. Vancomycin is not effective by the oral route for other types of infections. The usual adult total daily dosage is 500 mg to 2 g given in 3 or 4 divided doses for 7 to 10 days. The total daily dose in children is 40 mg/kg of body weight in 3 or 4 divided doses for 7 to 10 days. The total daily dosage should not exceed 2 g. The appropriate dose may be diluted in 1 oz of water and given to the patients to drink. Common flavoring syrups may be added to the solution to improve the taste for oral administration. The diluted solution may be administered via a nasogastric tube.
CONTRAINDICATIONS
Vancomycin hydrochloride for injection is contraindicated in patients with known hypersensitivity to this antibiotic.
ADVERSE REACTIONS
To report SUSPECTED ADVERSE REACTIONS, contact Plano Pharmaceuticals Inc. at 1-773-726-1154 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Infusion-Related Events
During or soon after rapid infusion of Sterile Vancomycin Hydrochloride, patients may develop anaphylactoid reactions, including hypotension (see ANIMAL PHARMACOLOGY ), wheezing, dyspnea, urticaria, or pruritus. Rapid infusion may also cause flushing of the upper body ("red neck") or pain and muscle spasm of the chest and back. These reactions usually resolve within 20 minutes but may persist for several hours. Such events are infrequent if Sterile Vancomycin Hydrochloride is given by a slow infusion over 60 minutes. In studies of normal volunteers, infusion-related events did not occur when Sterile Vancomycin Hydrochloride was administered at a rate of 10 mg/min or less.
Nephrotoxicity
Systemic vancomycin exposure may result in acute kidney injury (AKI). The risk of AKI increases as systemic exposure/serum levels increase. Additional risk factors for AKI in patients receiving vancomycin include receipt of concomitant drugs known to be nephrotoxic, in patients with pre-exiting renal impairment, or with co-morbidities that predispose to renal impairment. Interstitial nephritis has also been reported in patients receiving vancomycin.
Gastrointestinal
Onset of pseudomembranous colitis symptoms may occur during or after antibiotic treatment (see WARNINGS ).
Ototoxicity
A few dozen cases of hearing loss associated with Sterile Vancomycin Hydrochloride have been reported. Most of these patients had kidney dysfunction or a preexisting hearing loss or were receiving concomitant treatment with an ototoxic drug. Vertigo, dizziness, and tinnitus have been reported rarely.
Hematopoietic
Reversible neutropenia, usually starting 1 week or more after onset of therapy with vancomycin or after a total dosage of more than 25 g, has been reported for several dozen patients.
Neutropenia appears to be promptly reversible when vancomycin is discontinued. Thrombocytopenia has rarely been reported. Although a causal relationship has not been established, reversible agranulocytosis (granulocytes <500/mm3) has been reported rarely.
Phlebitis
Inflammation at the injection site has been reported.
Miscellaneous
Patients have been reported to have had anaphylaxis, drug fever, nausea, chills, eosinophilia, rashes including exfoliative dermatitis, Stevens-Johnson syndrome (see WARNINGS , Severe Dermatologic Reactions ), and vasculitis in association with the administration of vancomycin.
Chemical peritonitis has been reported following intraperitoneal administration (see PRECAUTIONS )
Post Marketing Reports
The following adverse reactions have been identified during post-approval use of vancomycin. Because these reactions are reported voluntarily from a population of uncertain size, it is not possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Skin and Subcutaneous Tissue Disorders
Severe dermatologic reactions such as toxic epidermal necrolysis (TEN), drug reaction with eosinophilia and systemic symptoms (DRESS), acute generalized exanthematous pustulosis (AGEP), and linear IgA bullous dermatosis (LABD) (see WARNINGS , Severe Dermatologic Reactions ).
Drug Interactions
Concomitant administration of vancomycin and anesthetic agents has been associated with erythema and histamine-like flushing (see Pediatric Use - PRECAUTIONS) and anaphylactoid reactions (see ADVERSE REACTIONS).
Monitor renal function in patients receiving vancomycin and concurrent and/or sequential systemic or topical use of other potentially, neurotoxic and/or nephrotoxic drugs, such as amphotericin B, aminoglycosides, bacitracin, polymixin B, colistin, viomycin, or cisplatin.
DESCRIPTION
Sterile Vancomycin Hydrochloride, USP is a lyophilized powder, for preparing intravenous (IV) infusions, in vials containing the equivalent of 5 g or 10 g vancomycin base. 500 mg of the base are equivalent to 0.34 mmol. When reconstituted with Sterile Water for Injection to a concentration of 50 mg/mL for the 5 g vial and 100 mg/mL for the 10 g vial, the pH of the solution is between 2.5 and 4.5. Sterile Vancomycin Hydrochloride, USP should be administered intravenously in diluted solution (see DOSAGE AND ADMINISTRATION).
