Get your patient on Vyalev - Foscarbidopa/foslevodopa injection (Foscarbidopa/Foslevodopa)
Vyalev - Foscarbidopa/Foslevodopa injection prescribing information
1 INDICATIONS AND USAGE
VYALEV is indicated for the treatment of motor fluctuations in adults with advanced Parkinson’s disease (PD).
2 DOSAGE AND ADMINISTRATION
- Evaluate vitamin B6 levels prior to starting treatment with carbidopa/levodopa therapies. (2.1 )
- For subcutaneous administration only, preferably in the abdomen, via the VYAFUSER pump. (2.2 , 2.3 , 2.4 )
- See the Full Prescribing Information for calculation of the base continuous dosage, hourly infusion rate, optional loading dose, and extra dose. (2.3 )
- The maximum recommended daily dosage of VYALEV is 3,525 mg of foslevodopa (approximately 2,500 mg levodopa). (2.3 )
2.1 Management of Vitamin B6 Levels
Evaluate vitamin B6 levels prior to initiating carbidopa/levodopa therapies, including VYALEV, periodically during treatment, and as clinically indicated [see Warnings and Precautions 5.7 ] . If vitamin B6 levels are low, supplement to sufficient levels per standard of care. Patients may initiate and continue treatment with VYALEV while supplementing vitamin B6.
2.000000000000000e+00 2 Important Information
For subcutaneous administration only.
Patients selected for treatment with VYALEV should be capable of understanding and using the delivery system [see Healthcare Professional Instructions for Use of VYAFUSER Pump , and Patient Instructions for Use of VYAFUSER Pump ] themselves or with assistance from a caregiver.
Patients should be trained on the proper use of VYALEV and the delivery system prior to initiating.
2.000000000000000e+00 3 Recommended Dosage
VYALEV (foscarbidopa and foslevodopa) is administered as a subcutaneous infusion with the VYAFUSER pump [ see Dosage and Administration (2.4 ), Healthcare Professional Instructions for Use of VYAFUSER Pump , and Patient Instructions for Use of VYAFUSER Pump ] .
VYALEV Base Continuous Dosage and Hourly Infusion Rate
The continuous infusion rate is based on total levodopa dosage (TLD). The hourly base continuous infusion rate (mL/hr) = [(TLD x 1.3) / 240] / [number of hours the patient is typically awake (e.g., 16 hours)], as shown in the steps below.
Step 1: Calculate the TLD for the levodopa-containing medications that VYALEV is replacing. All dosages should be converted to the equivalent dosage of immediate-release levodopa to obtain the TLD. Prescribers should adjust the total dose of levodopa-containing products for COMT inhibitor use. See the Prescribing Information for the respective drugs for conversions or adjustments. Do not include rescue or as needed levodopa or any other anti-Parkinsonian medication or therapy, including medications taken outside of awake time (e.g., night-time dosing) in this calculation.
Step 2: Determine the total daily dosage (mg) of VYALEV foslevodopa component by multiplying the TLD by 1.3. The conversion factor takes into account the molecular weight and bioavailability of foslevodopa compared to levodopa.
Step 3: Determine the total daily volume (mL) of VYALEV by dividing the total daily dosage (mg) of VYALEV by 240. Each 1 mL of VYALEV contains 240 mg of foslevodopa.
Step 4: Determine the hourly base continuous infusion rate of VYALEV by dividing the total daily volume (mL) of VYALEV by the number of hours the patient is typically awake (e.g., 16 hours). VYALEV may be administered over the patient’s waking hours or may be administered for 24 hours. See Dosage and Administration (2.4 ) for adjustment of and alternate infusion rates for lower overnight dosages.
Maximum Dosage
The maximum recommended daily dosage of VYALEV is 3,525 mg of the foslevodopa component (equivalent to approximately 2,500 mg levodopa).
Optional Loading Dose
If VYALEV therapy is being initiated in an “Off” state or the patient has not been receiving their base continuous infusion for more than 3 hours, a loading dose can be administered immediately prior to starting or re-starting the base continuous hourly infusion. Loading doses can be administered either with VYALEV or patients can continue using oral immediate-release carbidopa/levodopa tablets. The loading dose should be calculated from the first morning dose of oral immediate release carbidopa/levodopa the patient took before starting treatment with VYALEV. If VYALEV is used for the loading dose, multiply the first morning dose of oral immediate release levodopa by 1.3 and divide by 240 to determine the loading dose of VYALEV.
The pump is capable of delivering a loading dose ranging from 0.1 mL to a maximum of 3 mL, in increments of 0.1 mL.
Extra Dose
An extra dose volume can be programmed to 1 of 5 options (see Table 1). The “extra dose” feature is limited to no more than 1 extra dose per hour. If 2 or more extra doses are used by the patient during the 24-hour/day treatment period, a revision of the base continuous infusion rate should be considered (refer to the Healthcare Professional Instructions for Use of VYAFUSER Pump details).
| VYALEV extra dose volume | Approximate equivalent levodopa dose • |
| 0.1 mL | 17 mg |
| 0.15 mL | 25.5 mg |
| 0.2 mL | 34 mg |
| 0.25 mL | 42.5 mg |
| 0.3 mL | 51 mg |
• See Step 1
2.000000000000000e+00 4 Preparation and Administration Instructions
Patients should be trained on the proper use of VYALEV and the delivery system prior to initiating treatment with VYALEV and, as necessary, thereafter [see Dosage and Administration (2.3 ) and the Patient Instructions for Use of VYAFUSER Pump ] .
Preparation
- Use aseptic technique.
- Use sterile, single-patient-use infusion components (syringe, infusion set, and vial adapter) qualified for use with the pump to infuse VYALEV.
- Do not dilute.
- Do not mix VYALEV with other products.
- The medication vials are for single dose only. The entire contents of a VYALEV vial should be transferred into a syringe for administration. Do not withdraw a partial portion of the vial contents.
- Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit [see Dosage Forms and Strengths (3 ) ] .
- The infusion set (cannula) can remain in place for up to 3 days when VYALEV is infused continuously.
Administration
Site Selection
- Administer VYALEV subcutaneously, preferably in the abdomen, avoiding the area with a 2-inch radius from the navel.
- Rotate the infusion site and use a new infusion set at least every 3 days.
- Select new infusion sites at least 1 inch from sites used within the previous 12 days.
- Do not infuse VYALEV into areas where the site is tender, bruised, red, or hard to the touch.
Infusion Flow Rates
- Adjustment: Infusion rates may be adjusted in increments of 0.01 mL/hr (which is equivalent to approximately 1.7 mg of levodopa/hour).
- Alternative Flow Rates: The healthcare professional has the option to enable and pre-program the pump with 2 alternative continuous infusion rates (Low/High) that the patient may select to account for changes in functional demand (e.g., lowering the dosage at night or increasing the dose for prolonged intense activity) (see the Healthcare Professional Instructions for Use of VYAFUSER Pump ) .
Discarding VYALEV and Syringe
- Discard the vial after transfer of the product to the syringe. Discard the syringe and any unused VYALEV in the syringe after the product has been in the syringe for 24 hours.
2.000000000000000e+00 5 Interruption of Therapy
Prescribing a backup oral carbidopa and levodopa product is recommended in the event that delivery of VYALEV is interrupted, which may result in underdosing. Sudden discontinuation or rapid dose reduction of VYALEV, without administration of alternative dopaminergic therapy, should be generally avoided [see Warnings and Precautions (5.5 )].
Following interruptions of more than 1 hour, a new infusion set (tubing and cannula) should be used and rotated to a different infusion site. If the infusion has been interrupted for longer than 3 hours, the patient may also self-administer a loading dose, if enabled by their healthcare professional [see Dosage and Administration (2.3 )] .
3 DOSAGE FORMS AND STRENGTHS
Injection: 120 mg foscarbidopa and 2,400 mg foslevodopa per 10 mL (12 mg foscarbidopa and 240 mg foslevodopa per mL). Each single-dose vial contains 10 mL of a colorless to yellow to brown (may have a purple or red tint) and clear to slightly opalescent solution.
8 USE IN SPECIFIC POPULATIONS
Pregnancy: Based on animal data, may cause fetal harm. (8.1 )
8.1 Pregnancy
Risk Summary
There are no data on the developmental risk associated with the use of VYALEV (foscarbidopa and foslevodopa) in pregnant women. Foscarbidopa is a prodrug of carbidopa, and foslevodopa is a prodrug of levodopa. In animal studies, carbidopa-levodopa has been shown to be developmentally toxic (including teratogenic effects) at clinically relevant doses (see Data).
The estimated background risk of major birth defects and miscarriage in the indicated population is unknown. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Data
Animal Data
When administered to pregnant rabbits throughout organogenesis, carbidopa-levodopa caused both visceral and skeletal malformations in fetuses at all doses and ratios of carbidopa-levodopa tested. No teratogenic effects were observed when carbidopa-levodopa was administered to pregnant mice throughout organogenesis. There was a decrease in the number of live pups delivered by rats receiving carbidopa-levodopa during organogenesis.
8.2 Lactation
Risk Summary
Foscarbidopa and Foslevodopa
There are no adequate data on the presence of foscarbidopa or foslevodopa in human milk, the effects of foscarbidopa or foslevodopa on milk production or on the breastfed infant.
Foscarbidopa is a prodrug of carbidopa, and foslevodopa is a prodrug of levodopa.
Carbidopa
There are no adequate data on the presence of carbidopa in human milk, the effects on the breastfed infant, or the effects on milk production. Carbidopa is excreted in rat milk.
Levodopa
Levodopa has been detected in human milk after administration of carbidopa-levodopa. Levodopa decreases secretion of prolactin in humans, which may inhibit lactation. There are no adequate data on the effects of levodopa on the breastfed infant.
The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for VYALEV and any potential adverse effects on the breastfed infant from VYALEV or from the underlying maternal condition.
8.4 Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
8.5 Geriatric Use
Clinical studies of VYALEV did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function and of concomitant disease or other drug therapy.
4 CONTRAINDICATIONS
VYALEV is contraindicated in patients who are currently taking a non-selective monoamine oxidase (MAO) inhibitor or have recently (within 2 weeks) taken a nonselective MAO inhibitor. Hypertension can occur if these drugs are used concurrently [see Drug Interactions (7.1 )] .
5 WARNINGS AND PRECAUTIONS
- May cause falling asleep during activities of daily living. (5.1 )
- Hallucinations/Psychosis: May respond to dose reduction of VYALEV. (5.2 )
- Impulse Control Behaviors: Consider dose reductions or stopping VYALEV. (5.3 )
- Infusion Site Reactions and Infections: Monitor for infusion site infections: Following aseptic techniques while using this medication and frequent rotation of the infusion site is recommended to reduce the risk. (5.4 )
- Avoid sudden discontinuation or rapid dose reduction to reduce the risk of withdrawal-emergent hyperpyrexia and confusion. (5.5 )
- May cause or exacerbate dyskinesia: Consider dose reduction of VYALEV. (5.6 )
5.1 Falling Asleep During Activities of Daily Living and Somnolence
Patients treated with levodopa (the active metabolite of VYALEV) have reported falling asleep while engaged in activities of daily living, including the operation of motor vehicles, which sometimes resulted in accidents. Although many of these patients reported somnolence while on levodopa, some perceived that they had no warning signs, such as excessive drowsiness, and believed that they were alert immediately prior to the event (sleep attack). Some of these events have been reported more than one year after initiation of treatment.