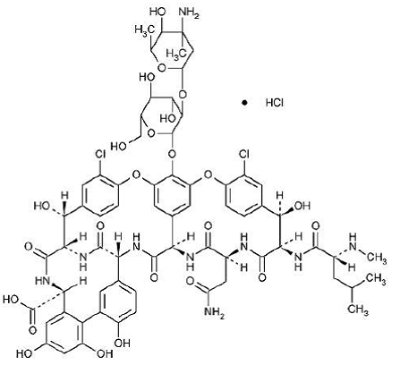
Sterile Vancomycin Hydrochloride is a tricyclic glycopeptide antibiotic derived from Amyco-latopasis orientalis (formerly Nocardia orientals). The chemical name for vancomycin hydrochloride is 3S-[3R•,6S•(S•),7S•,22S•,23R•,26R•,36S•,38aS•]]-3-(2-Amino-2-oxoethyl)-44-[[2-O-(3-amino-2,3,6-trideoxy-3-C-methyl-α-L-lyxo-hexopyranosyl)-ß-D-glucopyranosyl]oxy]-10,19-dichloro-2,3,4,5,6,7,23,24,25,26,36,37,38,38a-tetradecahydro-7,22,28,30,32-pentahydroxy-6-[[4-methyl-2-(methylamino)-1-oxopentyl]amino]-2,5,24,38,39-pentaoxo-22H-8,11:18,21-dietheno-23,36- (iminomethano)-13,16:31,35-dimetheno-1H,16H- [1,6,9]oxadiazacyclohexadecino[4,5-m][10,2,16]-benzoxadiazacyclotetracosine-26-carboxylic acid, monohydrochloride. The molecular formula is C66H75Cl2N9O24• HCl and the molecular weight is 1,485.74.Vancomycin hydrochloride has the following structural formula:
A pharmacy bulk package is a sterile dosage form containing many single doses. The contents are intended for use in a pharmacy admixture program and are restricted to the preparation of admixtures for IV infusion.
FURTHER DILUTION IS REQUIRED BEFORE USE.
CLINICAL PHARMACOLOGY
Vancomycin is poorly absorbed after oral administration.
In subjects with normal kidney function, multiple intravenous dosing of 1 g of vancomycin (15 mg/kg) infused over 60 minutes produces mean plasma concentrations of approximately 63 mcg/mL immediately at the completion of infusion, mean plasma concentrations of approximately 23 mcg/mL 2 hours after infusion, and mean plasma concentrations of approximately 8 mcg/mL 11 hours after the end of the infusion. Multiple dosing of 500 mg infused over 30 minutes produces mean plasma concentrations of about 49 mcg/mL at the completion of infusion, mean plasma concentrations of about 19 mcg/mL 2 hours after infusion, and mean plasma concentrations of about 10 mcg/mL 6 hours after infusion. The plasma concentrations during multiple dosing are similar to those after a single dose.
The mean elimination half-life of vancomycin from plasma is 4 to 6 hours in subjects with normal renal function. In the first 24 hours, about 75% of an administered dose of vancomycin is excreted in urine by glomerular filtration. Mean plasma clearance is about 0.058 L/kg/h, and mean renal clearance is about 0.048 L/kg/hr. Renal dysfunction slows excretion of vancomycin. In anephric patients, the average half-life of elimination is 7.5 days. The distribution coefficient is from 0.3 to 0.43 L/kg. There is no apparent metabolism of the drug. About 60% of an intraperitoneal dose of vancomycin administered during peritoneal dialysis is absorbed systemically in 6 hours. Serum concentrations of about 10 mcg/mL are achieved by intraperitoneal injection of 30 mg/kg of vancomycin.
However, the safety and efficacy of the intraperitoneal use of vancomycin has not been established in adequate and well-controlled trials (see PRECAUTIONS).
Total systemic and renal clearance of vancomycin may be reduced in the elderly. Vancomycin is approximately 55% serum protein bound as measured by ultrafiltration at vancomycin serum concentrations of 10 to 100 mcg/mL. After intravenous administration of vancomycin, inhibitory concentrations are present in pleural, pericardial, ascitic, and synovial fluids; in urine; in peritoneal dialysis fluid; and in atrial appendage tissue. Vancomycin does not readily diffuse across normal meninges into the spinal fluid; but, when the meninges are inflamed, penetration into the spinal fluid occurs.
Microbiology
The bactericidal action of vancomycin results primarily from inhibition of cell-wall biosynthesis. In addition, vancomycin alters bacterial-cell-membrane permeability and RNA synthesis. There is no cross-resistance between vancomycin and other antibiotics. Vancomycin is not active in vitro against gram-negative bacilli, mycobacteria, or fungi.
Synergy
The combination of vancomycin and an aminoglycoside acts synergistically in vitro against many strains of Staphylococcus aureus, Streptococcus bovis, enterococci, and the viridans group streptococci.
Vancomycin has been shown to be active against most strains of the following microorganisms, both in vitro and in clinical infections as described in the INDICATIONS AND USAGE section.
Aerobic gram-positive microorganisms
Diphtheroids
Enterococci (e.g., Enterococcus faecalis)
Staphylococci, including Staphylococcus aureus and Staphylococcus epidermidis (including heterogeneous methicillin-resistant strains)
Streptococcus bovis
Viridans group streptococci
The following in vitro data are available, but their clinical significance is unknown.
Vancomycin exhibits in vitro MIC's of 1 mcg/mL or less against most (≥90%) strains of streptococci listed below and MIC's of 4 mcg/mL or less against most (≥90%) strains of other listed microorganisms; however, the safety and effectiveness of vancomycin in treating clinical infections due to these microorganisms have not been established in adequate and well-controlled clinical trials.
Aerobic gram-positive microorganisms
Listeria monocytogenes
Streptococcus pyogenes
Streptococcus pneumoniae (including penicillin-resistant strains)
Streptococcus agalactiae
Anaerobic gram-positive microorganisms
Actinomyces species
Lactobacillus species
Susceptibility Testing
For specific information regarding susceptibility test interpretive criteria and associated test methods and quality control standards recognized by FDA for this drug, please see: https://www.fda.gov/STIC.
HOW SUPPLIED
Vancomycin Hydrochloride for Injection, USP, supplied as follows:
| Unit of Sale | Strength |
NDC 87188-203-01 Individually Packaged | Vancomycin hydrochloride equivalent to 5 grams vancomycin in a Pharmacy Bulk Package Bottle |
NDC 87188-204-01 Individually Packaged | Vancomycin hydrochloride equivalent to 10 grams vancomycin in a Pharmacy Bulk Package Bottle |
Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature].
The container closure is not made with natural rubber latex.