Falling asleep while engaged in activities of daily living usually occurs in patients experiencing preexisting somnolence, although patients may not give such a history. For this reason, prescribers should reassess patients for drowsiness or sleepiness while using VYALEV, especially since some of the events occur well after the start of treatment. Prescribers should be aware that patients may not acknowledge drowsiness or sleepiness until directly questioned about drowsiness or sleepiness during specific activities. Patients who have already experienced somnolence or an episode of sudden sleep onset should not participate in these activities while taking VYALEV.
Before initiating treatment with VYALEV, advise patients about the potential to develop drowsiness and specifically ask about factors that may increase the risk for somnolence with VYALEV such as the use of concomitant sedating medications or the presence of sleep disorders. Consider discontinuing VYALEV in patients who report significant daytime sleepiness or episodes of falling asleep during activities that require active participation (e.g., conversations, eating). If VYALEV is continued, patients should be advised not to drive and to avoid other potentially dangerous activities that might result in harm if the patient becomes somnolent. There is insufficient information to establish that dose reduction will eliminate episodes of falling asleep while engaged in activities of daily living.
5.2 Hallucinations/Psychosis
There is an increased risk for hallucinations and psychosis in patients taking VYALEV. In Study 1 [see Clinical Studies (14 )], hallucinations occurred in 12.2% of patients treated with VYALEV compared to 1.5% of patients treated with oral immediate-release carbidopa-levodopa. Psychosis occurred in 4.1% of patients treated with VYALEV compared to 1.5% of patients treated with oral immediate-release carbidopa-levodopa. Treatment with VYALEV was discontinued in 1 (1.4%) patient because of hallucinations.
Hallucinations associated with levodopa may present shortly after the initiation of therapy and may be responsive to dose reduction of VYALEV or other concomitantly administered medications. Confusion, insomnia, and excessive dreaming may accompany hallucinations. Abnormal thinking and behavior may present with one or more symptoms, including paranoid ideation, delusions, hallucinations, confusion, psychosis, disorientation, aggressive behavior, agitation, and delirium. Review of treatment is recommended if these symptoms develop.
Because of the risk of exacerbating psychosis, patients with a major psychotic disorder should not be treated with VYALEV. In addition, medications that antagonize the effects of dopamine used to treat psychosis may exacerbate the symptoms of PD and may decrease the effectiveness of VYALEV [see Drug Interactions (7.3 ) ] .
5.3 Impulse Control/Compulsive Behaviors
Patients may experience intense urges to gamble, increased sexual urges, intense urges to spend money, binge or compulsive eating, and/or other intense urges, and the inability to control these urges while taking one or more of the medications, including VYALEV, that increase central dopaminergic tone and that are generally used for the treatment of PD. In some cases, although not all, these urges were reported to have stopped when the dose was reduced, or the medication was discontinued.
Because patients may not recognize these behaviors as abnormal, it is important for prescribers to ask patients or their caregivers specifically about the development of new or increased gambling urges, sexual urges, uncontrolled spending, binge or compulsive eating, or other urges while being treated with VYALEV. Consider reducing the dose or discontinuing VYALEV if a patient develops such urges.
5.4 Infusion Site Reactions and Infections
VYALEV can cause infusion site reactions and infections.
In Study 1, one or more infusion site reactions were reported in 62% of patients treated with VYALEV and 8% of patients who received placebo subcutaneous infusion. Various types of reactions at the infusion site have been reported including: erythema, pain, edema, nodule, bruising, hemorrhage, induration, pruritus, extravasation, inflammation, mass, warmth, hematoma, pallor, rash, and swelling. In Study 1, 8% of patients treated with VYALEV and no patient who received placebo withdrew from treatment because of an infusion site reaction.
In Study 1, infusion site infections occurred in 28% of patients treated with VYALEV compared to 3% of patients who received placebo subcutaneous infusion. In Study 1, 5% of patients treated with VYALEV and 2% who received placebo withdrew from treatment because of an infusion site infection. The most frequent infusion site infection reported was cellulitis. If an infection is suspected at the infusion site, the cannula should be removed from the infusion site. If the cannula is removed for an infection, either a new canula should be placed at a new infusion site or, in the event of a prolonged interruption, the patient should be prescribed an oral carbidopa and levodopa product until they are able to resume VYALEV [see Dosage and Administration (2.4 , 2.5 )] .
5.5 Withdrawal-Emergent Hyperpyrexia and Confusion
A symptom complex that resembles neuroleptic malignant syndrome (characterized by elevated temperature, muscular rigidity, altered consciousness, and autonomic instability), with no other obvious etiology, has been reported in association with rapid dose reduction, withdrawal of, or changes in dopaminergic therapy. Avoid sudden discontinuation or rapid dose reduction in patients taking VYALEV. If VYALEV is discontinued, the dose should be tapered to reduce the risk of hyperpyrexia and confusion [see Dosage and Administration (2.5 ) ] .
5.6 Dyskinesia
VYALEV may cause or exacerbate dyskinesias. In Study 1, dyskinesia occurred in 11% of patients treated with VYALEV compared to 6% of patients treated with oral immediate-release carbidopa-levodopa. The occurrence of dyskinesias may require a dosage reduction of VYALEV or other medications used to treat PD.
5.7 Vitamin B6 Deficiency and Seizures
Treatment with carbidopa-levodopa (the active metabolites of VYALEV) may contribute to reduced vitamin B6 levels. Higher doses of carbidopa-levodopa may increase the risk of vitamin B6 deficiency. Seizures associated with vitamin B6 deficiency have been reported in the postmarketing setting in patients taking carbidopa/levodopa. In these reported cases, seizures were refractory to traditional antiseizure medications and only resolved after vitamin B6 administration. Other symptoms of vitamin B6 deficiency may occur, including depression, confusion, cheilosis, glossitis, dermatitis, anemia, and/or neuropathy.
Evaluate vitamin B6 levels prior to initiation of VYALEV and periodically while on treatment or if symptoms associated with vitamin B6 deficiency are identified. Supplement with vitamin B6 as necessary.
5.000000000000000e+00 8 Cardiovascular Ischemic Events
In clinical studies, myocardial infarction and arrhythmia were reported in patients taking carbidopa-levodopa (the active metabolites of VYALEV). Ask patients about symptoms of ischemic heart disease and arrhythmia, especially those with a history of myocardial infarction or cardiac arrhythmias.
5.000000000000000e+00 9 Glaucoma
Carbidopa-levodopa (the active metabolites of VYALEV) may cause increased intraocular pressure in patients with glaucoma. Monitor intraocular pressure in patients with glaucoma after starting VYALEV.
6 ADVERSE REACTIONS
The following serious adverse reactions are discussed below and elsewhere in labeling:
- Falling Asleep During Activities of Daily Living and Somnolence [see Warnings and Precautions (5.1 ) ]
- Hallucinations/Psychosis [see Warnings and Precautions (5.2 ) ]
- Impulse Control/Compulsive Behaviors [see Warnings and Precautions (5.3 ) ]
- Infusion Site Reactions and Infections [see Warnings and Precautions (5.4 )]
- Withdrawal-Emergent Hyperpyrexia and Confusion [see Warnings and Precautions (5.5 ) ]
- Dyskinesia [see Warnings and Precautions (5.6 ) ]
- Vitamin B6 Deficiency and Seizures [see Warnings and Precautions (5.7 ) ]
- Cardiovascular Ischemic Events [see Warnings and Precautions (5.8 ) ]
- Glaucoma [see Warnings and Precautions (5.9 )]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
In Study 1, a 12-week, active-controlled clinical trial, a total of 141 patients with advanced PD were enrolled [see Clinical Studies (14 )]. Of these, 74 patients received VYALEV and 67 received oral immediate-release carbidopa-levodopa with placebo subcutaneous infusion.
Adverse reactions led to discontinuation of VYALEV in 22% of patients, which included hallucinations, infusion site reactions, and infusion site infections [see Warnings and Precautions (5.2 , 5.5 )]. Table 2 presents the adverse reactions that occurred in ≥3% of patients who received VYALEV and with a difference of >2% between the VYALEV and the oral immediate release carbidopa-levodopa groups in Study 1.
| Adverse Reaction | VYALEV (n = 74 ) % | Oral immediate-release carbidopa-levodopa (n = 67 ) % |
| Infusion/catheter site reaction a | 62 | 8 |
| Infusion/catheter site infection b | 28 | 3 |
| Hallucination | 12 | 2 |
| Dyskinesia | 11 | 6 |
| On and off phenomenon | 8 | 0 |
| Balance disorder | 5 | 0 |
| Constipation | 5 | 0 |
| Peripheral swelling | 5 | 0 |
| Agitation | 4 | 2 |
| Insomnia | 4 | 2 |
| Psychotic disorder c | 4 | 2 |
| Dyspnea | 4 | 0 |
| ||
7 DRUG INTERACTIONS
- Selective MAO-B inhibitors: May cause orthostatic hypotension. (7.1 )
- Antihypertensive drugs: May cause symptomatic postural hypotension. Dosage adjustment of the antihypertensive drug may be needed. (7.2 )
- Dopamine D2 receptor antagonists and isoniazid may reduce the effectiveness of VYALEV. (7.3 )
7.1 Monoamine Oxidase (MAO) Inhibitors
Nonselective MAO Inhibitors
The use of nonselective MAO inhibitors (e.g., phenelzine and tranylcypromine) with VYALEV is contraindicated [see Contraindications (4 ) ] . Discontinue use of any nonselective MAO inhibitors at least two weeks prior to initiating VYALEV.
Selective MAO Inhibitors
The use of selective MAO-B inhibitors (e.g., rasagiline and selegiline) with VYALEV may be associated with orthostatic hypotension. Monitor patients who are taking these drugs.
7.2 Antihypertensive Drugs
The concurrent use of VYALEV with antihypertensive medications can cause symptomatic postural hypotension. A dose reduction of the antihypertensive medication may be needed after starting or increasing the dosage of VYALEV.
7.3 Dopamine D2 Receptor Antagonists and Isoniazid
Dopamine D2 receptor antagonists (e.g., phenothiazines, butyrophenones, risperidone, metoclopramide, papaverine) and isoniazid may reduce the effectiveness of foslevodopa. Monitor patients for worsening Parkinson’s symptoms when patients are taking these medications with VYALEV.
11 DESCRIPTION
VYALEV injection is a solution that is a combination of foscarbidopa (carbidopa-4´-monophosphate) and foslevodopa (levodopa-4´-monophosphate).
Foscarbidopa and foslevodopa are prodrugs that undergo enzymatic bioconversion via intrinsic alkaline phosphatase to carbidopa and levodopa, respectively, in vivo .
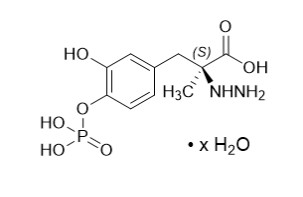
Foscarbidopa, an inhibitor of aromatic amino acid decarboxylation, is a white to light yellow powder, freely soluble in aqueous media, with a molecular weight of 306.21 g/mol anhydrous. It is designated chemically as (2 S )-2-hydrazinyl-3-[3-hydroxy-4-(phosphonooxy)phenyl]-2-methylpropanoic acid trihydrate. Its empirical formula is C 10 H 15 N 2 O 7 P (anhydrous basis), and its structural formula is:
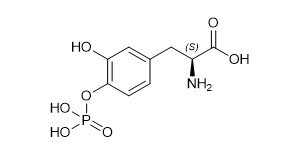
Foslevodopa, an aromatic amino acid, is a white to off-white powder, freely soluble in aqueous media, with a molecular weight of 277.17 g/mol. It is designated chemically as (2 S )-2-amino-3-[3-hydroxy-4-(phosphonooxy)phenyl] propanoic acid. Its empirical formula is C 9 H 12 NO 7 P, and its structural formula is:
VYALEV (foscarbidopa and foslevodopa) injection is a sterile, preservative-free solution for subcutaneous infusion. VYALEV is supplied in a 10 mL single-dose glass vial that contains 120 mg foscarbidopa and 2,400 mg foslevodopa per 10 mL (12 mg foscarbidopa and 240 mg foslevodopa per mL). VYALEV may contain sodium hydroxide and/or hydrochloric acid to adjust the pH to approximately 7.4.
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
VYALEV is a prodrug combination of foscarbidopa (carbidopa-4´-monophosphate) and foslevodopa (levodopa-4´-monophosphate). Foscarbidopa and foslevodopa are converted in vivo to carbidopa and levodopa.
Carbidopa
When levodopa is administered orally, it is rapidly decarboxylated to dopamine in extracerebral tissues so that only a small portion of a given dose is transported unchanged to the central nervous system. Carbidopa inhibits the decarboxylation of peripheral levodopa, making more levodopa available for delivery to the brain.
Levodopa
Levodopa is the metabolic precursor of dopamine, does cross the blood-brain barrier, and presumably is converted to dopamine in the brain. This is thought to be the mechanism whereby levodopa treats symptoms of Parkinson's disease.
12.2 Pharmacodynamics
Because the decarboxylase inhibiting activity is limited to extracerebral tissues, administration of carbidopa with levodopa makes more levodopa available to the brain. The addition of carbidopa to levodopa reduces the peripheral effects (e.g., nausea and vomiting) due to decarboxylation of levodopa; however, carbidopa does not decrease the adverse reactions due to the central effects of levodopa.
12.3 Pharmacokinetics
Absorption
VYALEV is administered directly into the subcutaneous space and is converted to carbidopa and levodopa by alkaline phosphatase. In a phase 1 study in healthy volunteers, carbidopa and levodopa were detectable in plasma within 30 minutes at the first pharmacokinetic (PK) collection point. In healthy volunteer subjects the steady state levodopa was achieved within 2 hours when VYALEV dosing was delivered as a loading dose followed by continuous infusion.
Healthy volunteers were administered VYALEV to different subcutaneous sites (i.e., abdomen, arm, and thigh) using a 3-way crossover design. PK analysis from this study showed that the 3 sites provided comparable carbidopa and levodopa exposure suggesting VYALEV absorption is similar at the different subcutaneous sites.
Effect of Food
VYALEV bypasses the gut, so consumption of food does not change absorption or systemic exposure of carbidopa/levodopa.
Distribution
Both foslevodopa and foscarbidopa have binding to plasma proteins between 24% to 26%.
Levodopa
Levodopa is distributed between erythrocytes and plasma in approximately 1:1 ratio. Levodopa binding to plasma proteins is< 10%. Levodopa is transported into the brain by the carrier mechanism for large neutral amino acids.
Carbidopa
Carbidopa is approximately 36% bound to plasma protein. Carbidopa does not cross the blood-brain barrier.
Metabolism and Excretion
The foscarbidopa and foslevodopa from VYALEV are converted by alkaline phosphatases into carbidopa and levodopa.
Levodopa
Levodopa is mainly eliminated via metabolism by the aromatic amino acid decarboxylase (AAAD) and the catechol-O-methyl-transferase (COMT) enzymes. Other routes of metabolism are transamination and oxidation. The decarboxylation of levodopa to dopamine by AAAD is the major enzymatic pathway when no enzyme inhibitor is co-administered. O-methylation of levodopa by COMT forms 3-O-methyldopa. When administered with carbidopa, the elimination half-life of levodopa is approximately 1.5 hours.
Carbidopa
Carbidopa is metabolized to two main metabolites (α-methyl-3-methoxy-4-hydroxyphenylpropionic acid and α-methyl-3,4-dihydroxyphenylpropionic acid). These 2 metabolites are primarily eliminated in the urine unchanged or as glucuronide conjugates. Unchanged carbidopa accounts for 30% of the total urinary excretion. The elimination half-life of carbidopa is approximately 2 hours.
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
In rat, oral administration of carbidopa and levodopa for two years resulted in no evidence of carcinogenicity. VYALEV contains hydrazine, a degradation product of carbidopa. In published studies, hydrazine has been demonstrated to be carcinogenic in multiple animal species. Increases in liver (adenoma, carcinoma) and lung (adenoma, adenocarcinoma) tumors have been reported with oral administration of hydrazine in mouse, rat, and hamster.
Mutagenesis
Carbidopa was positive in the in vitro Ames test, in the presence and absence of metabolic activation, and the in vitro mouse lymphoma tk assay in the absence of metabolic activation but was negative in the in vivo mouse micronucleus assay.
In published studies, hydrazine was reported to be positive in in vitro genotoxicity (Ames, chromosomal aberration in mammalian cells, and mouse lymphoma tk ) assays and in the in vivo mouse micronucleus assay.
Impairment of Fertility
In reproduction studies, no effects on fertility were observed in rats receiving carbidopa and levodopa.
14 CLINICAL STUDIES
The efficacy of VYALEV was established in a 12-week, randomized, double-blind, double-dummy, active-controlled, multicenter study (Study 1; NCT04380142) in patients with advanced Parkinson’s disease (PD). Study 1 enrolled patients who were responsive to levodopa treatment, had motor fluctuations inadequately controlled by their current medications, and who experienced a minimum of 2.5 hours of “Off” time per day as assessed by PD diaries. A total of 141 patients were randomized in 1:1 ratio and received either 24-hour/day continuous subcutaneous administration of VYALEV plus oral placebo capsules (N=74) or 24-hour/day continuous subcutaneous administration of placebo solution plus oral encapsulated carbidopa-levodopa immediate-release (IR) tablets (N=67).
Patients had a mean age of 66.4 years and a mean disease duration of 8.6 years. Most (93%) of the patients were white, 2% were Asian, 3% Black and 70% of the patients were male. At baseline, approximately 74% of patients in the VYALEV group and 66% of patients in the oral IR carbidopa-levodopa group were taking at least 1 or more classes of PD medications other than carbidopa-levodopa.
The primary clinical outcome measure was the mean change from baseline to Week 12 in the total daily mean “On” time without troublesome dyskinesia (defined as "On" time without dyskinesia plus "On" time with non-troublesome dyskinesia) based on PD diary.
The key secondary clinical outcome measure was the mean change from baseline to Week 12 in the total daily mean “Off” time. The “On” and “Off” time were normalized to a daily 16-hour awake period. Daily normalized "Off" and "On" times are averaged over valid PD diary days for each visit to obtain the average daily normalized times. VYALEV demonstrated statistically significant improvements from baseline to Week 12 in "On" time without troublesome dyskinesia compared with the oral IR carbidopa-levodopa group (p=0.0083; Table 3). VYALEV also demonstrated statistically significant improvements from baseline to Week 12 in “Off” time compared with the oral IR carbidopa-levodopa group (p=0.0054; Table 3).
| Oral IR carbidopa-levodopa b (N=67) | VYALEV (N=73) | |
| Primary Measure | ||
| “On” time without troublesome dyskinesia (hours) a | ||
| Baseline Mean (SD) | 9.49 (2.62) | 9.20 (2.42) |
| Change from Baseline to Endpoint Week 12 Mean (SD) | 0.85 (3.46) | 3.36 (3.62) |
| LS Mean (SE) of Change | 0.97 (0.50) | 2.72 (0.52) |
| LS Mean (SE) of Difference | 1.75 (0.65) | |
| P value | 0.0083 | |
| Secondary Measure | ||
| “Off” time (hours) a | ||
| Baseline Mean (SD) | 5.91 (1.88) | 6.34 (2.27) |
| Change from Baseline to Endpoint Week 12 Mean (SD) | -0.93 (3.31) | -3.41 (3.76) |
| LS Mean (SE) of Change | -0.96 (0.49) | -2.75 (0.50) |
| LS Mean (SE) of Difference | -1.79 (0.63) | |
| P value | 0.0054 | |
| LS = least squares; SD = standard deviation; SE = standard error. a Derived from Parkinson’s Disease (PD) diary. b Oral immediate release carbidopa-levodopa tablets. | ||
Figure 1 shows results over time according to treatment for the efficacy variable (mean change from baseline to week 12 in the total daily mean normalized “On” time without troublesome dyskinesia based on PD diary).
Figure 1 . LS Mean Change (±SE) from Baseline in “On” Time Without Troublesome Dyskinesia over 12 Weeks
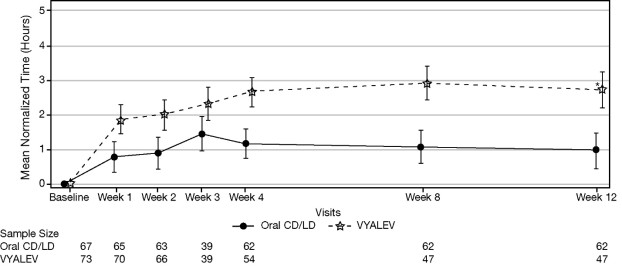
• p ≤ 0.01. P value reflects comparison between treatment groups
CD = carbidopa; LD = levodopa; LS = least squares; SE = standard error
Note - Week 3 was an optional visit.
Figure 2 shows results over time according to treatment for the efficacy variable (mean change from baseline to week 12 in the total daily mean normalized “Off” time based on PD diary).
Figure 2 . LS Mean Change (±SE) from Baseline in “Off” Time over 12 Weeks
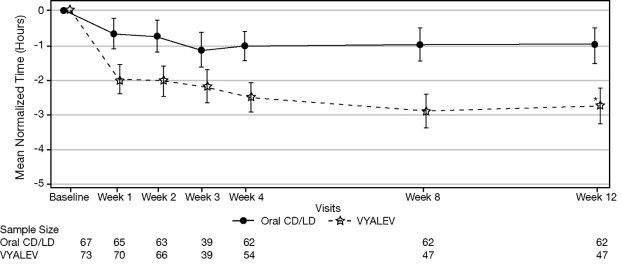
• p ≤ 0.01. P value reflects comparison between treatment groups
CD = carbidopa; LD = levodopa; LS = least squares; SE = standard error
Note - Week 3 was an optional visit.
16 HOW SUPPLIED/STORAGE AND HANDLING
16.1 How Supplied
VYALEV injection contains 120 mg foscarbidopa and 2,400 mg foslevodopa per 10 mL (12 mg foscarbidopa and 240 mg foslevodopa per mL) and is a colorless to yellow to brown (may have a purple or red tint), and clear to slightly opalescent solution. Each single-dose glass vial is filled with approximately 10 mL of solution and is fitted with a grey rubber stopper, aluminum crimp cap, and turquoise plastic flip-off cap.
The rubber stopper on the vial does not contain natural rubber latex.
Carton of 7 VYALEV vials: NDC 0074-0501-01
The VYAFUSER pump used to administer VYALEV (foscarbidopa and foslevodopa) is provided separately.
16.2 Storage and Handling
- Keep the medication vials in the outer carton to protect the vials from breaking.
- Store VYALEV refrigerated at 2°C to 8°C (36°F to 46°F).
- VYALEV may be stored at room temperature up to a maximum of 30°C (86°F) for a single period of up to 28 days.
- Once VYALEV has been stored at room temperature, do not return the product to the refrigerator.
- If stored at room temperature, discard VYALEV if not used within 28 days.
- Do not freeze.
- Do not shake.
INSTRUCTIONS FOR USE
VYALEV ™ [ vye-uh-lev ]
(foscarbidopa and foslevodopa)
injection , for subcutaneous use
Read this Instructions for Use before you start using VYALEV and each time you get a new refill. There may be new information. This information does not take the place of talking to your healthcare provider about your medical condition or your treatment. This Instructions for Use contains information on how to prepare VYALEV.
Before using VYALEV :
- Only use VYALEV by yourself after you have been shown the right way to use VYALEV and the delivery system (see the Patient Instructions for Use of VYAFUSER Pump ).
- Call your healthcare provider or call (866) 4-VYALEV or (866) 489-2538 for help or go to www.VYALEV.com.
How should I store VYALEV ?
- Keep the vials in the outer carton to protect the vials from breaking.
- Store VYALEV in the refrigerator between 36°F to 46°F (2°C to 8°C).
- Record the date when VYALEV is first removed from the refrigerator in the space provided on the carton.
- VYALEV may be stored at room temperature up to a maximum of 86°F (30°C) for a single period of up to 28 days.
- After VYALEV has been stored at room temperature, do not return the medicine to the refrigerator.
- If stored at room temperature, throw away (dispose of) VYALEV if not used within 28-days.
- Do not freeze.
- Do not shake.
- If VYALEV is stored in the refrigerator, take one VYALEV vial out of the carton and out of the refrigerator 30 minutes before use. Use the VYALEV at room temperature or you may not get the right amount of the medicine.
Keep VYALEV and all medicines out of the reach of children.
Important Information :
Solution
- The VYALEV s olution color may have changes in color and does not affect how well the medicine works . It may be colorless, or may have different colors anywhere between light yellow and brown, possibly with purple or red tint. It also may become darker while in the syringe.
- If refrigerated before use, remove the solution vial from refrigerated storage and allow to sit at room temperature out of direct sunlight for 30 minutes.
- Do not add any other liquid (dilute) the VYALEV solution or fill the syringe with anything other than what your healthcare provider prescribed.
- Do not use VYALEV if the expiration date (EXP:) shown on the carton and vial has passed.
Disposable Parts (Vial Adapter and Syringe)
- A new vial adapter must be used with each new vial of VYALEV.
- Do not use the VYALEV solution if it has been in the syringe for more than 24 hours.
A. T ransfer VYALEV from Solution V ial to Syringe
1.000000000000000e+00 Make sure your work space is clean.
Note: This will help to avoid contamination.
2.000000000000000e+00 Gather supplies, including (see Figure A ) :
- Solution vial
- Syringe (not included in VYALEV carton)
- Vial adapter (not included in VYALEV carton)
- Alcohol pads (not included in VYALEV carton)
Figure A
| Syringe | Solution | Vial | Alcohol |
| Vial | Adapter | Pads |
3.000000000000000e+00 Check over the parts for expiration and for any packaging damage. This should include:
• Solution vial (see Figure B)
• Vial adapter
• Syringe
- Check if the solution is the VYALEV solution prescribed by your healthcare provider.
- Do not use the VYALEV solution, vial adapter, or syringe if it is expired.
- Do not use any parts if their sterile packaging has been damaged before use.
Note: The product packaging for the infusion set, vial adapter, and syringe show that they are sterile and how they were sterilized.

Figure B
4.000000000000000e+00 Check over the contents of the VYALEV vial (see Figure C ) . Check the following:
- No cloudiness of the liquid.
- No particles seen in the liquid.
Figure C
- Do not use if the VYALEV solution is cloudy or contains flakes or particles.
Note: If refrigerated before use, remove the VYALEV solution vial from refrigerated storage and allow to sit at room temperature, out of direct sunlight, for 30 minutes.
- If refrigerated, do not warm VYALEV (in solution vial or syringe) in any way other than letting it warm at room temperature. For example, do not warm in a microwave or in hot water.
5.000000000000000e+00 Wash your hands with soap and water and dry them (see Figure D ).
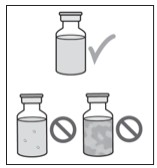
Figure D
6.000000000000000e+00 Prepare VYALEV solution v ial .
a. Remove the vial cap (see Figure E).
Figure E
b. Wipe the top of the solution vial with an alcohol pad and allow to dry (see Figure F).
Note: This will help to avoid contamination.
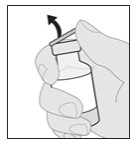
Figure F
7.000000000000000e+00 Attach the v ial a dapter to the s olution v ial .
Read your V ial Adapter Instructions f or Use for detailed steps.
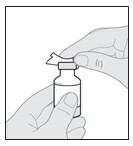
Vial Adapter
8.000000000000000e+00 Prepare the s yringe .
- To decrease the risk of infections, do not let the tip of any disposable parts come into contact with any unclean surfaces. If the tip of the vial adapter or syringe comes into contact with an unclean surface, throw (discard) it away and get a new one.
- Get a new syringe and remove it from its packaging.
- Push up on the rubber plunger to fully push out all air (see Figure G).

Figure G
9.000000000000000e+00 While holding the v ial a dapter firmly, attach the s yringe to the v ial a dapter by pushing and then screwing it into place (see Figure H ).
- Do not overtighten.
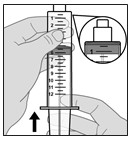
Figure H
1.000000000000000e+01 Hold the s yringe pointing straight up ( vertically ) with the s olution v ial above the s yringe (see Figure I ).
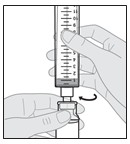
Figure I
1.100000000000000e+01 Withdraw all of the medicine from the vial into the s yringe .
- While holding the syringe firmly in one hand, pull down the plunger rod with the other hand to withdraw all of the medicine in the solution vial into the syringe to around the 12 mL mark, or until you see air at the tip of the syringe (see Figure J).
Note: It is important to hold the syringe pointing straight up.
Note: Always withdraw all of the medicine in the solution vial into the syringe.
Note: You will see air (head space) at the tip of the syringe.
Figure J
1.200000000000000e+01 Check for air bubbles.
- If there are large air bubbles, they must be removed. Air may affect if you get the right amount of medicine (your dose).
- As seen in Figure K, small bubbles are okay and the air at the tip of the syringe (the head space) is expected.
- As seen in Figure L, larger air bubbles are not okay. While the air at the top of the syringe (the head space) is expected, the larger bubbles are not.
- If you see large air bubbles , continue with Section B: Manually Remove Air Bubbles .
- If you see small air bubbles or do not see any air bubbles , skip the next section and go to Section C: Remove (Purge) Air from Syringe .
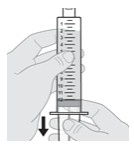
Figure K
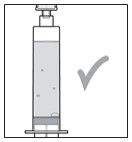
Figure L
B. Manually Remove Air Bubbles
1.300000000000000e+01 Bring the bubbles together into a single air bubble.
a. Slowly and gently rotate the syringe and tilt it back and forth (see Figure M).
- Do not shake or tap the syringe to remove the air bubbles.
Figure M
Note: If there are still air bubbles, bring the bubbles together by gently rotating the syringe end over end (see Figure N).
b. When the large air bubbles are brought together into a single air bubble, continue with the next step.
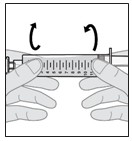
Figure N
C. Remove ( Purge ) Air from Syringe
1.400000000000000e+01 Push air out of the s yringe .
- With the solution vial still attached, point the syringe upward.
- Slowly push the air out of the syringe and into the vial (see Figure O).
- Continue pushing until all of the air is pushed out the syringe and into the solution vial and you can see that there is solution in the syringe tip.
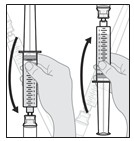
Figure O
Note: Some resistance will be felt as the air is pushed back into the solution vial.
Note: If the syringe is tilted slightly and not pointing straight up, you may see a small air bubble in the corner (see Figure P). This is okay.
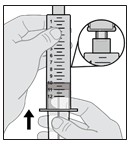
Figure P
1.500000000000000e+01 Turn the syringe and s olution v ial upside down (invert) so that the s olution v ial is upright on the table (see Figure Q ).
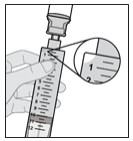
Figure Q
1.600000000000000e+01 Disconnect the s yringe from the v ial a dapter .
a. Hold the vial adapter firmly with one hand and the barrel of the syringe with the other.
b. Unscrew the syringe from the vial adapter (see Figure R).
Note: When disconnecting the syringe from the Vial, do not push the plunger or else the solution will leak.
c. Place the syringe on a clean surface, making sure the syringe tip does not contact an unclean surface.
- To decrease the risk of infections, do not let the tip of any disposable part come into contact with any unclean surfaces. If the tip of the vial adapter or syringe comes into contact with an unclean surface, throw it away (discard) and get a new one.
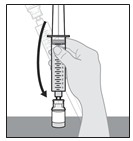
Figure R
1.700000000000000e+01 Your s yringe is now ready for use. Refer to your pump manual ( Patient Instructions for Use of VYAFUSER Pump ) for the next steps .
Patient Instructions for Use
of VYAFUSER Pump
D. Dispos ing of VYALEV
1.800000000000000e+01 Used s olution v ials with the v ial a dapters still attached should be thrown away ( disposed of ) in accordance with local regulations or as directed by your h ealthcare p ro vider .
This Instructions for Use for VYALEV (foscarbidopa and foslevodopa) is to be used with the VYAFUSER pump (see the Patient Instructions for Use of VYAFUSER Pump ).
Manufactured for: AbbVie Inc. North Chicago, IL 60064 U.S.A. VYALEV and its design are trademarks of AbbVie AB. VYAFUSER™ is a trademark of AbbVie AB. ©2024 AbbVie. All rights reserved. This Instructions for Use has been approved by the U.S. Food and Drug Administration. Approved: 10/2024 20065527
12.1 Mechanism of Action
VYALEV is a prodrug combination of foscarbidopa (carbidopa-4´-monophosphate) and foslevodopa (levodopa-4´-monophosphate). Foscarbidopa and foslevodopa are converted in vivo to carbidopa and levodopa.
Carbidopa
When levodopa is administered orally, it is rapidly decarboxylated to dopamine in extracerebral tissues so that only a small portion of a given dose is transported unchanged to the central nervous system. Carbidopa inhibits the decarboxylation of peripheral levodopa, making more levodopa available for delivery to the brain.
Levodopa
Levodopa is the metabolic precursor of dopamine, does cross the blood-brain barrier, and presumably is converted to dopamine in the brain. This is thought to be the mechanism whereby levodopa treats symptoms of Parkinson's disease.